EchoSolv AS deployed into Mount Sinai Health System, a leading US health system in New YorkMount Sinai Health System comprises seven hospitals, over 400 outpatient practices and +3,760 beds Mount Sinai Fuster Heart Hospital at The Mount Sinai Hospital is ranked No. 1 in New York and No. 2 in the nation for Cardiology, Heart & Vascular Surgery in U.S. News & World Report’s 2025–26 Best Hospitals rankings Mount Sinai Health System has a large echocardiography footprint and top-tier cardiovascular programAgreement represents another milestone in EIQ’s US commercial strategy to deploy in leading hospital systemsEIQ continues to engage with prospective US hospitals regarding potential EchoSolv AS opportunities SYDNEY, April 30, 2026 (GLOBE NEWSWIRE) — AI and Medical Technology company Echo IQ (“the Company”) (ASX: EIQ) is pleased to advise it has deployed its AI-based decision support software for the assessment of severe aortic stenosis, EchoSolv™ AS, with Mount Sinai Health System in New York, USA. “We are proud to work with Mount Sinai Health System, an institution recognised for excellence in cardiovascular care, clinical innovation, and leadership in medicine,” said Nick Lubbers, US President and General Manager for Echo IQ. “This deployment is an important step in our US commercial growth and underscores increasing interest in solutions that can support cardiologists within routine echocardiography workflows. We believe EchoSolv AS offers a practical, measurement-based second look that can help physicians assess severe aortic stenosis more consistently and look forward to working with The Mount Sinai Health System to demonstrate its utility at scale. “Mount Sinai is the kind of institution that helps set the standard for clinical excellence and innovation. For Echo IQ, this engagement represents more than a single large-scale deployment. It reflects our broader growth trajectory as we continue building partnerships with leading hospitals and expanding access to AI-enabled decision support in cardiology,” concluded Lubbers. Stamatios Lerakis MD, PhD*, Director of the Cardiovascular Imaging Program and Imaging for Structural and Valve Interventions for Mount Sinai Health System, said: “We are excited about the opportunity to bring EchoSolv AS into our echocardiography workflow for the evaluation of patients with aortic stenosis. This product can highlight patients with aortic stenosis that can be otherwise under-diagnosed, remain untreated and as a result, not benefit from the tremendous therapeutic advancements in the space of aortic valve diseases. I see this as an adjunct for the echocardiographer who will always have the final say on the interpretation! This product will only strengthen patient care.” The deployment marks a significant milestone for Echo IQ and reflects the Company’s continued growth as health systems and cardiology practices seek practical AI products that can be integrated into existing clinical workflows. Collaboration with a leading medical system such as Mount Sinai Health System also supports the Company’s stated strategy of expanding EchoSolv AS adoption at prominent cardiovascular institutions. Echo IQ views the deployment as an important milestone in not only building commercial traction but also providing future research opportunities as the Company pursues new product development opportunities. The Company continues to engage with prospective US healthcare providers regarding potential EchoSolv AS opportunities and will provide updates as appropriate. Mount Sinai Health System comprises seven hospitals, more than 400 outpatient practices and a globally recognised medical school, the Icahn School of Medicine at Mount Sinai. Specifically, Mount Sinai Fuster Heart Hospital at The Mount Sinai Hospital is ranked No. 1 in New York and No. 2 in the nation for Cardiology, Heart & Vascular Surgery in U.S. News & World Report’s 2025–26 Best Hospitals rankings. The system services millions of patient interactions annually, encompasses over 3,760 beds across campuses, and is consistently ranked among the top hospitals in the US across multiple specialties, including cardiology and heart surgery. Its scale, research pedigree and clinical leadership position it as a high-volume, high-complexity healthcare network at the forefront of adopting advanced digital health and AI-enabled technologies. About EchoSolv AS: EchoSolv AS is an AI-based decision support software indicated for use as an adjunct to echocardiography for the assessment of severe aortic stenosis. When used by an interpreting physician, it provides information to facilitate accurate diagnosis and support clinical