TRICAV II will evaluate the safety and effectiveness of the TricValve® Transcatheter Bicaval Valve System WILMINGTON, Del., Dec. 12, 2025 /PRNewswire/ — P&F USA, Inc., the U.S. subsidiary of heart valve manufacturer P&F Products and Features GmbH, today announced that the U.S. Food and…
Regulatory
SeaStar Medical Announces FDA Approval of Reduction in Mandatory Enrollment for SAVE Surveillance Registry Evaluating QUELIMMUNE™ Safety for Pediatric AKI
FDA lowers surveillance requirement to 50 patients from 300 patients in original HDE approval based on assessment of first 21 SAVE Surveillance Registry patients Compelling efficacy data to date along with completion of registry expected to expand QUELIMMUNE market opportunity DENVER, Dec. 02, 2025 (GLOBE NEWSWIRE) — SeaStar Medical Holding Corporation (Nasdaq: ICU), a commercial-stage healthcare company, announced today the Center for Biologics Evaluation and Research (CBER) of the U.S. Food and Drug Administration (FDA) has approved a reduction in the mandatory enrollment size of the SAVE Surveillance Registry from the originally-required 300 patients to only 50 patients. This new 50-patient registry size will satisfy the post-approval surveillance requirement by the FDA as outlined in the original Humanitarian Device Exemption (HDE) approval and indicates that FDA believes that continued device safety can be successfully shown with fewer patients than originally required. The SAVE Surveillance Registry is a Post-Approval Study (PAS) that is designed to confirm the safety of the QUELIMMUNE therapy, which was approved in 2024 as a treatment for children with acute kidney injury (AKI) and sepsis or a septic condition. To date, data from 32 pediatric patients have been entered into the SAVE Surveillance Registry. “We are grateful for this decision by the FDA and believe, once the registry is complete, it will enable us to expand the potential organ-sparing and life-saving benefits of our QUELIMMUNE therapy to more pediatric patients with AKI,” stated Eric Schlorff, CEO of SeaStar Medical. “These kids typically have about a 50% survival rate and our data from the first 21 patients in the SAVE Surveillance Registry showed zero device-related adverse events and a clear improvement to 70% survival at 90 days. This decision is a win-win for patients, their families, health care providers, and the overall health care system. We thank the FDA for their rapid evaluation of the required enrollment size of the mandatory surveillance requirements for the registry.” Preliminary results from the SAVE Surveillance Registry were recently presented at the 5th International Symposium on Acute Kidney Injury in Children. The SAVE Surveillance Registry presentation highlighted the role of QUELIMMUNE (Selective Cytopheretic Device for Pediatrics, or SCD-PED) therapy in the treatment of critically ill pediatric patients with life-threatening Acute Kidney Injury (AKI) and sepsis requiring Renal Replacement Therapy (RRT). Data from the first 21 pediatric patients with AKI and sepsis requiring RRT showed zero device-related adverse events or infections and no reports of immunosuppressive effects by the device. In addition, outcomes analyses show a 76% survival rate at Day 28 and Day 60, and a 71% survival rate at Day 90. These new data are on track to validate a 50% reduction in loss of life compared to historical data, as reported in Kidney Medicine. “The SAVE Surveillance Registry has provided valuable clinical outcomes data on both the safety and efficacy of our QUELIMMUNE therapy, but it has also slowed the adoption process and potential to treat these pediatric patients due to the complexities of setting up and running a patient registry at these medical institutions,” stated Tim Varacek, Senior Vice President, Commercial and Business Operations. “There has been very strong interest in QUELIMMUNE adoption and we are pleased that we can satisfy FDA’s registry requirements with far fewer patients than expected, enabling a smoother adoption process and potentially expanding the revenue opportunity to capture a larger portion of the total pediatric AKI market which we estimate to be about $100 million in the U.S. alone.” The QUELIMMUNE therapy has been adopted by some of the highest rated children’s medical centers in the United States. The patented technology behind QUELIMMUNE is known as the Selective Cytopheretic Device (SCD) therapy and has broad applications for treating the destructive hyperinflammation that shuts down organ function and causes loss of life. About Acute Kidney Injury (AKI) and Hyperinflammation AKI is characterized by a sudden and temporary loss of kidney function and can be caused by a variety of conditions such as sepsis, severe trauma, surgery, and COVID-19. AKI can cause destructive hyperinflammation, which is the overproduction or overactivity of inflammatory effector cells and other molecules that can be toxic. Damage resulting from this destructive hyperinflammation in AKI can progress to other organs, such as the heart or liver, and potentially