WESTMINSTER, Colo., Dec. 20, 2018 (GLOBE NEWSWIRE) — ARCA biopharma, Inc. (Nasdaq: ABIO), a biopharmaceutical company applying a precision medicine approach to developing genetically-targeted therapies for cardiovascular diseases, today announced that it has submitted an amendment to its Special Protocol Assessment (SPA)request to the U.S. Food and Drug Administration (FDA). The amendment addresses […]
Regulatory
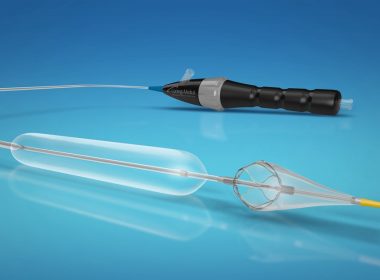
CathWorks FFRangio™ System Receives U.S. FDA Clearance
KFAR-SABA, Israel & ALISO VIEJO, Calif.–(BUSINESS WIRE)–CathWorks announced today that its FFRangio™ System received United States Food & Drug Administration (FDA) 510(k) clearance. The FFRangio system demonstrated accuracy versus the invasive FFR wire in a blinded comparative study, FAST-FFR. The results of the FAST-FFR pivotal study were used to establish substantial […]
Paragonix Technologies, Inc., Announces 510(k) Clearance Allowing for Use of the Paragonix SherpaPak™ Cardiac Transport System with Pediatric and Small Donor Hearts
BRAINTREE, Mass.–(BUSINESS WIRE)–Paragonix Technologies, Inc. today announced that it received clearance by the Food and Drug Administration (FDA) for a design enhancement allowing for the use of the Paragonix SherpaPak™ Cardiac Transport System (CTS)1,2 with small and pediatric donor hearts. Starting in Q1 2019, Paragonix SherpaPak™ CTS will now be shipped with […]
Cardiva Medical Announces FDA Approval of the VASCADE MVP Vascular Closure System for Multi-Site Vessel Closure Following Electrophysiology Procedures
SANTA CLARA, Calif.–(BUSINESS WIRE)–Cardiva Medical, Inc., an innovator in the field of vascular closure, today announced that the company has received PMA approval from the U.S. Food and Drug Administration (FDA) for the VASCADE® MVP Venous Vascular Closure System. VASCADE MVP is the first and only vascular closure system designed […]
Hancock Jaffe Receives Approval for First-in-Human VenoValve Study
IRVINE, Calif., Dec. 17, 2018 (GLOBE NEWSWIRE) — Hancock Jaffe Laboratories, Inc. (Nasdaq: HJLI, HJLIW), a company specializing in medical devices that restore cardiac and vascular health, announced today that it has received regulatory approval from INVIMA, the Colombian equivalent of the U.S. Food and Drug Administration, for its first-in-human […]
Aortica Corp. Announces FDA Approves Medtronic Valiant NAVION™ for Inclusion in Starnes’ Physician-Sponsored IDE
BELLEVUE, Wash.–(BUSINESS WIRE)–Aortica Corporation today announced that FDA has approved a supplement to an ongoing Physician Sponsored IDE study at the University of Washington (UW) sponsored by Principal Investigator & Chief of Vascular Surgery Dr. Benjamin Starnes. The positive decision allows the use of Medtronic’s Valiant NAVION™ stent graft system […]
Contego Medical Receives 510(k) Clearance for the Vanguard IEP Peripheral Angioplasty System with Integrated Embolic Protection
RALEIGH, N.C., Dec. 7, 2018 /PRNewswire/ — Contego Medical announced today that the U.S. Food and Drug Administration (FDA) has granted 510(k) clearance for its Vanguard IEP® Peripheral Balloon Angioplasty System with Integrated Embolic Protection. Contego Medical is a medical device company developing and commercializing a suite of next-generation devices that address […]
JenaValve Technology Receives FDA Approval for Expanded IDE Enrollment in the Treatment of Patients with Severe Aortic Stenosis and Severe Aortic Regurgitation
IRVINE, Calif.–(BUSINESS WIRE)–JenaValve Technology, Inc., a developer and manufacturer of differentiated transcatheter aortic valve replacement (TAVR) systems, today announced U.S. Food and Drug Administration (FDA) approval of expansion of its Investigational Device Exemption (IDE) feasibility studies for the JenaValve Pericardial TAVR System with the Everdur™ transcatheter heart valve (THV) and […]
Cardinal Health Announces FDA Approval of the INCRAFT® AAA Stent Graft System
DUBLIN, Ohio, November 28, 2018 — Cordis, a Cardinal Health company (NYSE: CAH), today announced that the U.S. Food and Drug Administration (FDA) has approved its INCRAFT® AAA Stent Graft System for use in complex access anatomies. The INCRAFT system is an ultra-low profile and flexible endovascular aneurysm repair (EVAR) system designed […]
MERIT MEDICAL RECEIVES 510(K) APPROVAL FOR EMBOCUBE EMBOLIZATION GELATIN FOR EMBOLIZATION OF HYPERVASCULAR TUMORS
SOUTH JORDAN, Utah – November 29, 2018 – Merit Medical Systems, Inc. (NASDAQ:MMSI), a leading manufacturer and marketer of proprietary disposable devices used primarily in interventional, diagnostic and therapeutic procedures, announces that it has received 510(k) clearance for EmboCube Embolization Gelatin. EmboCube is indicated for use in embolization of hypervascular tumors. The […]