SAN DIEGO, Sept. 30, 2024 /PRNewswire/ — Cortechs.ai, a leader in advanced imaging software medical analysis, is proud to announce a strategic partnership with Ascend Imaging, a premier provider of radiology products and services. This collaboration will enhance Cortechs.ai’s ability to…
Radiology
RenovoRx Increases Production of FDA-Cleared RenovoCath® Delivery System in Response to Strong Demand from Oncology and Interventional Radiology Physicians
Company expands relationship with manufacturing partner Medical Murray, and continues active exploration of standalone opportunities for RenovoCath Company expands relationship with manufacturing partner Medical Murray, and continues active exploration of standalone opportunities for RenovoCath
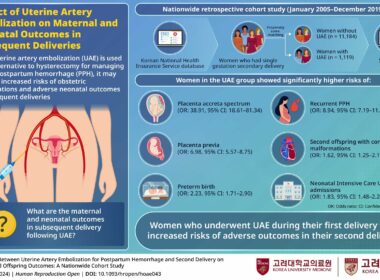
Korea University Study Reveals Potential Effects of Uterine Artery Embolization on Subsequent Deliveries
Korean nationwide cohort study shows increased complications for both mothers and newborns in deliveries following uterine artery embolization SEOUL, South Korea, Sept. 23, 2024 /PRNewswire/ — Postpartum hemorrhage (PPH) is a serious condition where a woman experiences significant blood…
NewVue Announces Strategic Partner Agreement with Konica Minolta Healthcare to Enhance Radiology Workflow
TAMPA, Fla. and WAYNE, N.J., Sept. 19, 2024 (GLOBE NEWSWIRE) — NewVue, a leader in cloud-native radiology workflow solutions, and Konica Minolta Healthcare Americas, Inc., a leading provider of medical diagnostic imaging and healthcare information technology, announce a strategic partner agreement. This partnership allows Konica Minolta to offer NewVue’s innovative EmpowerSuite Radiology Workflow Orchestrator and Command Center to its extensive network of healthcare customers across the US, integrating seamlessly with Konica Minolta’s Exa® Platform to enhance radiology workflows. EmpowerSuite is a key component of Konica Minolta’s enterprise imaging portfolio.
Sirtex Medical Announces Certification of SIR-Spheres® Y-90 Resin Microspheres According to Medical Device Regulation (EU) 2017/745 and European Launch of the Innovative SIROS™ Delivery System
Sirtex brings an innovative liver cancer treatment delivery system to Europe, enhancing options for physicians and their patients. WOBURN, Mass. (September 16, 2024) – Sirtex Medical (“Sirtex”), a leading manufacturer of interventional treatment solutions, today announced that it has received certification according to Medical Device Regulation (EU) 2017/745 for SIR-Spheres® […]
Top TeleRad Provider Aligned Radiology Selects InsiteOne, NewVue, and RADPAIR to Deliver Cloud-Native PACS, AI-Powered Workflow Orchestration, and Conversational Generative AI Diagnostic Reporting
WALLINGFORD, Conn. and TAMPA, Fla., Sept. 11, 2024 /PRNewswire/ — InsiteOne, NewVue.AI, and RADPAIR are thrilled to announce a groundbreaking strategic contract with Aligned Radiology, a leading teleradiology provider. This collaboration combines advanced AI-driven workflow…
Merit Medical to Present at the 2024 Cardiovascular and Interventional Radiological Society of Europe (CIRSE) Annual Congress
SOUTH JORDAN, Utah, Sept. 11, 2024 (GLOBE NEWSWIRE) — Merit Medical Systems, Inc. (NASDAQ: MMSI), a leading global manufacturer and marketer of healthcare technology, announced today that clinicians will present updates on two of its pivotal studies during scientific sessions held on September 14 at CIRSE in Lisbon, Portugal.
Allegheny Valley Hospital Completes $12 Million Imaging and Radiology Department Expansion
Allegheny Health Network’s (AHN) Allegheny Valley Hospital (AVH) has completed a $12 million expansion and renovation of its imaging and radiology department. The new 8,000-square-foot imaging center, located on the ground floor of AVH, offers state-of-the-art imaging technologies and…
Harrison.ai launches world leading AI model to transform healthcare
Harrison.rad.1 significantly outperforms other major models on radiology tasks The new model is being made accessible to select healthcare professionals and regulators to open up the conversation on responsible use of AI in healthcare SYDNEY, Sept. 4, 2024 /PRNewswire/ — Healthcare AI…
Positron Corporation Announces Sale of NeuSight PET-CT Scanner
Niagara Falls, NY, Aug. 26, 2024 (GLOBE NEWSWIRE) — Positron Corporation (“Positron” or the “Company”) (OTC: POSC), a leading molecular imaging medical device company offering PET and PET-CT (Positron Emission Tomography/Computed Tomography) imaging systems and clinical services, is pleased to announce the sale of Positron’s NeuSight PET-CT 64 slice scanner to a recognized leader in preventive, invasive and interventional cardiovascular services.