XARELTO® is available as both an oral tablet and oral suspension formulation for use in appropriate children less than 18 years of age Convenient oral suspension formulation advances standard of care for children; alleviates administration challenges found with injectable alternatives XARELTO® now has 11 indications, the most of any direct […]
Peripheral/Endo
Vesper Medical Announces Completion of Enrollment in the VIVID Clinical Trial
Company Achieves Key Milestone in the Development of Its Next Generation Deep Venous Stent System WAYNE, Pa.–(BUSINESS WIRE)–Vesper Medical, Inc., a developer of medical devices for deep venous disease, today announced the completion of enrollment in its pivotal study – Venous stent for the Iliofemoral Vein Investigational clinical trial using the Vesper DUO Venous Stent […]
Fist Assist Devices Receives FDA “Breakthrough Device” Designation for the World’s First Wearable Vein Dilation Device for ESRD Patients
Indication for Pre-Surgical Vein Dilation Use to Promote AV Fistula Creation LAS VEGAS–(BUSINESS WIRE)–Fist Assist Devices, LLC, an innovative medical device company focused on vein dilation to facilitate the Chronic Kidney Disease (CKD) patient’s journey through End-Stage Renal Disease (ESRD), announced today that it received “Breakthrough Device” designation from the […]
Sky wins FDA clearance to market the geko™ device for venous insufficiency and ischemia – a therapy area sorely in need of innovation.
The geko™ device increases blood flow in lower limb soft tissue of patients with venous insufficiency and/or ischemia. DARESBURY, England, Dec. 16, 2021 /PRNewswire/ — Medical devices company, Sky Medical Technology Ltd, has today announced U.S. Food and Drug Administration (FDA) 510(k) clearance to market the geko™ device for increasing microcirculatory blood flow in […]
Philips to further expand its image-guided therapy devices portfolio through acquisition of Vesper Medical
Amsterdam, the Netherlands – Royal Philips (NYSE: PHG; AEX: PHIA), a global leader in health technology, today announced that it has signed an agreement to acquire Vesper Medical Inc., a US-based medical technology company that develops minimally-invasive peripheral vascular devices. Vesper Medical will further expand Philips’ portfolio of diagnostic and therapeutic devices […]
InterVene’s BlueLeaf Endovenous Valve Formation (EVF) System Granted Breakthrough Device Designation by the FDA
SOUTH SAN FRANCISCO, Calif.–(BUSINESS WIRE)–InterVene Inc. announced today that it has received Breakthrough Device Designation by the FDA for the company’s BlueLeaf® Endovenous Valve Formation System. BlueLeaf is the first catheter-based solution developed for deep vein reflux — the failure of venous valves in the legs — which does not require […]
Bluegrass Vascular Announces New Paper Reporting Ability to Eliminate Use of a Femoral Catheter with the Surfacer® System
December 8, 2021, San Antonio, TX – Bluegrass Vascular Technologies, a private medical technology company focused on innovating lifesaving devices and methods for vascular access procedures, announced today the publication of a case report demonstrating the ability to utilize the Surfacer® System to perform the Inside-Out® procedure and obtain right-sided central venous access […]
Artio Medical’s Amplifi™ Vein Dilation System Demonstrates Encouraging Clinical Results for Hemodialysis Patients Requiring Vascular Access
PRAIRIE VILLAGE, Kan., Nov. 30, 2021 /PRNewswire/ — Artio Medical, Inc., a medical device company developing innovative products for the peripheral vascular, neurovascular, and structural heart markets, today announced that full results from its first-in-human (FIH) clinical study of the Amplifi™ Vein Dilation System were presented at the 2021 VEITH Symposium by Surendra Shenoy, MD-PhD, […]
Soundbite Medical Announces Issuance Of A Fifth U.S. Patent Covering Novel Active Balloon Devices Intended For Plaque Modification Of Calcified Arteries
MONTREAL–(BUSINESS WIRE)–Soundbite Medical Solutions Inc. (SBMS), a medical device company dedicated to developing solutions for the interventional treatment of calcific occlusive peripheral and coronary arterial diseases, today announced that the United States Patent and Trademark Office has issued US patent No. 11,179,169, entitled “Device for delivering mechanical waves through a balloon catheter”. […]
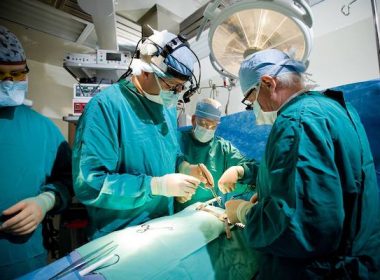
World’s First|Aopeng Medical Completes Clinical Trial of Robotic-Assisted Peripheral artery Intervention of iliac artery in human
SHANGHAI, Nov. 23, 2021 /PRNewswire/ — On November , 2021, Aopeng Medical’s ALLVAS™ Endovascular Intervention Surgical Robot, successfully completed the first-in-human clinical evaluation of robot-assisted peripheral artery intervention of iliac artery in the Department of Vascular Surgery at Shanghai Changhai Hospital. The procedure was operated by Prof. Qing-Sheng Lu and his colleagues. The endovascular […]