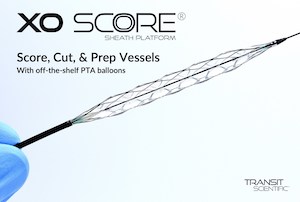
New metal-alloy scoring sheath CE Mark cleared for treatment of peripheral artery disease and end stage renal disease. PARK CITY, Utah, May 10, 2021 /PRNewswire/ — Transit Scientific announced it received CE Mark clearance in the European Union for the XO Score® Scoring Sheath Platform to facilitate dilation of stenotic material in the peripheral vasculature […]
Regulatory
Micro Medical Solutions Receives FDA Breakthrough Device Designation for MicroStent Vascular Stent
Breakthrough Designation Helps Advance Treatment Options, Targeting a Significant Unmet Need in the Treatment of CLTI. WILMINGTON, Mass., May 4, 2021 /PRNewswire/ — Micro Medical Solutions (MMS), an emerging leader in the treatment of CLTI, today announced that the U.S. Food and Drug Administration (FDA) has granted Breakthrough Device Designation for its […]
Biobeat’s AI-Powered Remote Patient Monitoring Platform Receives A Full CE Mark
Remote Patient Monitoring Platform Provides Continuous Vital Sign Measurement and Patient Deterioration Identification Capabilities In Hospital and Home Settings PETAH TIKVA, Israel , May 4, 2021 /PRNewswire/ — Biobeat, a global leader in wearable remote patient monitoring solutions for the healthcare continuum, announced today that its full AI-powered wearable remote patient monitoring platform has received a […]
AtriCure’s EPi-Sense System Approved by FDA for Treatment of Long-Standing Persistent Afib Patients
FDA approval results in the only label of its kind for more than 3 million patients in the United States, significantly expanding AtriCure’s addressable market Superiority trial showed a 29% difference in effectiveness at 12 months and a 35% difference in effectiveness at 18 months for long-standing persistent Afib patients. Study […]
FDA Clears IMS CloudVue for Mobile 3D with Patented Cinematic Rendering
Giving IMS a First Mover Advantage – The Only 100% Cloud-Based FDA Cleared Cinematic Solution on the Market TORONTO, April 28, 2021 /PRNewswire/ — International Medical Solutions (“IMS”), one of the first cloud-native technologies to enable the remote, seamless, and instant view of medical DICOM images in the cloud received FDA Class II […]
Soundbite Medical Solutions Announces FDA Approval for SoundBite® Crossing System – Peripheral (14P)
MONTREAL–(BUSINESS WIRE)–Soundbite announces FDA 510(k) Approval for SoundBite® Crossing System – Peripheral (SCS-P) with the 0.014” Active Wire (14P). The SoundBite® Crossing System – Peripheral (14P) is a recanalization tool, designed to help physician’s placement of conventional guidewires or treatment devices in the intraluminal space beyond chronic total occlusions (CTOs) in […]
Cerus Endovascular Announces IDE Approval from the U.S. FDA to Conduct Clinical Study of the Contour Neurovascular System™
Patient Enrollment to Begin Within Three Months FREMONT, Calif. and OXFORD, United Kingdom, April 21, 2021 /PRNewswire/ — Cerus Endovascular Ltd., a privately held, commercial-stage medical device company, today announced that the U.S. Food and Drug Administration (FDA) has approved its Investigational Device Exemption (IDE) application to conduct a U.S. trial for the Contour […]
Medtronic Receives U.S. FDA Approval for Pipeline™ Flex Embolization Device with Shield Technology™
First Patient Receives New Shield Surface Modification, An Advance in Flow Diversion Therapy, at NYU Langone Health DUBLIN, April 21, 2021 /PRNewswire/ — Medtronic plc (NYSE:MDT), the global leader in medical technology, today announced it has received approval from the United States Food and Drug Administration (FDA) for Pipeline™ Flex Embolization Device […]
LivaNova Receives FDA 510(k) Clearance for B-Capta, the New In-Line, Blood-Gas Monitoring System Integrated Into the S5 Heart-Lung Machine
Intuitive, optical-based technology system now available around the world LONDON–(BUSINESS WIRE)–LivaNova PLC (NASDAQ:LIVN), a market-leading medical technology and innovation company, today announced it received U.S. Food and Drug Administration (FDA) 510(k) clearance for B-Capta®, the new in-line, blood-gas monitoring system integrated into the market-leading S5® heart-lung machine (HLM). The system is […]
Contego Medical Receives CE Mark Approval for Next Generation Carotid Stent
RALEIGH, N.C., April 16, 2021 /PRNewswire/ — Contego Medical, Inc., a leading developer of innovative cardiovascular devices, today announced receipt of CE Mark approval for the Neuroguard IEP® 3-in-1 Carotid Stent and Post-Dilation Balloon System with Integrated Embolic Protection (Neuroguard IEP System). The patented Neuroguard IEP System contains a novel, next-generation nitinol stent, a […]