LATHAM, N.Y., February 15, 2024–(BUSINESS WIRE)–AngioDynamics, Inc. (NASDAQ: ANGO), a leading and transformative medical technology company focused on restoring healthy blood flow in the body’s vascular system, expanding cancer treatment options and improving patient quality of life, today announced that it has completed the sale of its PICC and Midline […]
Radiology
The Telerad Group’s AI-powered RIS-PACS Takes Center Stage at Arab Health
DUBAI, UAE, Feb. 8, 2024 /PRNewswire/ — The Telerad Group, a leading global provider of remote radiology services, participated in the Arab Health 2024 exhibition that provides a unique opportunity for healthcare professionals and organizations to explore cutting-edge innovations in…
UltraSight Partners with SELVAS Healthcare to Develop and Commercialize AI-Powered Cardiac Ultrasound Software in Asia
New joint venture creates pathway for introducing UltraSight’s technology to Korean market TEL AVIV, Israel, Feb. 5, 2024 /PRNewswire/ — Today, UltraSight, a digital health pioneer transforming cardiac imaging through the power of artificial intelligence, announced a partnership with…
SoniVie announces enrollment completion in the REDUCED-1 pilot study with TIVUS™ Ultra-Sound Renal Denervation
SoniVie announces enrollment completion in the REDUCED-1 pilot study TEL AVIV, Israel, Jan. 31, 2024 /PRNewswire/ — SoniVie, an Israeli company developing a novel, proprietary Therapeutic Intra-Vascular Ultrasound (TIVUSTM) System to treat a variety of hypertensive disorders, announced…
New Philips Mini TEE ultrasound transducer helps improve cardiac care for more patients
January 31, 2023 Latest innovation has the potential to reduce anesthesia during minimally invasive procedures [1] As a pioneer and leading innovator in cardiac ultrasound, Philips has developed the smaller X11- 4t Mini 3D TEE transducer to serve more patients with this valuable imaging tool, from pediatric patients to older […]
SS Innovations Announces Filing of Pre-Submission Application to the Center for Devices and Radiological Health (CDRH) of the United States Food and Drug Administration
FORT LAUDERDALE, Fla., Jan. 26, 2024 (GLOBE NEWSWIRE) — SS Innovations International, Inc. (the “Company” or “SS Innovations”) (OTC: SSII), a developer of innovative surgical robotic technologies dedicated to making robotic surgery affordable and accessible to a global population, today announced that it has filed a pre-submission application with the U.S. […]
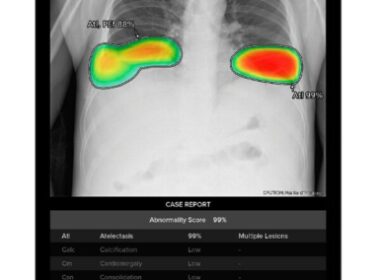
Lunit INSIGHT CXR Excels in Lung Nodule Detection – Exceptional Performance in Head-to-Head Study published in Radiology
– Lunit’s AI-powered chest X-ray analysis solution shows highest AUC of 0.93 in a recent multi-center, multi-use case, multi-vendor lung nodule detection study SEOUL, South Korea, Jan. 17, 2024 /PRNewswire/ — Lunit (KRX:328130.KQ), a leading provider of AI-powered solutions for cancer…
IceCure Medical’s ProSense® Featured at Indian Society of Vascular and Interventional Radiology Conference: Key Opinion Leaders Highlight Benefits of Cryoablation
ProSense® users speaking at the conference about cryoablation included ISVIR President Dr. Suyash Kulkarni, as well as Dr. Shuvro Roy-Choudhury CAESAREA, Israel, Jan. 16, 2024 /PRNewswire/ — IceCure Medical Ltd. (Nasdaq: ICCM) (“IceCure” or the “Company”), developer of the ProSense®…
InkSpace Imaging receives FDA Clearance for a 24-channel Small Body Array Compatible with Siemens Healthineers 3T Systems
PLEASANTON, Calif., Jan. 11, 2024 (GLOBE NEWSWIRE) — InkSpace Imaging, a leader in innovative diagnostic medical device technology, is proud to announce it received FDA clearance for its next-generation Small Body Array for the Siemens Healthineers Magnetom Skyra and Vida series 3T MRI scanners. This new medical device is part […]
United Imaging Intelligence Highlights Dedications to Advancing Intelligent Healthcare at RSNA Conference 2023
BOSTON, Jan. 11, 2024 /PRNewswire/ — The Radiological Society of North America (RSNA) has concluded its annual meeting in Chicago, IL, serving as a platform for revolutionary advancements in the field of radiology. United Imaging Intelligence (UII), an innovative subsidiary of United…