Stratford-upon-Avon, UK, 08 June 2018 – Celixir, a privately owned company discovering and developing life-saving advanced therapies, announces that the US Food and Drug Administration (FDA) has approved its Investigational New Drug application (IND) for HeartcelTM, its immuno-modulatory progenitor (iMP) cell therapy for the treatment of adult heart failure. Celixir announced […]
Regulatory
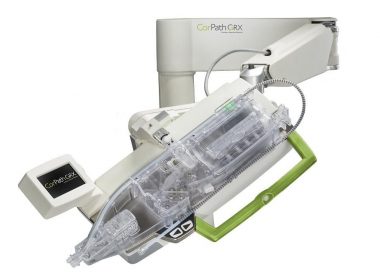
Corindus Announces Pharmaceutical and Medical Device Agency (PMDA) Approval of CorPath GRX System in Japan
WALTHAM, Mass.–(BUSINESS WIRE)–Corindus Vascular Robotics, Inc. [NYSE American: CVRS], a leading developer of precision vascular robotics, announced today that it received Pharmaceutical and Medical Device Agency (PMDA) Approval for commercialization of its CorPath® GRX System in Japan. Japan is one of the largest markets in the world for percutaneous coronary interventions […]
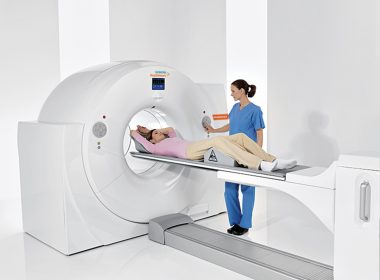
FDA Clears Biograph Vision PET/CT System From Siemens Healthineers
MALVERN, Pa.–(BUSINESS WIRE)–The Food and Drug Administration (FDA) has cleared the Biograph Vision, a new positron emission tomography/computed tomography system from Siemens Healthineers that delivers a new level of precision in PET/CT imaging. The Biograph Vision features new Optiso Ultra Dynamic Range (UDR) Detector Technology, which is based on silicon […]
FDA Classifies HeartWare(TM) HVAD(TM) Systems Unexpected Power Source Switching as Class I Recall
DUBLIN – June 1, 2018 – The United States Food and Drug Administration (FDA) has classified Medtronic plc’s (NYSE: MDT) recent voluntary urgent field action related to the HeartWare(TM) HVAD(TM) System unexpected power source switching as a Class I recall. Class I recalls describe situations where there is reasonable risk of […]
MicroVention® Announces FDA Approval For Neuro Stent Device
ALISO VIEJO, Calif., May 31, 2018 /PRNewswire/ — MicroVention, Inc., a U.S.-based subsidiary of Terumo and a global neurovascular company, announced today the FDA Premarket Approval (PMA) for the LVIS® and LVIS® Jr. stents for stent-assisted coil embolization of intracranial aneurysms. The LVIS® and LVIS® Jr. stents are the first and only stents PMA approved for […]
XableCath Receives Second FDA Clearance for Its Peripheral Arterial Catheters
SALT LAKE CITY–(BUSINESS WIRE)–XableCath, Inc., innovators of products designed to aid in the treatment of peripheral artery disease (PAD), announced that its second catheter, XableCath™ abrasion tip support catheter, was cleared by the FDA. The XableCath blunt tip catheter received FDA clearance at the end of 2017. This clearance gives […]
Avinger Receives FDA Clearance of Next Generation Pantheris Device
REDWOOD CITY, Calif., May 23, 2018 (GLOBE NEWSWIRE) — Avinger, Inc. (NASDAQ:AVGR), a leading developer of innovative treatments for peripheral artery disease (PAD), announced today that the Company received 510(k) clearance from the U.S. Food & Drug Administration (FDA) for its next generation Pantheris® Lumivascular atherectomy system, the first-ever image-guided […]
CERENOVUS Receives FDA Clearance For Next Generation Stent Retriever Device Used To Treat Ischemic Stroke
IRVINE, Calif., May 21, 2018 /PRNewswire/ — CERENOVUS, part of the Johnson & Johnson Medical Devices Companies, announced today it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for EMBOTRAP II Revascularization Device, a next generation stent retriever used to capture and remove life-threatening blood clots from […]
CardioFocus® Announces FDA Approval Of The Next-Generation HeartLight® Excalibur Balloon™ Designed For The Treatment Of Paroxysmal Atrial Fibrillation*
MARLBOROUGH, Mass., May 7, 2018 /PRNewswire/ — CardioFocus, Inc. today announced that the U.S. Food and Drug Administration (FDA) has granted approval for the HeartLight Excalibur Balloon, a next-generation technology designed for the treatment of paroxysmal atrial fibrillation (PAF)*. The Excalibur Balloon leverages the proven universal balloon design of the company’s FDA-approved HeartLight® Endoscopic Ablation System and introduces an […]
Abbott Expands Cardiac Arrhythmias Portfolio with FDA Clearance of Advanced Mapping Catheter
ABBOTT PARK, Ill., May 3, 2018 /PRNewswire/ — Abbott (NYSE: ABT) today announced U.S. Food and Drug Administration (FDA) clearance of the Advisor™ HD Grid Mapping Catheter, Sensor Enabled™. Advisor HD Grid employs a new design that allows physicians to see things differently, capturing and analyzing data in a novel manner to create […]