to multi-organ dysfunction or even failure that could result in worse outcomes, including increased risk of death. Even after resolution, these patients may face complications including chronic kidney disease or end-stage renal disease (ESRD) requiring dialysis. Extreme hyperinflammation may also contribute to added healthcare costs, such as prolonged ICU stays and increased reliance on dialysis and mechanical ventilation. About QUELIMMUNE The QUELIMMUNE™ therapy is being commercialized for children with AKI and sepsis or septic condition weighing 10 kilograms or more who are on antibiotics and being treated in the ICU with Renal Replacement Therapy (RRT). It was approved in February 2024 under a HDE application that requires medical institutions to also participate in the SAVE Surveillance Registry and complete Institutional Review Board approvals prior to adoption and use of the QUELIMMUNE therapy. This prolongs the adoption timeline by medical institutions, but provides important data on the use of QUELIMMUNE in the “real-world” setting. Data from two clinical studies of the QUELIMMUNE therapy, published in Kidney Medicine, showed a 77% survival rate in patient treated with QUELIMMUNE versus standard of care, representing an approximate 50% reduction in loss of life compared to historical data in this patient population. No dialysis was required for survivors and 87.5% of survivors had normal kidney function at Day 60 after ICU discharge. Recent data presented from the first 21 patients in the SAVE Surveillance Registry mirror the clinical trial results. In January 2025, SeaStar Medical was awarded the 2025 Corporate Innovator Award by the National Kidney Foundation for its significant contribution to improving the lives of pediatric patients with AKI based on the approval and introduction of the QUELIMMUNE therapy. About the NEUTRALIZE-AKI Pivotal Trial The NEUTRALIZE-AKI (NEUTRophil and monocyte deActivation via SeLective Cytopheretic Device – a randomIZEd clinical trial in Acute Kidney Injury) pivotal trial is evaluating the safety and efficacy of the SCD therapy in 200 adults with AKI in the ICU receiving CRRT. The trial’s primary endpoint is a composite of 90-day mortality or dialysis dependency of patients treated with the SCD therapy in addition to CRRT as the standard of care, compared with the control group receiving only CRRT standard of care. Secondary endpoints include mortality at 28 days, ICU-free days in the first 28 days, major adverse kidney events at Day 90 and dialysis dependency at one year. The study will also include subgroup analyses to explore the effectiveness of the SCD therapy in AKI patients with sepsis and acute respiratory distress syndrome. About the SeaStar Medical Selective Cytopheretic Device Therapy The Selective Cytopheretic Device (SCD) therapy is designed as a disease-modifying device that neutralizes over-active immune cells and stops the cytokine storm that yields destructive hyperinflammation and creates a cascade of events that wreak havoc in the patient’s body. The SCD therapy has broad applications in multiple acute and chronic kidney and cardiovascular diseases, representing patients who today have no FDA-approved options for treating their disease. Unlike pathogen removal and other blood-purification tools, the SCD therapy is integrated with an existing CRRT hemofiltration system to selectively target and transition proinflammatory monocytes to a reparative state and promote activated neutrophils to be less inflammatory. This unique immunomodulation approach may promote long-term organ recovery, eliminate the need for future RRT, including dialysis, and prevent loss of life. About SeaStar Medical SeaStar Medical is a commercial-stage healthcare company focused on transforming treatments for critically ill patients facing organ failure and potential loss of life. SeaStar’s first commercial product, QUELIMMUNE (SCD-PED), was approved in 2024 by the U.S. Food and Drug Administration (FDA). It is the only FDA approved product for the ultra-rare condition of life-threatening acute kidney injury (AKI) due to sepsis or a septic condition in critically ill pediatric patients. SeaStar’s Selective Cytopheretic Device (SCD) therapy has been awarded Breakthrough Device Designation for six therapeutic indications by the FDA, enabling the potential for a speedier pathway to approval and preferable reimbursement dynamics at commercial launch. The company is currently conducting a pivotal trial of its SCD therapy in adult patients with AKI requiring continuous renal replacement therapy (CRRT), a life-threatening condition with no effective treatment options that impacts over 200,000 adults in the U.S. annually. For more information visit www.seastarmedical.com or visit us on LinkedIn or X. Forward-Looking Statements This press release contains certain forward-looking statements within the meaning of the “safe harbor” provisions of the Private Securities Litigation Reform Act of 1955. These forward-looking statements include, without limitation, SeaStar