Stroke Risk Is Dramatically Reduced During and After Carotid Artery Stenting with Innovative New Procedure TINLEY PARK, Ill.–(BUSINESS WIRE)–When Delnora Kizzee Talbert met Dr. Eugene Tanquilut in 2017, neither of them had any idea that in 3 short years they would be making history together. “I’ll never forget meeting Dr. T,” says […]
Peripheral/Endo
Micro Medical Solutions Announces CE Mark Approval of 2.5mm MicroStent and MicroBalloon XL
The 2.5 mm stent and 120 cm OTW balloon catheter will make it possible for more patients with CLI to be treated, reducing the rate of amputation. WILMINGTON, Mass., Oct. 13, 2020 /PRNewswire/ — Micro Medical Solutions (MMS) today announced CE Mark approval for the 2.5 mm MicroStent vascular stent, the smallest […]
WaveClear, Inc Raises $1.125 Million in Series-A Funding Led by Hunniwell Lake Ventures
SAN FRANCISCO, Oct. 13, 2020 /PRNewswire/ — WaveClear, Inc., a medical device start-up pioneering the deployment of therapeutic ultrasound technologies to combat vascular diseases, recently announced today the closing of a $1,125,000 Series-A round of investment led by Palo Alto-based medical device venture capital firm, Hunniwell Lake Ventures (“HLV”). Peripheral vascular diseases are increasing worldwide, and […]
Enrolment of First Two Patients in PRISTINE Study with SELUTION SLR™ Sirolimus Drug Eluting Balloon
NYON, Switzerland, Oct. 11, 2020 /PRNewswire/ — MedAlliance has announced enrolment of the first two patients in the PRISTINE registry with SELUTION SLR™ 018 DEB for the treatment of patients with Below The Knee disease (Chronic Limb Threatening Ischemia). This is the first DEB accepted by the FDA for its “Breakthrough Program”. SELUTION […]
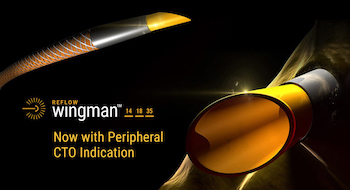
Reflow Medical Receives Approval in Japan for the Wingman Catheter to Cross Chronic Total Occlusions (CTOs) in Peripheral Artery Disease
SAN CLEMENTE, CA /October 7, 2020 /Business Wire/ — Reflow Medical, Inc., a California- based medical device company, announced that Japan’s Pharmaceuticals and Medical Devices Agency (PMDA) has approved the Wingman™ CTO Catheter. Reflow Medical has partnered with Century Medical, Inc. (CMI), a leading medical device distributor based in Tokyo, […]
Cardiovascular Systems Announces Presentation of Data From Large Coronary Orbital Atherectomy Study at the TCT Connect 2020 Conference
ST. PAUL, Minn.–(BUSINESS WIRE)–Cardiovascular Systems, Inc. (CSI®) (NASDAQ: CSII), a medical device company developing and commercializing innovative interventional treatment systems for patients with peripheral and coronary artery disease, announced today that results from a large retrospective observational study of coronary orbital atherectomy will be released at TCT Connect 2020. Severe calcification […]
PQ Bypass Completes Enrollment in DETOUR2 Percutaneous Femoral-Popliteal Bypass Pivotal Study for Patients With Complex Peripheral Arterial Disease
FDA-designated Breakthrough Device now one step closer to PMA submission MILPITAS, Calif.–(BUSINESS WIRE)–PQ Bypass, an innovative medical device company pioneering advancements in the treatment of complex peripheral artery disease (PAD), announces enrollment of the final subject in the company’s flagship IDE, the DETOUR2 clinical trial. This important milestone occurs only […]
Teleflex Announces Expanded Indications for the Arrow® EZ-IO® Intraosseous Vascular Access System
Now Cleared for up to 48-Hour Dwell WAYNE, Pa., Oct. 01, 2020 (GLOBE NEWSWIRE) — Teleflex Incorporated (NYSE: TFX), a leading global provider of medical devices for critical care and surgery, has announced it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) to expand the Indications […]
Mentice to Acquire Vascular Simulations
– Combined portfolio will enhance Mentice’s endovascular simulation offerings with world class replication solutions – Acquisition to expand Mentice’s global customer base and offer a strong entry into previously untapped market segments STOCKHOLM, Oct. 1, 2020 /PRNewswire/ — Mentice AB (STO: MNTC) today signed a definitive agreement to acquire the New York-based medical […]
Vascular Surgeon Eugene Tanquilut Performs First-Ever “Ellipsys” Dialysis Access Procedure in Chicago Southland
Groundbreaking, minimally invasive procedure means dialysis in half the time for late stage renal failure patient TINLEY PARK, Ill.–(BUSINESS WIRE)–Vascular Specialists in Tinley Park, Illinois, is pleased to announce that Dr. Eugene Tanquilut, has successfully completed the first Ellipsys Vascular Access System procedure in the Chicago Southland region. “Peter Song was a perfect candidate […]