review. EchoSolv AS is intended for qualified healthcare professionals and is not intended to replace physician judgment. Authorised for release by the Board of Directors of Echo IQ Limited. Investor Enquiries: Andrew Grover, Executive ChairHenry Jordan, Six Degrees Investor RelationsAndrew.grover@echoiq.ai / investor@echoiq.aiHenry.jordan@sdir.com.au / +61 (0) 431 271 538 ABOUT ECHO IQEcho IQ uses AI-driven technology and proprietary software to improve decision making in Cardiology. The company is based in Sydney, Australia. Disclosures: *Dr. Stamatios Lerakis serves as a paid key opinion leader, speaker and advisory board member for Echo IQ Limited. Forward-Looking Statements: This press release contains forward-looking statements within the meaning of the U.S. Private Securities Litigation Reform Act of 1995 and for the purposes of ASX Listing Rule 3.1 and ASX Guidance Note 8. Forward-looking statements are statements that are not historical facts and may include, without limitation, statements regarding Echo IQ’s commercial growth strategy, anticipated expansion of EchoSolv AS adoption, development of partnerships with health systems and cardiology practices, expected market adoption of AI-enabled decision support tools, and the potential benefits of EchoSolv AS for clinical decision-making and patient care. These forward-looking statements reflect the current expectations, assumptions, and beliefs of Echo IQ Limited based on information currently available to the company. Forward-looking statements are subject to known and unknown risks, uncertainties, and other factors that may cause actual results, performance, or achievements to differ materially from those expressed or implied by such statements. Such factors include, but are not limited to:Regulatory Risks: the ability of Echo IQ to maintain FDA 510(k) clearance for EchoSolv AS, obtain or maintain any additional required regulatory approvals in the United States or other jurisdictions, and achieve reimbursement coverage from government and private payors;Commercial Risks: the rate at which health systems and cardiology practices adopt EchoSolv AS, the ability of the company to execute on existing commercial agreements including with The Mount Sinai Hospital, performance under and renewal of customer contracts, and the competitive environment for AI-enabled medical software;Financial Risks: the availability of sufficient funding to execute the company’s commercial strategy, the timing and magnitude of revenue generation, and the company’s ability to achieve and sustain profitability; andOperational Risks: the performance and reliability of EchoSolv AS technology in real-world clinical environments, the ability to integrate EchoSolv AS into existing clinical workflows, and broader market acceptance of AI-based decision support tools in cardiology. The assumptions underlying the forward-looking statements in this release are made as of the date of this release and are subject to change without notice. Echo IQ cautions that the foregoing list of risk factors is not exhaustive. Investors and prospective investors should carefully consider these factors, as well as those disclosed in Echo IQ’s periodic filings with the ASX and other public disclosures, before making any investment decision. Echo IQ undertakes no obligation to publicly update or revise any forward-looking statement, whether as a result of new information, future events, or otherwise, except as required by applicable law or regulation, including the ASX Listing Rules and the U.S. securities laws. ™ Registered trademark or trademark of Echo IQ Ltd. or its affiliates. © 2025 Echo IQ Ltd. All rights reserved.
Peripheral/Endo
VVT Med Announces Positive Clinical Data for ScleroSafe(R) Published in Peer-Reviewed Indian Journal of Vascular and Endovascular Surgery
Prospective Study Conducted at Holy Spirit Hospital in Mumbai Reports 95% Vein Occlusion Rate at 30 Prospective Study Conducted at Holy Spirit Hospital in Mumbai Reports 95% Vein Occlusion Rate at 30
enVVeno Medical Receives First-Ever FDA IDE Approval for a U.S. Pivotal Study of a Non-Surgical Replacement Venous Valve
Apr 29, 2026 First-in-Class enVVe system to be evaluated in TAVVE U.S. Pivotal Trial Clinical site activation and patient enrollment expected to commence later this year Large unmet clinical need for approximately 3 million U.S. patients with severe deep Chronic Venous Insufficiency Potential multi-billion-dollar untapped U.S. market Company has approximately […]
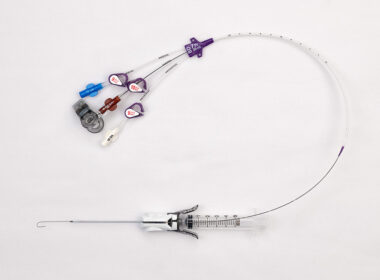
BD Reimagines Central Line Insertion, Advancing Leadership in Patient Safety Through Breakthrough Vascular Access Technology