Medical’s expectations with respect to the anticipated adoption our products; anticipated cost savings for patients, healthcare providers, and the healthcare system more generally from the adoption of the SCD therapy; the total addressable market for pediatric SCD applications; the ability of SeaStar Medical to increase market share and generate sales with respect to the total addressable market for pediatric SCD applications; the ability of SCD to treat patients with AKI and other diseases; the expected regulatory approval process and timeline for commercialization; and the ability of SeaStar Medical to meet the expected timeline. Words such as “believe,” “project,” “expect,” “anticipate,” “estimate,” “intend,” “strategy,” “future,” “opportunity,” “plan,” “may,” “should,” “will,” “would,” “will be,” “will continue,” “will likely result,” and similar expressions are intended to identify such forward-looking statements. Forward-looking statements are predictions, projections and other statements about future events that are based on current expectations and assumptions and, as a result, are subject to significant risks and uncertainties that could cause the actual results to differ materially from the expected results. Most of these factors are outside SeaStar Medical’s control and are difficult to predict. Factors that may cause actual future events to differ materially from the expected results include, but are not limited to: (i) the risk that SeaStar Medical may not be able to obtain regulatory approval of its SCD product candidates; (ii) the risk that SeaStar Medical may not be able to raise sufficient capital to fund its operations, including current or future clinical trials; (iii) the risk that SeaStar Medical and its current and future collaborators are unable to successfully develop and commercialize its products or services, or experience significant delays in doing so, including failure to achieve approval of its products by applicable federal and state regulators, (iv) the risk that SeaStar Medical may never achieve or sustain profitability; (v) the risk that SeaStar Medical may not be able to secure additional financing on acceptable terms; (vi) the risk that third-party suppliers and manufacturers are not able to fully and timely meet their obligations, (vii) the risk of product liability or regulatory lawsuits or proceedings relating to SeaStar Medical’s products and services, (viii) the risk that SeaStar Medical is unable to secure or protect its intellectual property, and (ix) other risks and uncertainties indicated from time to time in SeaStar Medical’s Annual Report on Form 10-K, including those under the “Risk Factors” section therein and in SeaStar Medical’s other filings with the SEC. The foregoing list of factors is not exhaustive. Forward-looking statements speak only as of the date they are made. Readers are cautioned not to put undue reliance on forward-looking statements, and SeaStar Medical assumes no obligation and do not intend to update or revise these forward-looking statements, whether as a result of new information, future events, or otherwise. Contact: IR@SEASTARMED.COM
RapidAI Earns FDA Clearance for Rapid Aortic, Bringing AI-Driven Aortic Measurements and Surveillance to Care Teams
SAN MATEO, Calif.–(BUSINESS WIRE)–RapidAI, the pioneer of deep clinical AI and global leader in enterprise imaging, today announced U.S. Food and Drug Administration (FDA) clearance of Aortic Management, part of the Rapid Aortic product, a comprehensive deep clinical AI solution designed to transform the acute assessment and longitudinal management of aortic disease. […]
ViTAA Medical Secures FDA 510(k) Clearance for AiORTA™ Plan — Marking the First Step in Its Hyper-Precise Aortic Care Platform
MONTREAL–(BUSINESS WIRE)–ViTAA Medical Solutions Inc. today announced that the U.S. Food and Drug Administration (FDA) has granted 510(k) clearance for AiORTA™ Plan, the company’s fully automated, hyper-precise aortic surgery planning solution. This clearance represents a key milestone in the evolution of ViTAA’s broader AiORTA™ platform, built to support physicians across the full continuum of […]
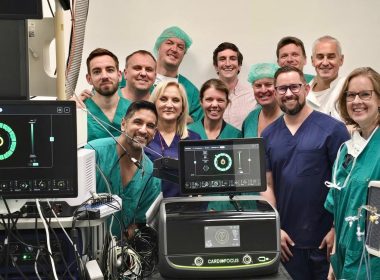
CardioFocus Announces First Enrollments in CE Mark Clinical Trial of QuickShot™ PFA Catheter and CardioWave™ PFA System
MARLBOROUGH, Mass.–(BUSINESS WIRE)–CardioFocus, Inc., a medical device company dedicated to advancing ablation treatment for cardiac arrhythmias, today announced a series of major milestones advancing its pulsed field ablation (PFA) program. The company has initiated its CE Mark pivotal clinical trial for the QuickShot™ Nav large-area focal PFA Catheter and the […]
GenesisX Robotic Magnetic Navigation System Receives U.S. FDA Clearance
ST. LOUIS, Nov. 10, 2025 (GLOBE NEWSWIRE) — Stereotaxis (NYSE: STXS), a pioneer and global leader in surgical robotics for minimally invasive endovascular intervention, today announced that it received U.S. Food and Drug Administration 510(k) clearance for its latest generation robotic system, GenesisX™.
