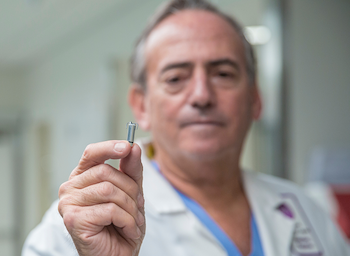
World’s Smallest Pacemaker Can Now Treat Patients with AV Block, Allowing More Potential Candidates for Leadless Pacing DUBLIN, June 15, 2020 (GLOBE NEWSWIRE) — Medtronic plc (NYSE:MDT) today announced it has received CE (Conformité Européenne) Mark for Micra™ AV Transcatheter Pacing System (TPS), the world’s smallest pacemaker with atrioventricular (AV) synchrony. Micra […]
Rhythm
Radcliffe Cardiology Announces New Educational Hub, Arrhythmia Academy
LONDON, June 10, 2020 /PRNewswire/ — An exciting new digital platform – ‘Arrhythmia Academy’ launches today – dedicated to arrhythmia and electrophysiology specialists. This new scientific content hub is led by Editor-in-Chief Prof Angelo Auricchio, MD, PhD, (Lugano, Switzerland) and is supported by a stellar, international editorial board. In association with leading Cardiovascular content provider Radcliffe […]
CorWave Reports Two Critical Technical Milestones Required to Proceed to the Clinical Phase at the American Society for Artificial Internal Organs Annual Meeting
The presentation was selected as Top Bioengineering Abstract and confirms CorWave’s commitment to scientific excellence, which enabled demonstration of its Left Ventricular Assist Device’s (LVAD) capability to provide durable, long-term circulatory support with high-fidelity physiologic flow CLICHY, France–(BUSINESS WIRE)–CorWave announced today that the latest results of its preclinical Left Ventricular […]
Espero BioPharma, Inc. Expands Collaboration Effort for Tecarfarin
JACKSONVILLE, Fla. and IRVINE, Calif., June 4, 2020 /PRNewswire/ — Espero BioPharma, Inc., a pharmaceutical company focused on the development of therapeutics for unmet needs in thrombosis and cardiac rhythm control, today expanded a collaborative partnering effort for its lead product candidate tecarfarin. Tecarfarin is a Phase III-ready, vitamin K antagonist, taken once a day […]
Novadoz Pharmaceuticals FDA Approvals of Oseltamivir and Dofetilide Continues the Company’s Rise in the Generic Market
PISCATAWAY, N.J., June 4, 2020 /PRNewswire/ — Novadoz Pharmaceuticals, the U.S based sales & marketing affiliate for MSN Labs, located in Hyderabad India, recently received FDA approval for their generic versions of Oseltamivir Phosphate Capsules and Dofetilide Capsules. The company was notified earlier this year, that Dofetilide 125mcg, 250mcg, 500mcg capsules, AB rated to […]
Coala launches first real-time Telemedicine solution for virtual diagnostics of heart and lungs
Integrated medical solution that meets new clinical demands of home-based, real-time cardiac and respiratory diagnostics IRVINE, Calif., June 3, 2020 /PRNewswire/ — Coala Life, a Swedish innovator in remote cardiac and respiratory monitoring, has launched the first smartphone powered virtual cardiac and respiratory diagnostics solution with real-time, p-wave based detection of nine common […]
Adagio Medical Receives CE Mark Approval for its iCLAS™ Cryoablation System and Prepares for European Commercial Launch
LAGUNA HILLS, Calif., June 1, 2020 /PRNewswire/ — Adagio Medical, Inc., announces that it has received CE Mark approval for its ultra-low temperature intelligent Continuous Lesion Ablation System (iCLAS) for the endocardial treatment of paroxysmal (PAF) and persistent (PsAF) atrial fibrillation and atrial flutter. “More than half of all atrial fibrillation (AF) patients are diagnosed with PsAF. There […]
ZOLL Introduces New AED With Enhanced Real CPR Help, Integrated Child Rescue, and Wireless Connectivity
CHELMSFORD, Mass.–(BUSINESS WIRE)–ZOLL® Medical Corporation, an Asahi Kasei Group Company that manufactures medical devices and related software solutions, announced today that the ZOLL AED 3® defibrillator received premarket approval (PMA) by the FDA for use by lay rescuers. “ZOLL is committed to supporting the public with all emergency preparedness needs during this […]
Stereotaxis Robotic Technology Now Referenced In Over 400 Scientific Publications
Over 400 scientific publications now document clinical benefits of Stereotaxis robotic technology Most recent publication reviews outcomes of 1,003 consecutive robotic procedures at a single center ST. LOUIS, June 02, 2020 (GLOBE NEWSWIRE) — Stereotaxis (NYSE: STXS), the global leader in innovative robotic technologies for the treatment of cardiac arrhythmias, today announced […]
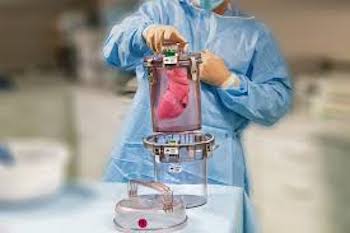
Paragonix Technologies Announces Enrollment Milestone for its Global Registry for Donor Heart Preservation
The Paragonix GUARDIAN Registry has reached its 100th patient enrollment milestone CAMBRIDGE, Mass.–(BUSINESS WIRE)–Paragonix Technologies, Inc. today announced that its clinical global registry, the Global Utilization And Registry Database for Improved Heart Preservation (GUARDIAN), has reached enrollment of over 100 patients that have received a donor heart. Patients that have received donor […]