With the launch of CentroVena One™, BD brings a first‑of‑its‑kind insertion system to market that simplifies central line placement for clinicians and enhances care in acute settings FRANKLIN LAKES, N.J., April 29, 2026 /PRNewswire/ — BD (Becton, Dickinson and Company) (NYSE: BDX), a…
Saving Limbs Where Others Stop: Cagent Vascular Announces Landmark Peer-Reviewed Data Demonstrating Safety and Efficacy of Serration Remodeling Therapy™ in the Most Challenging Arteries Below-the-Ankle
WAYNE, Pa.–(BUSINESS WIRE)– #CLI–Cagent Vascular, a leader in innovative endovascular technologies, today announced the publication of a peer-reviewed study in the Journal of Endovascular Therapy (JEVT) demonstrating the safety and efficacy of the Serranator® PTA Serration Balloon Catheter in pedal (inframalleolar) interventions for patients with chronic limb-threatening ischemia (CLTI). The investigator-initiated, multi-institutional, retrospective study, led by Dr. Edward Gifford, vascular surgeo
Cordis Launches SELUTION SLR™ PTA Drug-Eluting Balloon (DEB) in Japan
TOKYO–(BUSINESS WIRE)– #Cordis–Cordis, a global leader in interventional cardiovascular technologies, today announces the launch and availability of the SELUTION SLR™ PTA Drug-Eluting Balloon (DEB)1 for use in Japan. SELUTION SLR™ DEB is designed to deliver sirolimus in a controlled, sustained manner to reduce restenosis and restore vessel function. The SELUTION SFA Japan clinical trial results demonstrate durable clinical outcomes in a complex patient population, including 81.5% primary patency and 9
Humacyte to Host Virtual KOL Event to Discuss ATEV for Arteriovenous (AV) Access for Hemodialysis Patients on April 28, 2026
DURHAM, N.C., April 27, 2026 (GLOBE NEWSWIRE) — Humacyte, Inc. (Nasdaq: HUMA), a commercial-stage biotechnology platform company developing universally implantable, bioengineered human tissues at commercial scale, today announced that it will host a virtual key opinion leader (KOL) event on Tuesday, April 28, 2026 at 8:00 AM ET featuring Prabir Roy-Chaudhury MD, PhD, FASN (University of North Carolina (UNC) Kidney Center, Salisbury VA Medical Center), and Mohamad A. Hussain, MD, PhD, RPVI, FAHA, FRCSC, FACS (Brigham & Women’s Hospital, Center for Surgery and Public Health, Harvard Medical School), who will join company management to discuss the unmet need and current treatment landscape for hemodialysis patients with End-Stage Renal Disease (ESRD). To register, click here. The event will provide an overview of the Company’s Acellular Tissue Engineered Vessel (ATEV) in arteriovenous (AV) access for hemodialysis patients. The current standard of care for providing AV access involves connecting the patient’s artery with a vein, known as an arteriovenous fistula or AV fistula (AVF). Humacyte’s ATEV is designed to provide an alternative to the current standard of care for hemodialysis patients with ESRD, as AVFs can take weeks or months to mature and become usable for hemodialysis, and in many patients fail to mature at all. One Phase 3 trial has already been completed showing the potential advantages of the ATEV in AV Access compared to autologous AV fistula in patients at high risk for fistula failure. Company management will also review the ongoing V012 Phase 3 trial in hemodialysis for which interim top-line results are expected this quarter. A live question and answer session will follow the formal presentations. For uses other than the FDA approval in the extremity vascular trauma indication, the ATEV is an investigational product and has not been approved for sale by the FDA or any other regulatory agency. About Prabir Roy-Chaudhury MD, PhD, FASNPrabir Roy-Chaudhury MD, PhD, FASN is the Drs. Ronald and Katherine Falk Eminent Professor and Co-Director of the University of North Carolina (UNC) Kidney Center. He is also a Staff Nephrologist at the Salisbury VA Medical Center. After graduating from the Armed Forces Medical College, Pune, India, he trained in Internal Medicine and Nephrology at the University of Aberdeen, Scotland and at the Beth Israel Hospital, Harvard Medical School, Boston, USA. In addition to being an active transplant nephrologist, Dr. Roy-Chaudhury’s main research interest is in uremic vascular biology (including both dialysis vascular access dysfunction and cardiovascular complications in kidney disease patients). Dr. Roy-Chaudhury has been the recipient of extensive NIH, VA, and industry research grant funding (over 20 million USD) has published over 250 manuscripts and book chapters (h index = 65), and has delivered over 450 lectures (including multiple named and plenary