ST. LOUIS and BARCELONA, Spain, Nov. 01, 2019 (GLOBE NEWSWIRE) — Stereotaxis (NYSE American: STXS) and ADAS 3D Medical SL today announced the first patients have been successfully treated with the integration of ADAS 3D’s advanced preoperative substrate mapping and Stereotaxis’ Robotic Magnetic Navigation technologies. ADAS 3D helps identify possible sources of dangerous irregular heartbeats and […]
Rhythm
Baylis Medical Introduces a 2-French Electrophysiology Catheter into North America
TORONTO, Oct. 31, 2019 /PRNewswire/ – Baylis Medical announced today the first North American use of its EPstar™ Fixed Electrophysiology Catheters. The EPstar catheters make available the smallest diagnostic catheter in the North American electrophysiology market, allowing physicians to reach previously inaccessible areas of the heart for procedure mapping. The EPstar […]
ANI Announces Plans to Launch Bretylium Tosylate Injection, USP 500mg/10ml (50mg/ml) for Ventricular Arrhythmias
BAUDETTE, Minn., Oct. 31, 2019 /PRNewswire/ — ANI Pharmaceuticals, Inc. (“ANI”) (Nasdaq: ANIP) today announced that its partner Pharmaceutics International Inc. (Pii) has received FDA approval of a Prior Approval Supplement for Bretylium Tosylate Injection, USP 500mg/10ml (50mg/ml). ANI plans to launch this currently unavailable drug in December, introducing this critical drug for the treatment of ventricular fibrillation and life-threatening […]
CathVision Receives Competitive EU Grant
COPENHAGEN, Denmark, Oct. 31, 2019 /PRNewswire/ — CathVision ApS, a medical device company developing, manufacturing and selling cardiac electrophysiology products, has been awarded a competitive EU grant of 2.2M Euro for the clinical development of its software and clinical decision support platform. The EU grant is for an SME Instrument, Phase 2 under the Horizon2020 […]
Impulse Dynamics Announces U.S. FDA Supplemental-PMA Approval of its Next Generation Optimizer® Smart System
Commercial-Ready Device to Deliver Improved Outcomes to Heart Failure Patients MOUNT LAUREL, N.J.–(BUSINESS WIRE)–Impulse Dynamics, developer of the implantable Optimizer Smart System for delivering CCM™ (Cardiac Contractility Modulation) therapy, announced today that it has received FDA approval of a PMA supplement for its next generation, two-lead Optimizer Smart System for […]
AliveCor and Huami Partner To Deliver The Next Generation of Medical Wearables
NEW YORK and MOUNTAIN VIEW, Calif., Oct. 30, 2019 /PRNewswire/ — AliveCor, the leader in AI based, personal ECG technology, and provider of enterprise cardiology solutions; and Huami Corporation (NYSE:HMI), a global leader in biometric, data-driven wearables, today announced a multi-faceted global strategic alliance that combines the technologies of each company to deliver a […]
CardioFocus® Treats First Patients With HeartLight® Endoscopic Ablation System In France
MARLBOROUGH, Mass., Oct. 29, 2019 /PRNewswire/ — CardioFocus, Inc., a medical device company dedicated to advancing ablation treatment for atrial fibrillation (AFib), announced today that the first patients in France have been treated in the University Public Hospital of Nancy with the HeartLight Endoscopic Ablation System as part of an exclusive distribution partnership with MicroPort® CRM France. To […]
Medtronic Receives FDA “Breakthrough Device Designation” for Developing Fully Implantable Heart Pump
DUBLIN, Oct. 29, 2019 (GLOBE NEWSWIRE) — Medtronic plc (NYSE:MDT), the global leader in medical technology, today announced it has received Breakthrough Device designation from the U.S. Food and Drug Administration (FDA) for the Medtronic Fully Implantable Left Ventricular Assist Device (LVAD) for patients with advanced heart failure, currently in development. […]
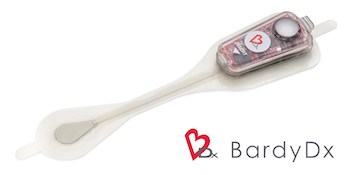
Bardy Diagnostics™ Wins the Remote Monitoring in Arrhythmias Digital Health Pitch Session at European Society of Cardiology Congress 2019 and Presents at UK Heart Rhythm Congress 2019
SEATTLE, Oct. 24, 2019 /PRNewswire/ — Bardy Diagnostics, Inc., (“BardyDx”), a leading provider of ambulatory cardiac monitoring technologies and custom data solutions, announced that it was selected as winner of the “Remote Monitoring in Arrhythmias” Technology and Innovation Pitch Session held as part of the Digital Health program at the European Society […]
Vygon Group Acquires Pilot, Specialist in ECG Central Venous Catheter Tip Location and Navigation Devices
Écouen, France, October 21, 2019 – Vygon Group, a specialist single-use medical devices group, today announces its acquisition of Italian firm Pilot, which specializes in ECG guidance devices. The financial terms of the agreement have not been disclosed. The aim of this transaction is to acquire the ECG location and navigation […]