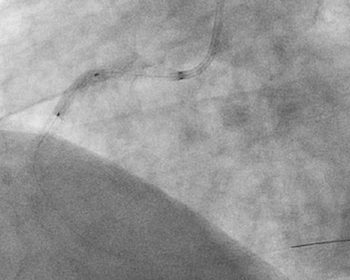
Novel dual-balloon technology designed for true 360° stent apposition CAMPBELL, Calif., May 12, 2020 /PRNewswire/ — Ostial Corporation (Ostial), a private medical technology company focused on addressing the clinical challenges of aorto-ostial interventions, today reached a new milestone, passing the mark of 15,000 commercial units sold of its FLASH™ Aorto-Ostial Angioplasty System (FLASH System) in […]
Peripheral/Endo
FDA Clears New XO Cross Microcatheter Platform
New non-tapered metal-alloy catheter platform FDA cleared for use in peripheral vasculature PARK CITY, Utah, May 12, 2020 /PRNewswire/ — Transit Scientific announced the FDA cleared the XO Cross® Microcatheter platform for guidewire support, exchange, and contrast media injection in the peripheral vasculature. Microcatheters are small 0.70-1.30mm outer diameter (OD) catheters used to provide guidewire […]
Micro Medical Solutions: First Case Completed in STAND Study, the Pivotal Clinical Trial of MicroStent
Major milestone reached in journey toward FDA approval for potentially limb-saving treatment of peripheral artery disease WILMINGTON, Mass., May 12, 2020 /PRNewswire/ — Micro Medical Solutions (MMS) today announced the first implantation of the MicroStent vascular stent in the FDA randomized, multicenter pivotal clinical study, STAND (A Clinical Evaluation of the MicroSTent PeripherAl Vascular […]
Straub Medical AG acquired by Becton, Dickinson and Company (BD)
(PresseBox) ( Wangs,04/21/20) Straub Medical AG, a privately held Swiss company, announces that it has approved a full acquisition by Becton, Dickinson and Company (BD). The acquisition was completed on April 20, 2020. Straub Medical AG is headquartered in Wangs, where it manufactures, develops and sells medical devices for the treatment of arterial […]
FDA Clears New Mechanical Thrombectomy Platform
New large catheter platform FDA cleared to remove blood clots from peripheral vessels PARK CITY, Utah, May 6, 2020 /PRNewswire/ — Control Medical Technology announced the FDA cleared the Aspire MAX 7 – 11F Mechanical Thrombectomy platform to remove blood clots from peripheral vessels. Blood clot removal (thrombectomy) is a common procedure. Coronary […]
Centerline Biomedical Completes First Human Patient in the United States with IOPS™ (Intra-Operative Positioning System)
Fast-growing startup company continues to execute key milestones and begins first in-market use of novel endovascular navigation technology CLEVELAND, May 4, 2020 /PRNewswire/ — Surgical navigation startup Centerline Biomedical, Inc. (Centerline), a Cleveland Clinic spin-off company, has announced the successful completion of the first in a series clinical cases in the United States as […]
Cagent Vascular Announces FDA 510(k) Clearance for its Serranator Device for the Infrapopliteal Indication and Completion of its PRELUDE-BTK Clinical Trial Enrollment
WAYNE, Pa.–(BUSINESS WIRE)–Cagent Vascular, a developer of serration technology for vessel dilatation in cardiovascular disease interventions, announces FDA 510(k) Clearance of its Serranator® PTA Serration Balloon Catheter for treating below-the-knee (BTK) lesions. The Serranator device is the first and only angioplasty balloon FDA Cleared and CE Marked that embeds serration technology into a […]
Intact Vascular Announces First U.S. Commercial Use of Tack Endovascular System® (4F) in Below-the-Knee Arteries
WAYNE, Pa.–(BUSINESS WIRE)–Intact Vascular, Inc., a developer of medical devices for minimally invasive peripheral vascular procedures, today announced the first commercial use of its Tack Endovascular System® (4F) in multiple sites across the United States. Notably the first FDA-approved vascular implant for below-the-knee post-angioplasty dissection repair, Tack® implants are designed to optimize balloon […]
FDA Grants NEXUS™ Aortic Arch Stent Graft System Breakthrough Designation
Endospan is Granted Breakthrough Device Designation from the U.S. Food and Drug Administration (FDA) for the NEXUS™ Aortic Arch Stent Graft System. HERZLIA, Israel–(BUSINESS WIRE)–Endospan, a pioneer in off-the-shelf endovascular repair of aortic arch disease was recently granted Breakthrough Device Designation from the U.S. Food and Drug Administration (FDA) for […]
Proximo Medical Named Commercialization Partner for Elucid Bioimaging
CASTLE PINES, Colo., April 16, 2020 /PRNewswire/ — Today Proximo Medical, LLC, a business acceleration solution that addresses medical device commercialization challenges among startup and established medical device technologies looking to expand adoption in the U.S., announced a partnership with Elucid Bioimaging, a developer of diagnostic image analysis software used to assist in the detection and treatment […]