presentations) across the globe. He is also the recipient of many national and international awards including the ASDIN Lifetime Achievement Award and the KS Chugh Memorial Oration. Dr. Roy-Chaudhury has also been actively involved in the public policy and administrative aspects of dialysis vascular access care and hemodialysis through leadership roles in multiple societies such as ASN, ASDIN and VASA, and is a Past President of the American Nephrologists of Indian Origin (ANIO). Dr. Roy-Chaudhury was also the founding ASN co-chair of the Kidney Health Initiative, a public-private partnership between ASN and the US FDA and is a regular member of the FDA’s Cardiovascular and Renal Drugs Advisory Committee (CRDAC). He is also currently the Immediate Past President of the American Society of Nephrology, which is the largest professional kidney organization in the world with over 21,000 members in 141 countries across the globe. About Mohamad A. Hussain, MD, PhD, RPVI, FAHA, FRCSC, FACSMohamad A. Hussain, MD, PhD, RPVI, FAHA, FRCSC, FACS is a Vascular and Endovascular Surgeon-Scientist at Brigham and Women’s Hospital, Core Faculty at the Center for Surgery and Public Health, and Associate Professor of Surgery at Harvard Medical School in Boston. He is board certified in vascular surgery by both the American Board of Surgery and the Royal College of Physicians and Surgeons of Canada. He obtained his medical degree from the Michael G. DeGroote School of Medicine at McMaster University. He completed vascular surgery residency and a PhD in clinical epidemiology and health services research through the Surgeon Scientist Training Program at the University of Toronto. He also completed a cardiovascular research fellowship at the Brigham. Dr. Hussain’s clinical practice is focused on general vascular and endovascular surgery, with special interests in complex hemodialysis access, aortic dissections and aneurysms, and thoracic outlet syndrome. He also co-directs the Heart & Vascular Hemodialysis Access workgroup across the MGB health system. Dr. Hussain’s research lab VESEL (Vascular & Endovascular Surgery Epidemiology Lab) conducts observational and clinical research to improve the care of patients with vascular diseases with special interests in population-based health services research, prediction with machine learning, enhancing causal inference research using target trial emulation, and clinical trials. Dr. Hussain also serves as the Associate Program Director of Surgical Residency and Associate Clerkship Director in the Department of Surgery at the Brigham. About HumacyteHumacyte, Inc. (Nasdaq: HUMA) is developing a disruptive biotechnology platform to deliver universally implantable bioengineered human tissues, advanced tissue constructs, and organ systems designed to improve the lives of patients and transform the practice of medicine. The Company develops and manufactures acellular tissues to treat a wide range of diseases, injuries, and chronic conditions. Humacyte’s Biologics License Application for the acellular tissue engineered vessel (ATEV) in the vascular trauma indication was approved by the FDA in December 2024. ATEVs are also currently in late-stage clinical trials targeting other vascular applications, including arteriovenous (AV) access for hemodialysis and peripheral artery disease (PAD). Preclinical development is also underway in coronary artery bypass grafts, pediatric heart surgery, treatment of type 1 diabetes, and multiple novel cell and tissue applications. Humacyte’s 6mm ATEV for AV access in hemodialysis was the first product candidate to receive the FDA’s Regenerative Medicine Advanced Therapy (RMAT) designation and has also received FDA Fast Track designation. Humacyte’s 6mm ATEV for urgent arterial repair following extremity vascular trauma and for advanced PAD also have received RMAT designations. The ATEV received priority designation for the treatment of vascular trauma by the U.S. Secretary of Defense. For more information, visit www.Humacyte.com. Forward-Looking StatementsThis press release contains forward-looking statements that are based on beliefs and assumptions and on information currently available. In some cases, you can identify forward-looking statements by the following words: “may,” “will,” “could,” “would,” “should,” “expect,” “intend,” “plan,” “anticipate,” “believe,” “estimate,” “predict,” “project,” “potential,” “continue,” “ongoing” or the negative of these terms or other comparable terminology, although not all forward-looking statements contain these words. These statements involve risks, uncertainties, and other factors that may cause actual