BEAUFORT, S.C.–(BUSINESS WIRE)–A new analysis published by THE SAGE GROUP concludes that ALI represents a substantial market opportunity for pharmaceutical and interventional therapies. “Although the number of patients currently treated for an acute occlusion in the lower limbs is relatively small, the total market is surprisingly large,” stated Mary L. […]
Peripheral/Endo
Hemostemix Provides Highlights of 2018 Accomplishments
CALGARY, Alberta, Jan. 17, 2019 (GLOBE NEWSWIRE) — Hemostemix Inc. (“Hemostemix” or the “Company”) (TSX VENTURE: HEM; OTCQB: HMTXF) is pleased to provide the following corporate update on accomplishments and milestones that it has achieved in 2018. The past twelve months has seen a transformation in the Company, with a […]
Chocolate Touch® Drug-Coated Balloon IDE Study Expanded To Include Atherectomy
PLEASANTON, Calif., Jan. 16, 2019 /PRNewswire/ — QT Vascular Ltd., (the “Company” or “QT Vascular“, and together with its subsidiaries, the “Group“), is a global company engaged in the design, assembly and distribution of advanced therapeutic solutions for the minimally invasive treatment of vascular disease. The Company is pleased to announce that […]
Euphrates Vascular announces FDA clearance of the ICHOR Reperfusion System
St. Louis, Missouri – Euphrates Vascular (www.euphratesvascular.com), a company focused on the treatment of vascular occlusions, recently received 510(k) clearance for sale of its ICHOR Percutaneous Reperfusion System. ICHOR is a Percutaneous Reperfusion System designed to treat organized thrombus and embolic events in the peripheral vasculature. ICHOR is a “one size fits […]
InspireMD Announces the Distribution of CGuard™ Embolic Prevention System in South Africa
TEL AVIV, Israel, Jan. 14, 2019 (GLOBE NEWSWIRE) — InspireMD, Inc. (NYSE American: NSPR), developer of the CGuard™ Embolic Prevention System (EPS) for the prevention of stroke caused by the treatment of carotid artery disease, today announced that its novel CGuard EPS will be distributed by its local distribution partner, […]
Intact Vascular Announces Launch of the Tack Endovascular System® in the EU and First Commercial Use in Germany
WAYNE, Pa.–(BUSINESS WIRE)–Intact Vascular, Inc., a developer of medical devices for minimally invasive peripheral vascular procedures, today announced the first commercial use of its Tack Endovascular System® in multiple hospitals within Germany. A novel therapy for dissection repair following balloon angioplasty, the Tack® implant is a first-of-its kind device for patients with peripheral […]
Access Scientific announces the first non-coated intravascular catheter material–ChronoFlex®C with BioGUARD™–proven in vitro to inhibit bacterial attachment and biofilm formation
SAN DIEGO, Jan. 7, 2019 /PRNewswire/ — Access Scientific, LLC today announced publication, in the journal Medical Devices: Evidence & Research, of a groundbreaking study proving the superiority of POWERWAND™ catheter material, Chronoflex®C with BioGUARD™ (as compared with the standard polyurethane) with respect to bacterial attachment and biofilm formation. Utilizing state-of-the-art fluorescent microscopy, the […]
Endologix Takes Decisive Action to Optimize Patient Outcomes by Ensuring Nellix System Used Only within Current Indications
IRVINE, Calif.–(BUSINESS WIRE)–Endologix® Inc. (Nasdaq: ELGX) (“Endologix” or the “Company”), a developer and marketer of innovative treatments for aortic disorders, announced today that in order to ensure optimal outcomes for patients, unrestricted sales and use of the Nellix System will cease immediately, and the product will only be available for […]
Hemostemix Announces Two Additional Sites Enrolled in Phase II Clinical Trial
CALGARY, Alberta, Jan. 03, 2019 (GLOBE NEWSWIRE) — Hemostemix Inc. (“Hemostemix” or the “Company”) (TSX VENTURE: HEM; OTCQB:HMTXF) is pleased to announce that it has received all approvals from two new medical centers to be clinical trial sites for the Company’s Phase II clinical trial for critical limb ischemia (“CLI”). […]
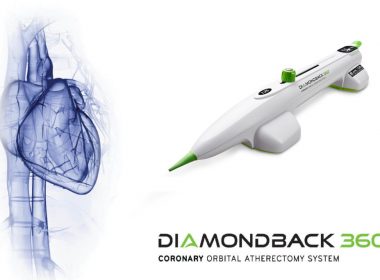
Cardiovascular Systems, Inc. Receives Approval for the Diamondback 360® Coronary Orbital Atherectomy System with Classic Crown and New ViperWire Advance® FlexTip Guidewire in Japan
ST. PAUL, Minn.–(BUSINESS WIRE)–Cardiovascular Systems, Inc. (CSI®) (NASDAQ: CSII), a medical device company developing and commercializing innovative interventional treatment systems for patients with peripheral and coronary artery disease, announced today that Japan’s Ministry of Health, Labor and Welfare (MHLW) has approved the Diamondback 360®Coronary Orbital Atherectomy System (OAS) with Classic Crown […]