Late-breaking data demonstrate superior rates of primary patency and statistically significant sustained clinical improvement LAS VEGAS and MARLBOROUGH, Mass., Oct. 6, 2021 /PRNewswire/ — Today, Boston Scientific Corporation (NYSE: BSX) announced positive data for the Eluvia™ Drug-Eluting Vascular Stent System (Eluvia stent) during a late-breaking clinical trial presentation at the Vascular InterVentional Advances (VIVA) meeting […]
Peripheral/Endo
Positive Late-breaking Clinical Trial Data for the EkoSonic™ Endovascular System Presented at VIVA21
Analysis found no intracerebral hemorrhages, low major bleeding rates; registry represents largest data set in interventional treatment of pulmonary embolism LAS VEGAS and MARLBOROUGH, Mass., Oct. 5, 2021 /PRNewswire/ — Today, Boston Scientific (NYSE: BSX) announced positive results for the EkoSonic™ Endovascular System (EKOS system) during a late-breaking clinical trial presentation at the Vascular InterVentional Advances […]
Boston Scientific Announces Positive Late-Breaking Clinical Trial Data for the Ranger™ Drug-Coated Balloon
Two-year data demonstrate continued high rates of primary patency including in patients with complex lesions LAS VEGAS and MARLBOROUGH, Mass., Oct. 5, 2021 /PRNewswire/ — Today, Boston Scientific (NYSE: BSX) announced positive results for the Ranger™ Drug-Coated Balloon (DCB) during a late-breaking clinical trial presentation at the Vascular InterVentional Advances (VIVA) meeting in Las Vegas. The data […]
First Round of Late-Breaking Clinical Trial Results Announced at VIVA21
LAS VEGAS, Oct. 5, 2021 /PRNewswire/ — The VIVA Foundation, a not-for-profit organization dedicated to advancing the field of vascular medicine and intervention through education and research, announces the first round of 16 highly anticipated late-breaking clinical trial results at VIVA121 hosted at Wynn Las Vegas. VIVA (Vascular InterVentional Advances) is an […]
Cardiovascular Systems, Inc. Announces First Patient Treated with ViperCross™ Peripheral Support Catheter
ST. PAUL, Minn.–(BUSINESS WIRE)–Cardiovascular Systems, Inc. (CSI) (NASDAQ: CSII), a medical device company developing and commercializing innovative interventional treatment systems for patients with peripheral and coronary artery disease, announced today that the first patient has been successfully treated with its ViperCross peripheral support catheter. Dr. Billy J. Kim, MD, The […]
JanOne Encouraged By Recent FDA Communications Regarding Phase 2B Clinical Trial Design of Lead Drug Candidate JAN101
JanOne’s management is encouraged regarding Recent Discussions on Potential Phase 2b Trial Endpoints and Possible Regulatory Routes for JAN101 as Treatment for Peripheral Artery Disease (PAD) LAS VEGAS, Sept. 28, 2021 /PRNewswire/ — JanOne Inc. (Nasdaq: JAN), a company that focuses on the development of treatments for conditions that cause severe pain – in […]
Surmodics Announces Successful First Patient Uses of Sublime™ Radial Access .018 RX PTA Dilatation Catheter
Latest addition to the Sublime™ Radial Access Platform expands market opportunity by increasing size offering to treat larger vessels and providing the shaft length needed to treat more distal lesions using a radial approach EDEN PRAIRIE, Minn.–(BUSINESS WIRE)–Surmodics, Inc. (NASDAQ:SRDX), a leading provider of medical device and in vitro diagnostic […]
Endologix Initiates Randomized Study of ALTO® Abdominal Stent Graft System
JAGUAR Study Evaluates ALTO Compared to Other Commercially Available EVAR Devices IRVINE, Calif.–(BUSINESS WIRE)–Endologix LLC, a leader in the treatment of vascular disease, today announced that the first patient has been enrolled in the company’s JAGUAR study to compare outcomes for the company’s ALTO® Abdominal Stent Graft System to other commercially […]
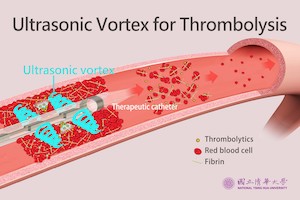
NTHU Researchers Develop Ultrasonic Vortex Thrombolytic Device
HSINCHU, Taiwan–(BUSINESS WIRE)–As COVID-19 vaccination has raised the issue of thrombosis, Dr. Chih-Kuang Yeh, Distinguished Professor of the Department of Biomedical Engineering and Environmental Sciences, has led a research team to developed the world’s first ultrasonic vortex thrombolytic device and to treat thrombosis quickly and safely. The research has been […]
W. L. Gore & Associates Enhances GORE® VIABAHN® Endoprosthesis Portfolio With Lower Profile Delivery
Improvements to large diameter devices include accessibility through smaller sheaths while providing enhanced visualization by adding radiopaque markers 25 years of continued innovation PUTZBRUNN, Germany, Sept. 22, 2021 /PRNewswire/ — As part of efforts to continuously improve medical solutions for patients with complex vascular disease, W. L. Gore & Associates, Inc. (Gore) announced […]