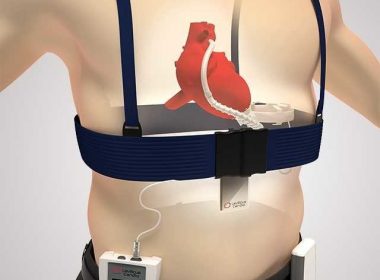
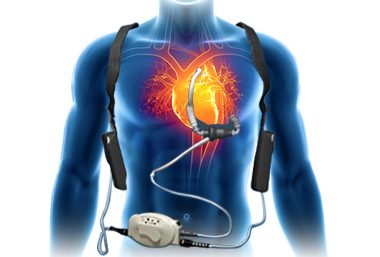
PETAH TIKVA, Israel, April 10, 2018 /PRNewswire/ — Leviticus Cardio, the inventor of wireless power transfer for Left Ventricular Assisted Devices, or “LVADs”, for patients with severe cardiac issues, has announced another round of successful animal trials, well beyond periods required to demonstrate suitability for chronic conditions. The company confirms that the successful studies […]
Rhythm
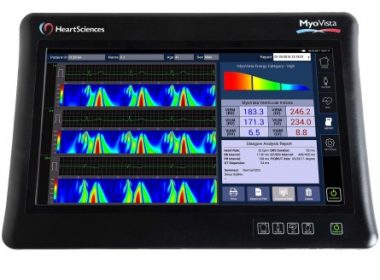
HeartSciences Showcases MyoVista® Wavelet ECG at 2018 China International Medicinal Equipment Fair (CMEF)
SOUTHLAKE, Texas–(BUSINESS WIRE)–HeartSciences, an innovative medical device company, is proud to announce its participation as an exhibitor at the upcoming 2018 China International Medicinal Equipment Fair (CMEF) Shanghai, China, April 11-14, 2018. CMEF has become the largest fair of medical devices, related products and services in the Asia-Pacific region. The […]
Cardiac Monitoring and Cardiac Rhythm Management Devices Market Worth USD 25.12 Billion by 2022
PUNE, India, April 9, 2018 /PRNewswire/ — According to a new market research report “Cardiac Monitoring & Cardiac Rhythm Management Devices Market by Product (ECG, Cardiac Output Monitoring, ILR), CRM [ICDs, External Defibrillator, Pacemaker (Dual Chamber Pacemaker)], End User (Home and Ambulatory, Hospital) – Global Forecast to 2022“, published by MarketsandMarkets™, the market is […]
Fischer Medical Receives U.S. FDA 510(k) Clearance to Market Bloom2 Cardiac Stimulator
DENVER, April 9, 2018 /PRNewswire/ — Fischer Medical has announced that the Bloom2 cardiac electrophysiology stimulator device has received U.S. Food and Drug Administration (FDA) 510(k) clearance. The Bloom2stimulator is a fundamental component of nearly all electrophysiology (EP) procedures to diagnose and treat cardiac rhythm disorders. The release of the digital version of […]
LivaNova Completes Acquisition of TandemLife
LONDON–(BUSINESS WIRE)–LivaNova PLC (NASDAQ:LIVN) (“LivaNova” or the “Company”), a market-leading medical technology company, today announced it closed its acquisition of TandemLife. Previously, on Feb. 14, LivaNova announced its intent to acquire TandemLife, a privately held company focused on advanced cardiopulmonary temporary support solutions. “The acquisition of TandemLife allows us to […]
Sotera Medical Secures Seed Financing
CLEVELAND–(BUSINESS WIRE)–Sotera Medical Corp., a medical device company developing a novel device for use in cardiac procedures to treat atrial fibrillation, has closed initial seed funding in excess of one million dollars. The round was led by a syndicate of investors, including JumpStart Inc., North Coast Angel Fund, Northwest Ohio […]
AliveCor Data Yield Reaches 25MM ECGs
MOUNTAIN VIEW, Calif., April 5, 2018 /PRNewswire/ — AliveCor, the leader in FDA-cleared personal electrocardiogram (ECG) technology, announced today that it had surpassed the milestone of 25 million recorded ECGs – by far the largest data set ever collected by any consumer ECG. The company also announced that its KardiaBand product, paired with […]
Improving Cardiology Care: BIOTRONIK US to Distribute Aziyo ECM Envelopes
LAKE OSWEGO, Ore. and SILVER SPRING, Md., April 3, 2018 /PRNewswire/ — BIOTRONIK US and Aziyo today announced a strategic agreement allowing BIOTRONIK to distribute Aziyo’s CanGaroo® extracellular matrix (ECM) cardiovascular implantable electronic device (CIED) envelopes in the United States. BioEnvelope will be available from BIOTRONIK starting in April 2018. BioEnvelope reduces scar formation and has been shown to mitigate the […]
CorWave is Set to Receive about $3.5 Million in Financing Through France’s Worldwide Innovation Challenge for Its NovaPulse Program, a Mini-Invasive Solution for Cardiac Support
CLICHY, France–(BUSINESS WIRE) CorWave, a French medical technology company that develops innovative implanted cardiac support devices for patients suffering from heart failure, announced today that it has been awarded almost $3.5 million in financing for its R&D program NovaPulse in the Silver Economy category. This is part of phase 2 of […]
Bardy Diagnostics™ Names Ken Nelson Chief Commercial Officer
SEATTLE, April 2, 2018 /PRNewswire/ — Bardy Diagnostics, Inc., (“BardyDx”), a leading provider of ambulatory cardiac monitoring technologies and custom data solutions, including the Carnation Ambulatory Monitor (“CAM™”), the world’s smallest and lightest P-wave centric™ ambulatory cardiac monitor and arrhythmia detection device, announced today the appointment of Kenneth W. Nelson III as Chief Commercial Officer […]