NEW YORK, Nov. 20, 2025 /PRNewswire/ — Frank J. Veith and Rasit Dinc presented new expert insights on the role of advancing artificial intelligence (AI) technologies in contemporary vascular medicine during a featured session at the 52nd annual VEITH Symposium. Their discussion outlined…
Peripheral/Endo
AIROS Medical Launches Custom Travel Case for Lymphedema Compression Therapy Devices
AUDUBON, Pa., Nov. 20, 2025 /PRNewswire/ — AIROS® Medical., Inc., a leading medical technology manufacturer and designer specializing in compression therapy devices that treat lymphedema and venous disorders, today announced the release of the AIROS Medical Travel Case. The case will…
Frank J Veith and Rasit Dinc Evaluate Next-Generation Technologies in Vascular Treatments
AI-driven innovations in digital health and imaging were discussed at the VEITH Symposium, where Frank Veith and Rasit Dinc evaluated the impact of next-generation technologies on vascular treatments NEW YORK, Nov. 19, 2025 /PRNewswire-PRWeb/ — During a special session at the 52nd annual…
RapidAI Earns FDA Clearance for Rapid Aortic, Bringing AI-Driven Aortic Measurements and Surveillance to Care Teams
SAN MATEO, Calif.–(BUSINESS WIRE)–RapidAI, the pioneer of deep clinical AI and global leader in enterprise imaging, today announced U.S. Food and Drug Administration (FDA) clearance of Aortic Management, part of the Rapid Aortic product, a comprehensive deep clinical AI solution designed to transform the acute assessment and longitudinal management of aortic disease. […]
ViTAA Medical Secures FDA 510(k) Clearance for AiORTA™ Plan — Marking the First Step in Its Hyper-Precise Aortic Care Platform
MONTREAL–(BUSINESS WIRE)–ViTAA Medical Solutions Inc. today announced that the U.S. Food and Drug Administration (FDA) has granted 510(k) clearance for AiORTA™ Plan, the company’s fully automated, hyper-precise aortic surgery planning solution. This clearance represents a key milestone in the evolution of ViTAA’s broader AiORTA™ platform, built to support physicians across the full continuum of […]
Humacyte Announces Six Presentations Scheduled for the Upcoming 52nd Annual Symposium on Vascular and Endovascular Issues, Techniques and Horizons (VEITHsymposium)
DURHAM, N.C., Nov. 13, 2025 (GLOBE NEWSWIRE) — Humacyte, Inc. (Nasdaq: HUMA), a commercial-stage biotechnology platform company developing universally implantable, bioengineered human tissues at commercial scale, today announced the details of six presentations on the Company’s acellular tissue engineered vessel (ATEV™) that are scheduled for the 52nd Annual Symposium on Vascular and Endovascular Issues, Techniques And Horizons (VEITHsymposium), to be held November 18-22, 2025 in New York, NY. The VEITHsymposium is a premier educational event for vascular surgeons, interventional radiologists, interventional cardiologists, and other vascular specialists.
GenesisX Robotic Magnetic Navigation System Receives U.S. FDA Clearance
ST. LOUIS, Nov. 10, 2025 (GLOBE NEWSWIRE) — Stereotaxis (NYSE: STXS), a pioneer and global leader in surgical robotics for minimally invasive endovascular intervention, today announced that it received U.S. Food and Drug Administration 510(k) clearance for its latest generation robotic system, GenesisX™.
First Surgery With Polymer-Based Coil Takes Place, Using Embolization, Inc., Device
— Polymer-based coil minimizes artifacts in imaging — BOULDER, Colo., Nov. 10, 2025 /PRNewswire/ — A woman in Delaware has become the first patient to undergo minimally invasive, endovascular surgery with a polymer-based coil. The coil, manufactured by Embolization, Inc., is a vascular…
GORE ANNOUNCES POSITIVE CLINICAL TRIAL RESULTS OF THE INVESTIGATIONAL GORE® VIABAHN® FORTEGRA VENOUS STENT FOR TREATMENT OF PATIENTS WITH DEEP VENOUS ILIOCAVAL OBSTRUCTION
The investigational device evaluated in the trial, previously known as the GORE® VIAFORT Vascular Stent, will now be referred to as the GORE® VIABAHN® FORTEGRA Venous Stent. CAUTION: Investigational device. Limited by United States law to investigational use. LAS VEGAS and FLAGSTAFF, Ariz….
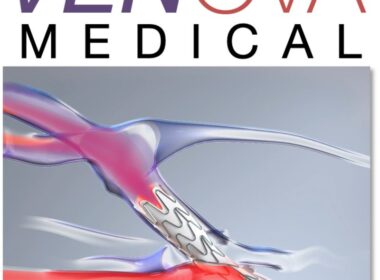
Venova Medical Announces First Subjects Enrolled in VENOS-3 Pivotal IDE Study of the Velocity® Percutaneous AVF System
The Velocity System is a next generation percutaneous AVF technology designed to reduce the need for reinterventions to achieve fistula maturation and improve hemodialysis vascular access outcomes while reducing costs. LOS GATOS, Calif., Nov. 6, 2025 /PRNewswire-PRWeb/ — Venova Medical,…