CAMBRIDGE, Mass. & BEIJING–(BUSINESS WIRE)–BeiGene (NASDAQ: BGNE; HKEX: 06160) announced positive results from a planned interim analysis of the Phase 3 ALPINE trial comparing BRUKINSA® (zanubrutinib) against ibrutinib in adults with relapsed or refractory (R/R) chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma (SLL). BRUKINSA met the primary endpoint of the […]
Rhythm
BioSig’s PURE EP(tm) System Evaluated with Patients at Mayo Clinic in Arizona
Westport, CT, April 27, 2021 (GLOBE NEWSWIRE) — The clinic is conducting an evaluation of the PURE EP(tm) System, a signal processing technology for cardiac arrhythmia treatments New installation follows Mayo Clinic in Florida that has been conducting patient cases with the technology since January 2020 BioSig Technologies, Inc. (NASDAQ: […]
New two-years follow-up data from ETNA-AF-Europe reaffirms benefit of LIXIANA® (edoxaban) in people with atrial fibrillation
Large routine clinical practice study, ETNA-AF-Europe, demonstrates continued benefits of edoxaban across age groups and supports treatment in elderly patient populations, in line with those observed in ETNA-AF after one year and in ENGAGE AF-TIMI 48 ETNA-AF-Europe is part of the global ETNA-AF programme, which is the largest prospective, non-interventional […]
Implicity Earns Coveted Spot in the Plug and Play Cleveland Health Accelerator Program
Remote patient monitoring and cardiac data management solution provider is one of only six innovators accepted into the Batch 5 program for Spring 2021 BOSTON, April 15, 2021 /PRNewswire/ — Implicity, a leader in remote patient monitoring and cardiac data management solutions, today announced the company has been selected to join Plug and Play […]
BioSig Launches Enhanced Signal Processing Capabilities for Advanced Cardiac Signal Analysis Through its Electrophysiology Platform
Westport, CT, April 14, 2021 (GLOBE NEWSWIRE) — The latest release represents the most advanced software version of the PURE EPTM System, signal processing technology for treatments of irregular heartbeats BioSig Technologies, Inc. (NASDAQ: BSGM) (“BioSig” or the “Company”), a medical technology company commercializing an innovative signal processing platform designed to improve […]
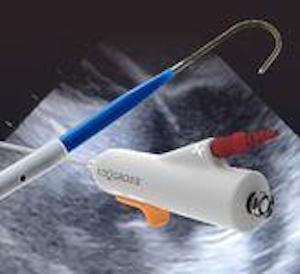
Acutus Medical Announces FDA Clearance of AcQCross™, a Full Suite of Universal Transseptal Crossing Devices
Product Family Targets Over 300,000 Annual US Electrophysiology and Structural Heart Procedures CARLSBAD, Calif., April 13, 2021 (GLOBE NEWSWIRE) — Acutus Medical, Inc. (“Acutus”) (Nasdaq: AFIB), an arrhythmia management company focused on improving the way cardiac arrhythmias are diagnosed and treated, today announced the FDA clearance of the AcQCross™ family […]
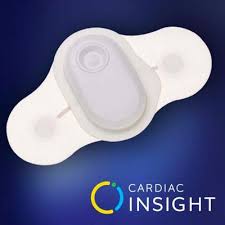
Cardiac Insight Remains Committed to Serving Seniors and Medically Underserved in ECG Wearable Market
Continued focus on providing nationwide access to patients and physicians to innovative wearable ECG technologies despite updated Novitas and First Coast Reimbursement Rates and Competitive Market Exit SEATTLE–(BUSINESS WIRE)–Cardiac Insight, Inc., a healthcare innovator specializing in wearable cardiac sensors and proven automated electrocardiogram (ECG) analysis, remains committed to delivering highly […]
Cardialen Announces Positive Results from Low-Voltage MultiPulse Therapy to Terminate Atrial Fibrillation
MultiPulse Therapy™ (MPT™) achieved 71% termination of acute, induced atrial fibrillation (AF) episodes at energy levels deemed to be tolerable for patients1,2 MINNEAPOLIS–(BUSINESS WIRE)–Cardialen, Inc., has published a first-in-human study in the Journal of the American College of Cardiology: Clinical Electrophysiology titled Novel Low-Voltage MultiPulse Therapy to Terminate Atrial Fibrillation. The study […]
EP Lab Digest Features Physician Experience with BioSig’s Cardiac Signal Acquisition Technology
Westport, CT, April 08, 2021 (GLOBE NEWSWIRE) — Leading industry publication highlights the rising importance of intracardiac signals in complex cardiac ablations that treat irregular heart rhythm disorders High-quality intracardiac signals are deemed essential to determine ablation endpoints PURE EP™’s signals are described as ‘indispensable’ by the physician user BioSig […]
Broward Health Medical Center Becomes First Hospital in Florida to Adopt Advanced Genesis Robotic Technology to Treat Heart Rhythm Disorders
ST. LOUIS, April 07, 2021 (GLOBE NEWSWIRE) — Stereotaxis (NYSE: STXS), the global leader in innovative robotic technologies for the treatment of cardiac arrhythmias, announces that Broward Health Medical Center is establishing a robotic electrophysiology program to expand access to advanced minimally-invasive treatment of heart rhythm disorders in Florida. Broward Health Medical […]