Clinical Trial Demonstrates Procedural Efficiencies, Safety, Effectiveness and Non-Inferiority of DiamondTemp System DUBLIN, Jan. 29, 2021 /PRNewswire/ — Medtronic plc (NYSE:MDT), the global leader in medical technology, today announced it has received U.S. Food and Drug Administration (FDA) approval of the DiamondTemp™ Ablation (DTA) system which treats patients with recurrent, symptomatic paroxysmal atrial […]
Regulatory
Alleviant Medical Receives Breakthrough Device Designation From FDA for Transcatheter Technology
Innovative technology offers an implant-free approach for individuals with chronic heart failure AUSTIN, Texas–(BUSINESS WIRE)–Alleviant Medical Inc., a privately-held medical device company, today announced that the US Food and Drug Administration (FDA) has granted the company a Breakthrough Device designation for its transcatheter technology. The technology offers a no-implant interatrial […]
PEDRA™ Technology Receives FDA Breakthrough Device Designation for its PEDRA™ Xauron™ Real-Time Tissue Perfusion System
Novel perfusion monitor achieves FDA Breakthrough Device Designation for real-time, periprocedural monitoring of tissue perfusion in patients with critical limb threatening ischemia SINGAPORE, Jan 25, 2021 /PRNewswire/ — PEDRA™ Technology, a privately-held company, announced today that the U.S Food and Drug Administration (FDA) has granted the company a Breakthrough Device Designation for the […]
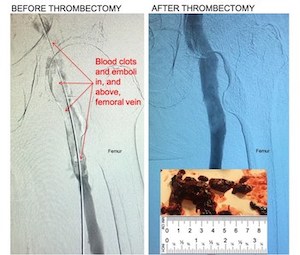
Kishor Vora, M.D. Owensboro Heart & Vascular, Removes Large Blood Clots in 1st Global Use of New Device
11F mechanical thrombectomy system removes large blood clots in deep vein thrombosis (DVT) HALLANDALE BEACH, Flo., Jan. 22, 2021 /PRNewswire/ — Control Medical Technology announced the FDA cleared Control 11F Mechanical Thrombectomy system was used to remove large blood clots from patients with deep vein thrombosis (DVT). “Control removed large blood […]
Canon Medical’s AI-Powered, Premium Large Bore CT Receives FDA Clearance
Aquilion Exceed LB CT System Offers Industry’s Largest Bore and Widest Field-of-View TUSTIN, Calif.–(BUSINESS WIRE)–Canon Medical Systems USA, Inc. has received FDA clearance for the Aquilion Exceed LB™ CT system, giving clinicians the opportunity to see more during radiation therapy planning for accuracy, precision and speed. As cancer cases continue to […]
Merck Announces U.S. FDA Approval of VERQUVO® (vericiguat)
VERQUVO Approved for Reduction of Risk of Cardiovascular Death and Heart Failure (HF) Hospitalization Following a Hospitalization for HF or Need for Outpatient Intravenous (IV) Diuretics in Adults with Symptomatic Chronic Heart Failure and Ejection Fraction Less than 45% VERQUVO is the First Soluble Guanylate Cyclase Stimulator, Approved to Treat […]
Impulse Dynamics Announces FDA Approval for Magnetic Resonance Imaging
FDA Clears Potential Hurdle for Many Heart Failure Patients MARLTON, N.J.–(BUSINESS WIRE)–Impulse Dynamics, a company dedicated to improving the lives of people with heart failure (HF), today announced the U.S. Food and Drug Administration (FDA) has approved the conditional use of Magnetic Resonance Imaging (MRI) for Optimizer® CCM® delivery systems. This approval […]
Occlutech’s Atrial Flow Regulator (AFR) Receives U.S. FDA Breakthrough Device Designation for Heart Failure (HF)
SCHAFFHAUSEN, Switzerland, Jan. 19, 2021 /PRNewswire/ — Occlutech, a privately-held company, announced today that the U.S Food and Drug Administration (FDA) has granted the Company a Breakthrough Device designation for its first-in-class, implantable Atrial Flow Regulator (AFR) for heart failure (HF) patients with preserved (HFpEF) or reduced (HFrEF) ejection fraction. Heart failure (HF) […]
US FDA accepts supplemental New Drug Application for Jardiance® (empagliflozin) for adults with heart failure with reduced ejection fraction
RIDGEFIELD, Conn. and INDIANAPOLIS, Jan. 11, 2021 /PRNewswire/ — The U.S. Food and Drug Administration (FDA) has accepted a supplemental New Drug Application (sNDA) for Jardiance® (empagliflozin) which is being investigated as a potential new treatment to reduce the risk of cardiovascular death and hospitalization for heart failure and to slow kidney function decline in adults with […]
Ultromics receives FDA Clearance for a first-of-kind solution in Echocardiography to help clinicians diagnose disease
EchoGo Pro is an AI service for automated identification of coronary artery disease – FDA gives 510(K) clearance for EchoGo Pro, expanding the use of Ultromics’ EchoGo service to support clinicians diagnosing coronary artery disease – Every year in the U.S. over 800,000 people die from heart disease – accounting […]