results, levels of activity, performance, or achievements to be materially different from the information expressed or implied by these forward-looking statements. Although we believe that we have a reasonable basis for each forward-looking statement contained in this press release, we caution you that these statements are based on a combination of facts and factors currently known by us and our projections of the future, about which we cannot be certain. Forward-looking statements in this press release include, but are not limited to, our plans and ability to commercialize Symvess and, if approved by regulatory authorities, our product candidates, successfully and on our anticipated timelines; the degree of market acceptance of and the availability of third-party coverage and reimbursement for Symvess and, if approved by regulatory authorities, our product candidates; our ability to manufacture Symvess and, if approved by regulatory authorities, our product candidates in sufficient quantities to satisfy our clinical trial and commercial needs; the anticipated benefits of our ATEVs relative to existing alternatives; our plans and ability to execute product development, process development and preclinical development efforts successfully and on our anticipated timelines; our ability to design, initiate and successfully complete clinical trials and other studies for our product candidates and our plans and expectations regarding our ongoing or planned clinical trials, including for our V007 and V012 Phase 3 clinical trials; the anticipated characteristics and performance of our ATEVs and the public perception thereof; the implementation of our business model and strategic plans for our business; and the timing or likelihood of regulatory filings, acceptances and approvals. We cannot assure you that the forward-looking statements in this press release will prove to be accurate. These forward-looking statements are subject to a number of significant risks and uncertainties that could cause actual results to differ materially from expected results, including, among others, changes in applicable laws or regulations, the possibility that Humacyte may be adversely affected by other economic, business, competitive and/or reputational factors, and other risks and uncertainties, including those described under the header “Risk Factors” in our Annual Report on Form 10-K for the year ended December 31, 2024 filed by Humacyte with the SEC, and in future SEC filings. Most of these factors are outside of Humacyte’s control and are difficult to predict. Furthermore, if the forward-looking statements prove to be inaccurate, the inaccuracy may be material. In light of the significant uncertainties in these forward-looking statements, you should not regard these statements as a representation or warranty by us or any other person that we will achieve our objectives and plans in any specified time frame, or at all. Except as required by law, we have no current intention of updating any of the forward-looking statements in this press release. You should, therefore, not rely on these forward-looking statements as representing our views as of any date subsequent to the date of this press release. Humacyte Investor Contact:Joyce AllaireLifeSci Advisors LLC+1-617-435-6602jallaire@lifesciadvisors.cominvestors@humacyte.com Humacyte Media Contact:Rich LuchettePrecision Strategies+1-202-845-3924rich@precisionstrategies.commedia@humacyte.com
Guerbet announces an important milestone achievement in the “Vascular Embolization” indication for Lipiodol® Ultra Fluid (Iodinated ethyl esters of fatty acids of poppy seed oil)
VILLEPINTE, France, April 24, 2026 /PRNewswire/ — GUERBET (FR0000032526 GBT): Guerbet, a global leader in medical imaging, announces a positive outcome from a worksharing procedure, involving several EU member states, for a new Lipiodol® indication in interventional radiology procedures…
Reflow Medical Announces U.S. Launch of Next-Generation Cora Flex® and Cora Force® Torqueable Microcatheters
SAN CLEMENTE, Calif.–(BUSINESS WIRE)– #coraflex–Reflow Medical announces US launch of next-generation Cora Flex® and Cora Force® Torqueable Microcatheters for complex coronary interventions.
FLEX Vascular Announces 12-Month Real-World Outcomes Demonstrating Strong Safety, Durability, and Reduced Reinterventions with FLEX™ Vessel Prep System
LONDON, April 23, 2026 /PRNewswire/ — Minneapolis-based FLEX Vascular announced the presentation of 12-month results from the FLEX FIRST AV Registry, a prospective, multi-center, real-world study evaluating the FLEX™ Vessel Prep System in patients with dysfunctional hemodialysis access….