Next-generation deliverability and control for peripheral embolization — available September 8, 2025 IRVINE, Calif., Sept. 8, 2025 /PRNewswire/ — ASAHI INTECC today announced the U.S. commercial launch of Veloute™ and Tellus™. These medical devices are indicated for angiography and…
Peripheral/Endo
FLEX Vessel Prep™ System Spotlighted for Clinical Outcomes, Staff Efficiency Gains, and Real-World Physician Experience in the Journal of Vascular Access, Vascular News, and Endovascular Today
MINNEAPOLIS, Sept. 4, 2025 /PRNewswire/ — VentureMed Group, Inc., a privately held leader in medical device innovations for arteriovenous (AV) access and peripheral arterial disease (PAD), was recently featured in a published study on the outcomes of FLEX VP + DCB in the Journal of…
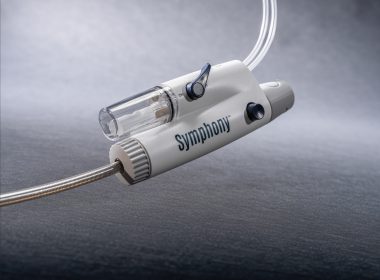
Imperative Care Announces FDA Clearance of Symphony Thrombectomy System, the First Large-Bore Continuous Vacuum System for Treatment of Pulmonary Embolism
CAMPBELL, Calif.–(BUSINESS WIRE)–Imperative Care, Inc. today announced U.S. Food and Drug Administration (FDA) 510(k) clearance of its Symphony® Thrombectomy System to treat pulmonary embolism (PE), a life-threatening condition caused by blood clots blocking an artery in the lungs. This clearance expands the use of Symphony – previously for the treatment of […]
Phraxis Announces First-Ever Commercial Case of EndoForce™ Anastomotic Connector at Spartanburg Regional Medical Center
MINNEAPOLIS, Aug. 29, 2025 /PRNewswire/ — Phraxis, Inc., a leader in vascular access innovation, today announced the successful completion of the first commercial case using the EndoForce™ Anastomotic Connector, marking a historic milestone in the treatment of dialysis patients worldwide….
Venova Medical Receives FDA IDE Approval for the VENOS-3 Pivotal Study of the Velocity™ Percutaneous AVF System
Venova Medical Receives FDA IDE Approval for the VENOS-3 Study LOS GATOS, Calif., Aug. 21, 2025 /PRNewswire-PRWeb/ — Venova Medical, a privately held company developing a next generation technology for the creation of percutaneous arteriovenous fistulas (pAVF) for hemodialysis access, today announced approval from the US Food and Drug Administration (FDA) to […]
Instylla Gains U.S. FDA Premarket Approval for Embrace™ Hydrogel Embolic System
First and Only Liquid Embolic Approved for Hypervascular Tumor Embolization Supported by a Prospective, Randomized, Controlled, Clinical Trial BEDFORD, Mass., Aug. 7, 2025 /PRNewswire/ — Instylla, Inc., a privately held company developing novel resorbable embolics for peripheral vascular…
Instylla Appoints MedTech Leader Sean Boyle as Chief Executive Officer
BEDFORD, Mass., Aug. 5, 2025 /PRNewswire/ — Instylla, Inc., a privately held clinical-stage company developing solutions for peripheral vascular embolization, has announced the appointment of Sean Boyle as Chief Executive Officer. I’m honored to join Instylla at such a crucial moment in…
Inari Medical, now part of Stryker, launches the all-new InThrill® Thrombectomy System, designed to address arteriovenous access and small vessel thrombus cases
PORTAGE, Mich., July 29, 2025 /PRNewswire/ — Inari Medical, now part of Stryker (NYSE: SYK), a global leader in medical technologies, announced the launch of its next-generation InThrill Thrombectomy System, the first and only purpose-built small vessel and arteriovenous (AV) access…
AngioDynamics Enrolls First Patient in AMBITION BTK Trial Advancing Treatment for Critical Limb Ischemia
Trial Investigates Use of Auryon Atherectomy System in Cases with Challenging Below-the-Knee Blockages LATHAM, N.Y.–(BUSINESS WIRE)–AngioDynamics, Inc. (NASDAQ: ANGO), a medical technology company focused on restoring healthy blood flow in the body’s vascular system, expanding cancer treatment options and improving patient quality of life, today announced enrollment of the first […]
Embolx Launches Sniper G3: Revolutionizing Embolization with Enhanced Efficiency, Ease of Use, and Extended Shelf Life
SUNNYVALE, Calif., July 28, 2025 /PRNewswire/ — Embolx, a leader in advanced microcatheters for targeted embolization procedures, today announced the release of its next-generation Sniper G3 Balloon Occlusion Microcatheter. This third iteration of the innovative device delivers…