PARIS–(BUSINESS WIRE)–TISSIUM, a privately-owned life science company developing fully synthetic, biomorphic programmable polymers, announced today the publication of its manuscript detailing preclinical and clinical evaluations of SETALIUMTM Vascular Sealant, the company’s first synthetic, biodegradable, on-demand, light-activated sealant. The results of these evaluations, published in The Journal of Cardiovascular Surgery on May 10th, 2019, […]
Peripheral/Endo
Gore Receives FDA Approval for the GORE® TAG® Conformable Thoracic Stent Graft with ACTIVE CONTROL
FLAGSTAFF, Ariz.–(BUSINESS WIRE)–W. L. Gore & Associates, Inc. (Gore) today announced that the U.S. Food and Drug Administration (FDA) has granted regulatory approval for commercial distribution for the GORE® TAG® Conformable Thoracic Stent Graft with ACTIVE CONTROL System, a unique Thoracic Endovascular Aortic Repair (TEVAR) solution combining new levels of control […]
Teleflex to participate at the European Association for Percutaneous Cardiovascular Interventions Course (EuroPCR) 2019 with its complex PCI product portfolio and the newly acquired MANTA™ Vascular Closure Device
WAYNE, Pa., May 15, 2019 (GLOBE NEWSWIRE) — Teleflex Incorporated (NYSE: TFX), a leading global provider of medical technologies for critical care, urology and surgery, coronary and peripheral interventions, will showcase its complex PCI product portfolio, the Arrow® AC3 Optimus™ Intra-Aortic Balloon Pump (IABP) and the newly acquired MANTA™ Vascular Closure […]
Eximo Medical Ltd. Announces the First Successful Use of the B-Laser™ Atherectomy System in Crossing Chronic Total Occlusion (CTO) without a guidewire
REHOVOT, Israel–(BUSINESS WIRE)–Eximo Medical Ltd., announced today the first successful use of the B-Laser™Atherectomy System in a “step-by-step” approach (crossing a non-crossable CTO cap without guidewire) for the therapy of patients afflicted by highly complex, arterial occlusive peripheral arterial disease (PAD) as well as the first ever utilization of On-line Photo-Acoustic Monitoring […]
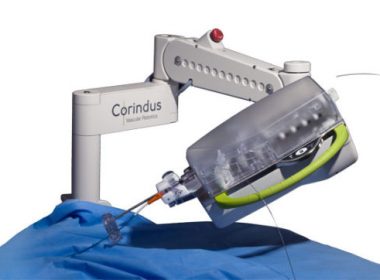
Corindus Equips the World’s First Fully Robotic Catheterization Laboratory
WALTHAM, Mass.–(BUSINESS WIRE)–Corindus Vascular Robotics, Inc. (“Corindus” or the “Company”) (NYSE American: CVRS), a leading developer of precision vascular robotics, announced today that Chesapeake Regional Healthcare in Chesapeake, VA has equipped both of its catheterization labs at Chesapeake Regional Medical Center with CorPath GRX Vascular Robotic Systems, making it the first hospital […]
Inari Medical Announces Publication of FLARE IDE Study Results
IRVINE, Calif., May 7, 2019 /PRNewswire/ — Inari Medical, Inc. announced today the publication of its 106-patient prospective multicenter FlowTriever Mechanical Pulmonary Embolectomy (FLARE) study for the treatment of intermediate-risk pulmonary embolism (PE). The study was published in JACC: Cardiovascular Interventions. FLARE was conducted under the direction of co-principal investigators, Dr. Kenneth Rosenfield, Section Head […]
Boston Scientific Receives U.S. FDA Approval for The VICI VENOUS STENT™ System
MARLBOROUGH, Mass., May 6, 2019 /PRNewswire/ — Boston Scientific (NYSE: BSX) announced today that the U.S Food and Drug Administration (FDA) has approved the VICI VENOUS STENT™ System for the treatment of iliofemoral venous obstructive disease, which occurs when the flow of blood through the veins located deep in the pelvic region becomes blocked […]
Intact Vascular Announces US Launch of the Tack Endovascular System® and First Commercial Use
WAYNE, Pa.–(BUSINESS WIRE)–Intact Vascular, Inc., a developer of medical devices for minimally invasive peripheral vascular procedures, today announced the first commercial use of its Tack Endovascular System in multiple sites across the United States. Recently FDA-approved for above-the-knee interventions, the first-of-its-kind dissection repair device is implanted post-angioplasty to resolve dissections […]
NuCryo Vascular Announces Series B Financing and the Launch of its Extended PolarCath Catheter
SAN JOSE, Calif.–(BUSINESS WIRE)–NuCryo Vascular, the manufacturer and marketer of the PolarCath Balloon Dilatation System, announced today that the company is raising a Series B round of financing and is launching its Extended PolarCath Balloon Dilatation Catheter. The Series B financing will provide the necessary working capital to support the […]
Surmodics Provides Update Regarding TRANSCEND Clinical Trial
EDEN PRAIRIE, Minn.–(BUSINESS WIRE)–Surmodics, Inc. (NASDAQ: SRDX), a leading provider of medical device and in vitro diagnostic technologies to the healthcare industry, has resumed patient enrollment into its TRANSCEND clinical trial and is nearly 75 percent of the way to its goal of 446 randomized patients. Patient enrollment in TRANSCEND, the […]