Venova Medical Announces First Subjects Enrolled in VENOS-3 Pivotal IDE Study of the Velocity® Percutaneous AVF System
The Velocity System is a next generation percutaneous AVF technology designed to reduce the need for reinterventions to achieve fistula maturation and improve hemodialysis vascular access outcomes while reducing costs. LOS GATOS, Calif., Nov. 6, 2025 /PRNewswire-PRWeb/ — Venova Medical,…
FDA Approves Anteris’s DurAVR® THV Global Pivotal Trial (the “PARADIGM Trial”)
PARADIGM: A Prospective rAndomized tRial Assessing the safety and effectiveness of the DurAVR® bIomimetic valve designed for physioloGic flow compared to CoMmercial TAVR devices MINNEAPOLIS and BRISBANE, Australia, Nov. 03, 2025 (GLOBE NEWSWIRE) — Anteris announced today it has received U.S. Food and Drug Administration (FDA) approval to initiate PARADIGM, its global Investigational Device Exemption (IDE) clinical trial which is designed to evaluate the DurAVR® Transcatheter Heart Valve (THV) in patients with severe calcific aortic stenosis and to support a future PMA* submission. “We are extremely pleased to receive FDA approval for the PARADIGM Trial, which allows us to commence patient recruitment in the United States**. This milestone, together with the recent launch of the trial and first patients treated in Denmark, represents a significant achievement and a key step forward in advancing this life-saving technology worldwide for patients living with aortic stenosis, a debilitating and progressive condition,” commented Vice Chairman and CEO, Wayne Paterson. The PARADIGM Trial is co-chaired by Dr. Michael J. Reardon, Allison Family Distinguished Chair of Cardiovascular Research and Professor of Cardiothoracic Surgery at the Houston Methodist Hospital, Texas and Professor Stephan Windecker, Chairman of the Department of Cardiology at Bern University Hospital, Switzerland. About the PARADIGM Trial The PARADIGM Trial is a prospective randomized controlled trial (RCT) which will evaluate the safety and effectiveness of the DurAVR® THV compared to commercially available transcatheter aortic valve replacements (TAVRs). This head-to-head study will enroll approximately 1000 patients across the Unites States, Europe and Canada in the ‘All Comers Randomized Cohort’ with 1:1 randomization of patients who will receive either the DurAVR® THV or TAVR using commercially available and approved THVs. The PARADIGM Trial will assess non-inferiority on a primary composite endpoint of all-cause mortality, all stroke and cardiovascular hospitalization at one year post procedure. The PARADIGM Trial is designed to provide the robust clinical evidence required to support an application to the FDA for Premarket Approval in the United States, with CE Mark approval anticipated to progress in parallel to the PMA. For further information, please refer to ClinicalTrials.gov (ClinicaTrials.gov ID NCT07194265). *A Premarket Approval (PMA) application requires a high level of clinical evidence to demonstrate reasonable assurance of safety and effectiveness for the intended use. Randomized controlled trials are generally considered Level 1 evidence, the highest level for determining the effectiveness of interventions in evidence-based medicine given RCTs minimize bias and allow a clear comparison between treatment groups. **Subject to Institutional Review Board (IRB) approval About Anteris Anteris Technologies Global Corp. (NASDAQ: AVR, ASX: AVR) is a global structural heart company committed to designing, developing, and commercializing cutting-edge medical devices to restore healthy heart function. Founded in Australia, with a significant presence in Minneapolis, USA, Anteris is a science-driven company with an experienced team of multidisciplinary professionals delivering restorative solutions to structural heart disease patients. Anteris’ lead product, the DurAVR® Transcatheter Heart Valve (THV), was designed in partnership with the world’s leading interventional cardiologists and cardiac surgeons to treat aortic stenosis – a potentially life-threatening condition resulting from the narrowing of the aortic valve. The balloon-expandable DurAVR® THV is the first biomimetic valve, which is shaped to mimic the performance of a healthy human aortic valve and aims to replicate normal aortic blood flow. DurAVR® THV is made using a single piece of molded ADAPT® tissue, Anteris’ patented anti-calcification tissue technology. ADAPT® tissue, which is FDA-cleared, has been used clinically for over 10 years and distributed for use in over 55,000 patients worldwide. The DurAVR® THV System is comprised of the DurAVR® valve, the ADAPT® tissue, and the balloon-expandable ComASUR® Delivery