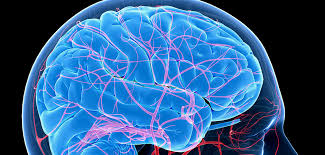
TUSTIN, Calif.–(BUSINESS WIRE)– The Aquilion ONE™ / GENESIS Edition, capable of imaging the entire brain with a single 640-slice* rotation, covering 16 cm, gives clinicians the potential to diagnose and evaluate acute stroke quickly. The system can also capture both anatomy and function, which provides physicians with additional tools to […]
Neuro
MRI Interventions, Inc. Reports 34% Year-Over-Year Increase in Functional Neurology Revenues, 50% increase in Capital Sales
Record 629 ClearPoint® Procedures Performed in 2017 New Initiatives Position for Further Growth in 2018 IRVINE, Calif., March 15, 2018 (GLOBE NEWSWIRE) — MRI Interventions, Inc. (OTCQB:MRIC) (the “Company”) today announced financial results for the fourth quarter and full year ended December 31, 2017. Full-Year and Fourth Quarter 2017 Highlights Total […]
The global neurovascular devices market is expected to grow at a CAGR of 10.3%
LONDON, March 14, 2018 /PRNewswire/ — Download the full report: https://www.reportbuyer.com/product/5351730 Report Details The global neurovascular devices market is expected to grow at a CAGR of 10.3% in the first half of the forecast period. In 2016, the Cerebral Embolization and Aneurysm Coiling Devices segment held the largest share in the global neurovascular […]
Neurovascular Embolization Coils: Healthcare Provider Letter – Potential for Increased Image Artifact When Using Magnetic Resonance Angiography For Follow-Up
ISSUE: The FDA is providing information about the potential for increased image artifact associated with Magnetic Resonance Angiography (MRA) imaging for patient follow-up of certain post neurovascular embolization coil procedures. Neurovascular embolization coils are used in the treatment of brain aneurysms and other vascular abnormalities. These coils are prescription devices […]
Perflow Medical Closes $12 Million in Financing to Treat Complex Neurovascular Disorders
TEL AVIV, Israel, Feb. 15, 2018 /PRNewswire/ — Perflow Medical, an Israeli-based medical device company that develops innovative tools for neurovascular interventions, today announced that the company has closed $12 million in financing. The syndicate included existing investors, two new international investors, and an unnamed strategic investor. Following a recent launch in Europe, Perflow stated that […]
FDA Permits Marketing of Clinical Decision Support Software for Alerting Providers of a Potential Stroke in Patients
SILVER SPRING, Md., Feb. 13, 2018 /PRNewswire-USNewswire/ — Today, the U.S. Food and Drug Administration permitted marketing of the Viz.AI Contact application, a type of clinical decision support software designed to analyze computed tomography (CT) results that may notify providers of a potential stroke in their patients. A stroke is […]
Stryker’s WEAVE Trial Demonstrates Positive Results of Endovascular Treatment for Patients With Intracranial Atherosclerotic Disease
KALAMAZOO, Michigan, USA, Jan. 26, 2018 /PRNewswire/ — Results from the Wingspan® StEnt System Post MArket SurVEillance Study (WEAVE™ Trial) were presented yesterday at the International Stroke Conference, providing compelling evidence that more patients suffering from intracranial atherosclerotic disease (ICAD) may benefit from endovascular treatment with the Wingspan Stent System. Results from […]
INDEPENDENT STUDY REAFFIRMS PENUMBRA ASPIRATION AS FRONTLINE THROMBECTOMY APPROACH FOR ACUTE ISCHEMIC STROKE
Results of COMPASS Trial Presented at International Stroke Conference LOS ANGELES – Jan. 25, 2018 – Penumbra, Inc. (NYSE: PEN), a global healthcare company focused on innovative therapies, today announced the presentation of the results of the COMPASS Trial, an independent, prospective, multi-center randomized trial that showed the use of […]
NEW STUDY SHOWS USE OF EMBOTRAP® DEVICE CLOT RETRIEVER DURING TREATMENT OF STROKE YIELDS POSITIVE RATES OF REVASCULARIZATION
ARISE II Clinical Trial Results Presented at International Stroke Conference LOS ANGELES – Jan. 25, 2018 – EMBOTRAP® Device, a device designed to remove clots from the brain following an ischemic stroke, demonstrated high success rates in restoring blood flow and patients achieved high rates of functional independence, according to […]
Stryker’s Wingspan Stent Impresses in Post-Market Study
KALAMAZOO, Michigan, USA, Jan. 25, 2018 /PRNewswire/ — Yesterday the American Heart Association and American Stroke Association updated the ischemic stroke guidelines. The new recommendation expanded the treatment window from six to 16 hours based on overwhelming clinical evidence from the Stryker-sponsored DAWN Trial and confirmed by DEFUSE 3. The guidelines state that […]