Dr. Michael Lichtenberg Treats First Patient in Arnsberg, Germany ST. PAUL, Minn.–(BUSINESS WIRE)–Cardiovascular Systems, Inc. (CSI®) (NASDAQ: CSII), a medical device company developing and commercializing innovative interventional treatment systems for patients with peripheral and coronary artery disease, announced today that the first patient in Europe has been treated with its […]
Peripheral/Endo
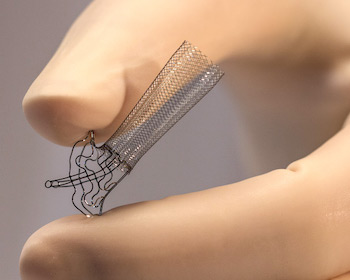
Enrollment Complete for VALUE, the Prospective EU VasQ™ External Support Post-Market Study
TEL AVIV, Israel, Dec. 11, 2019 /PRNewswire/ — Laminate Medical Technologies (Laminate) has announced the completion enrollment of the VALUE study for the VasQ™ External Support. The post-market study enrolled 80 patients (50 upper arm and 30 forearm fistulas) for sites across Germany, France, Spain and the UK and will be followed for one year. The study is the […]
CORRECTING and REPLACING First Patient Enrolled in PROMISE II U.S. Pivotal Study of LimFlow System to Treat Chronic Limb-threatening Ischemia
Clinical Study to Evaluate Device’s Ability to Prevent Amputation and Promote Wound Healing in Patients with No Other Options CORRECTION…by LimFlow SA PARIS–(BUSINESS WIRE)–First paragraph, last sentence should read: The successful first case was performed by Mark Archie, MD, principal investigator for the PROMISE II trial at Harbor-UCLA Medical Center, […]
Canon Medical’s Alphenix Platform at the Forefront of Helping Hospitals Prepare for the Future of Interventional Medicine
Canon Medical to Showcase High-Definition Image Resolution Technology at RSNA 2019 CHICAGO–(BUSINESS WIRE)–Since its launch in 2018, Canon Medical’s Alphenix™ platform has helped clinicians think beyond the traditional boundaries of interventional procedures, with its streamlined workflow, dose optimization and improved image resolutions. For example, the specialized team of neuroendovascular surgeons […]
Shape Memory Medical Receives PMDA Approval for the IMPEDE® Embolization Plug Family
SANTA CLARA, Calif.–(BUSINESS WIRE)–Shape Memory Medical Inc. announced today that its IMPEDE® Embolization Plugs have been approved in Japan by the Pharmaceuticals and Medical Devices Agency (PMDA). Cosmotec, which is a Group Company of M3 Inc https://corporate.m3.com/en/ listed on the Tokyo Stock Exchange (TYO:2413), championed the approval process in Japan and is […]
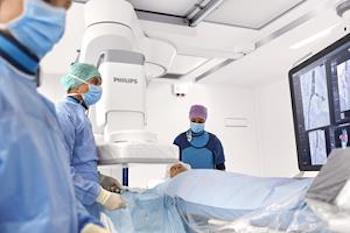
One millionth procedure carried out on Philips Azurion advanced image-guided therapy platform
Industry-leading platform combines clinical excellence and workflow innovation, helping clinicians and hospitals to deliver outstanding patient care Amsterdam, the Netherlands – Royal Philips (NYSE: PHG, AEX: PHIA), a global leader in health technology, today announced the one millionth procedure on its flagship Azurion image-guided therapy platform. Since its introduction in February 2017, Azurion […]
CryoLife Receives CE Mark for E-nside Thoraco-abdominal Stent Graft
First Off-the-Shelf Thoraco-abdominal Stent Graft with Pre-Cannulated Inner Branch Technology ATLANTA, Dec. 2, 2019 /PRNewswire/ — CryoLife, Inc. (NYSE: CRY), a leading cardiac and vascular surgery company focused on aortic disease, announced today that it has received CE Mark for the E-nside TAAA multibranch stent graft system for the endovascular treatment of thoraco-abdominal aneurysms. Approximately […]
CryoLife Receives CE Mark for E-nya Thoracic Stent Graft
Product Launch Anticipated in the First Quarter 2020 ATLANTA, Dec. 2, 2019 /PRNewswire/ — CryoLife, Inc. (NYSE: CRY), a leading cardiac and vascular surgery company focused on aortic disease, announced today it has received CE Mark for the E-nya thoracic stent graft system for the minimally invasive repair of lesions of the descending thoracic aorta, […]
One millionth procedure carried out on Philips Azurion advanced image-guided therapy platform
November 27, 2019 Industry-leading platform combines clinical excellence and workflow innovation, helping clinicians and hospitals to deliver outstanding patient care Amsterdam, the Netherlands – Royal Philips (NYSE: PHG, AEX: PHIA), a global leader in health technology, today announced the one millionth procedure on its flagship Azurion image-guided therapy platform. Since its introduction […]
InspireMD Reports Updated Positive CGuard™ EPS Data Presented at VEITH 2019
CGuard™ EPS clinical data featured as a prominent discussion point in multiple key presentations Data from investigator-initiated multicenter, 729-patient IRONGUARD 2 study suggests that the use of CGuard™ EPS in routine clinical practice is associated with no major periprocedural, 30-day or one-year neurological complications TEL AVIV, Israel, Nov. 26, 2019 […]