System. Forward-Looking Statements This announcement contains forward-looking statements including statements regarding the intent for the PARADIGM Trial. Forward-looking statements include all statements that are not historical facts. Forward-looking statements generally are identified by the words “believe,” “project,” “expect,” “anticipate,” “estimate,” “intend,” “budget,” “target,” “aim,” “strategy,” “plan,” “guidance,” “outlook,” “may,” “should,” “could,” “will,” “would,” “will be,” “will continue,” “will likely result” and similar expressions, although not all forward-looking statements contain these identifying words. These forward-looking statements are subject to a number of risks, uncertainties, and assumptions, including those described under “Risk Factors” in Anteris’ Annual Report on Form 10-K for the fiscal period ended December 31, 2024 that was filed with the SEC and ASX. Readers are cautioned not to put undue reliance on forward-looking statements, and except as required by law, neither ATL or Anteris assume any obligation to update any of these forward-looking statements to conform these statements to actual results or revised expectations. For more information: Investor Relationsinvestor@anteristech.comDebbie OrmsbyAnteris Technologies Global Corp.+61 1300 550 310 | +61 7 3152 3200 Investor Relations (US)mchatterjee@bplifescience.comMalini Chatterjee, Ph.D.Blueprint Life Science Group+1 917 330 4269 Website www.anteristech.comX @AnterisTechLinkedIn https://www.linkedin.com/company/anteristech
Anteris Receives First European Regulatory Clearance to Commence DurAVR® THV Global Pivotal Trial (the “PARADIGM Trial”)
MINNEAPOLIS and BRISBANE, Australia, Oct. 15, 2025 (GLOBE NEWSWIRE) — Anteris Technologies Global Corp. (Anteris or the Company) (NASDAQ: AVR, ASX: AVR) a global structural heart company committed to designing, developing, and commercializing cutting-edge medical devices to restore healthy heart function, announced today it has received regulatory clearance from the Danish Medicines Agency to initiate the DurAVR® Transcatheter Heart Valve (THV) global pivotal trial in patients with severe calcific aortic stenosis (the “PARADIGM Trial”). Patient recruitment at Danish centers is expected to begin in 4Q 2025. PARADIGM: A Prospective rAndomized tRial Assessing the safety and effectiveness of the DurAVR® bIomimetic valve designed for physioloGic flow compared to CoMmercial TAVR devices With the first regulatory clearance secured, Anteris is positioned to drive the global PARADIGM Trial through the addition of further countries and sites in the near term, with planned expansion across the United States, Europe and Canada. Management believes strong enthusiasm from investigators is expected to translate into efficient recruitment and timely study advancement. “Receiving initial approval in Europe is an important milestone as it signals the launch of the global PARADIGM Trial. This groundbreaking study has been designed with world-leading experts and is attracting significant interest from clinicians globally, reflecting the potential for DurAVR® to transform care for patients with aortic stenosis,” said Anteris Chief Medical Officer, Chris Meduri, M.D. “The PARADIGM Trial enables us to bring promising new technology to patients across all surgical risk groups, building on the growing body of evidence supporting the DurAVR® THV’s favorable hemodynamic performance. We look forward to contributing meaningful data which could support both PMA* and CE Mark approvals,” said Dr. Michael Reardon and Professor Stephan Windecker, Study Co-Chairs of the global PARADIGM Trial. “The PARADIGM trial is a multi center global study which, if successful, will result in multiple approvals and labels for the DurAVR® THV. This is a watershed moment in the company’s life cycle and marks the beginning of the commercialization planning phase as we march towards global approvals. Anteris is excited to be able to allow access to an increasing pool of patients globally who will benefit from this life saving technology. The commencement of this trial reflects the commitment and tireless work of our talented Anteris team, as well as our Physician and Scientific Advisors. The company would like to express its gratitude to the physicians, patients and shareholders who have been fundamental in developing this important new therapy,” said Vice Chairman and CEO, Wayne Paterson. In parallel, an Investigational Device Exemption (IDE) application remains under review by the U.S. Food and Drug Administation (FDA). Anteris continues to expect FDA approval in the near term, which will allow initiation of study sites in the PARADIGM Trial in the United States, pending