An article published by the Heart Rhythm Society’s journal Heart Rhythm O2 concludes that Vektor’s Computational ECG Mapping System (vMap™) and protocol-based respiratory gating may help facilitate radioablation planning and maintain efficacy during therapy. CARLSBAD, Calif.–(BUSINESS WIRE)–Vektor Medical, Inc., a private company founded to develop the next-generation in arrhythmia mapping systems, today […]
Rhythm
Resverlogix Announces Apabetalone Meets Primary Endpoint in a Pulmonary Arterial Hypertension Pilot Study
Apabetalone, a First-in-Class Epigenetic Therapeutic Candidate, Met Key Endpoint of Lowering Pulmonary Vascular Resistance (PVR) and Was Well Tolerated Successful Completion and Promising Results of Investigator-led APPROACH-p Trial Sets the Stage for a Larger Multicenter Trial CALGARY, Alberta, Sept. 08, 2021 (GLOBE NEWSWIRE) — Resverlogix Corp. (“Resverlogix” or the “Company”) […]
IDTechEx Dissects the Market for Solid-state Batteries in New Report
BOSTON, Sept. 2, 2021 /PRNewswire/ — IDTechEx has recently launched the new version of their market research report on solid-state batteries – “Solid-State and Polymer Batteries 2021-2031: Technology, Forecasts, Players“. This report identifies key players within the market; discusses manufacturing challenges and how companies are addressing limitations; explores lithium metal as […]
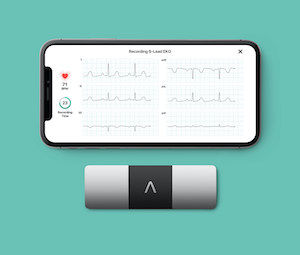
KardiaMobile 6L is Superior to One Lead Personal Devices
MOUNTAIN VIEW, Calif., Sept. 1, 2021 /PRNewswire/ — AliveCor, a leading innovator in FDA-cleared personal electrocardiogram (ECG) technology and services, today announced the KardiaMobile® 6L device was found to be superior to single lead devices according to results from the largest study to assess the reliability of the detection of atrial fibrillation (AF) through […]
BioSig Confirmed as a Participant of The Mark E. Josephson Twenty-Eight Annual State-of-the-Art Arrhythmia Virtual Symposium
Physician-only event, organized by the University of Pennsylvania, is a tribute to the pioneer of cardiac electrophysiology Westport, CT, Aug. 31, 2021 (GLOBE NEWSWIRE) — BioSig Technologies, Inc. (Nasdaq: BSGM) (“BioSig” or the “Company”), a medical technology company commercializing an innovative signal processing platform designed to improve signal fidelity and […]
Late-Breaking Data Shows Abbott’s Amplatzer™ Amulet™ Occluder Offers Superior Left Atrial Appendage Closure Compared to Watchman‡ Device for People With Atrial Fibrillation at Risk of Stroke
– New data presented at ESC Congress 2021 and simultaneously published in Circulation show Amulet Occluder was superior in left atrial appendage (LAA) closure and noninferior on safety and effectiveness endpoints compared to Watchman device – Amulet device is the first and only minimally invasive option approved for LAA occlusion […]
Stereotaxis and MicroPort EP Collaborate to Advance Innovation and Adoption of Robotic Electrophysiology in China
ST. LOUIS and SHANGHAI, China, Aug. 30, 2021 (GLOBE NEWSWIRE) — Stereotaxis (NYSE: STXS) and Shanghai Microport EP Medtech Co., Ltd. (“MicroPort EP”) today announced a broad collaboration to advance technology innovation and commercial adoption of robotics in electrophysiology in China. The agreement brings together MicroPort EP’s commercial and product leadership in […]
Real-World Data Demonstrate Significant Reduction in Complications and Reinterventions with Medtronic Micra Leadless Pacemaker
Results Presented at ESC 2021 Show Micra TPS Compares Favorably to Traditional Pacemakers DUBLIN, Aug. 27, 2021 /PRNewswire/ — Medtronic plc (NYSE:MDT), the global leader in medical technology, today announced new data from the Micra Coverage with Evidence Development (CED) Study, the largest evaluation of leadless pacemakers to date, which showed the Micra™ Transcatheter Pacing […]
Landmark trial demonstrates Jardiance® (empagliflozin) is the first therapy to show statistically significant improvement in heart failure outcomes in adults with preserved ejection fraction
– In this clinical first for adults with heart failure with preserved ejection fraction, Jardiance demonstrated an impressive 21% relative risk reduction in the composite primary endpoint of cardiovascular death or hospitalization for heart failure – The benefit in the primary endpoint was independent of ejection fraction or diabetes status […]
Veepoo technology releases the health smartwatch: Watch Rig, it can accurately monitor EKG/ECG, SPo2, heart rate and other health information
NEW YORK, Aug. 27, 2021 /PRNewswire/ — VEEPOO WATCH RIG, equipped with multiple health monitoring functions featuring its EKG/ECG monitoring service, can effectively detect a series of heart diseases such as atrial fibrillation and premature beat. The sampling of EKG/ECG signal frequency can reach up to 512hz, which is a medical application level. […]