Institutional Review Board (IRB) approval. About the PARADIGM Trial The PARADIGM Trial is a prospective randomized controlled trial (RCT) which will evaluate the safety and effectiveness of the DurAVR® THV compared to commercially available transcatheter aoric valve replacements (TAVRs). This head-to-head study will enroll approximately 1000 patients in the ‘All Comers Randomized Cohort’ with 1:1 randomization of patients who will receive either the DurAVR® THV or TAVR using commercially available and approved THVs. The PARADIGM Trial will assess non-inferiority on a primary composite endpoint of all-cause mortality, all stroke and cardiovascular hospitalization at one year post procedure. For further information, please refer to ClinicalTrials.gov (ClinicaTrials.gov ID NCT07194265). The planned expansion across other geographies includes additional cohorts. *A Premarket Approval (PMA) application requires a high level of clinical evidence to demonstrate reasonable assurance of safety and effectiveness for the intended use. Randomized controlled trials are generally considered Level 1 evidence, the highest level for determining the effectiveness of interventions in evidence-based medicine given RCTs minimize bias and allow a clear comparison between treatment groups. About Anteris Anteris Technologies Global Corp. (NASDAQ: AVR, ASX: AVR) is a global structural heart company committed to designing, developing, and commercializing cutting-edge medical devices to restore healthy heart function. Founded in Australia, with a significant presence in Minneapolis, USA, Anteris is a science-driven company with an experienced team of multidisciplinary professionals delivering restorative solutions to structural heart disease patients. Anteris’ lead product, the DurAVR® Transcatheter Heart Valve (THV), was designed in partnership with the world’s leading interventional cardiologists and cardiac surgeons to treat aortic stenosis – a potentially life-threatening condition resulting from the narrowing of the aortic valve. The balloon-expandable DurAVR® THV is the first biomimetic valve, which is shaped to mimic the performance of a healthy human aortic valve and aims to replicate normal aortic blood flow. DurAVR® THV is made using a single piece of molded ADAPT® tissue, Anteris’ patented anti-calcification tissue technology. ADAPT® tissue, which is FDA-cleared, has been used clinically for over 10 years and distributed for use in over 55,000 patients worldwide. The DurAVR® THV System is comprised of the DurAVR® valve, the ADAPT® tissue, and the balloon-expandable ComASUR® Delivery System. Forward-Looking Statements This announcement contains forward-looking statements, including statements regarding the planned expansion of the PARADIGM Trial, the results of the PARADIGM Trial, the contours of the PARADIGM Trial, and the timing of the IDE approval. Forward-looking statements include all statements that are not historical facts. Forward-looking statements generally are identified by the words “believe,” “project,” “expect,” “anticipate,” “estimate,” “intend,” “budget,” “target,” “aim,” “strategy,” “plan,” “guidance,” “outlook,” “may,” “should,” “could,” “will,” “would,” “will be,” “will continue,” “will likely result” and similar expressions, although not all forward-looking statements contain these identifying words. These forward-looking statements are subject to a number of risks, uncertainties, and assumptions, including those described under “Risk Factors” in Anteris’ Annual Report on Form 10-K for the fiscal period ended December 31, 2024 that was filed with the SEC and ASX. Readers are cautioned not to put undue reliance on forward-looking statements, and except as required by law, Anteris does not assume any obligation to update any of these forward-looking statements to conform these statements to actual results or revised expectations. For more information: Investor Relations Investor Relations (US)investor@anteristech.com mchatterjee@bplifescience.comDebbie Ormsby Malini Chatterjee, Ph.D.Anteris Technologies Global Corp. Blueprint Life Science Group+61 1300 550 310 | +61 7 3152 3200 +1 917 330 4269 Websitewww.anteristech.com X@AnterisTech LinkedInhttps://www.linkedin.com/company/anteristech
Vasa Therapeutics Receives FDA IND Clearance to Advance Novel Small Molecule VS-041 for the Treatment of Heart Failure with Preserved Ejection Fraction (HFpEF)
Phase 1c Clinical Trial to Begin Immediately ENCINITAS, Calif. and WROCLAW, Poland, Oct. 13, 2025 /PRNewswire/ — Vasa Therapeutics, (“Vasa”), a private, clinical stage biotechnology company developing novel therapies for cardiovascular and metabolic aging, today announced that the U.S….