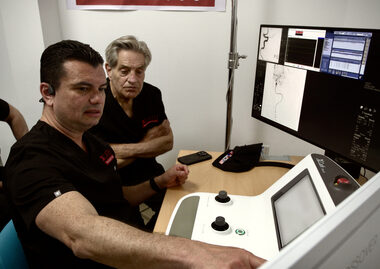
HOUSTON & PANAMA CITY, Panama–(BUSINESS WIRE)–XCath announced successful completion of world’s first telerobotic mechanical thrombectomy for stroke using the XCath Iris Surgical Robotic System.
Uncategorized
AngioDynamics to Report Fiscal 2026 Third Quarter Results on April 2, 2026
LATHAM, N.Y.–(BUSINESS WIRE)–AngioDynamics, Inc. (NASDAQ: ANGO), a leading and transformative medical technology company focused on restoring healthy blood flow in the body’s vascular system, expanding cancer treatment options and improving patient quality of life, today announced that it will report financial results for the third quarter of fiscal year 2026 before the market open on Thursday, April 2, 2026. The Company’s management will host a conference call at 8:00 am ET the same day to d
Heartflow Reports Fourth Quarter and Full Year 2025 Financial Results
MOUNTAIN VIEW, Calif., March 18, 2026 (GLOBE NEWSWIRE) — Heartflow, Inc. (Heartflow) (Nasdaq: HTFL), the leader in AI technology for coronary artery disease (CAD), today reported financial results for the fourth quarter and full year ended December 31, 2025. Fourth Quarter 2025 Highlights Total revenue of $49.1 million, a 40% increase year-over-yearGross margin of 79.5%, non-GAAP gross margin of 79.9%Net operating loss of $17.8 million, non-GAAP net operating loss of $12.5 millionU.S. installed base of 1,465 accounts as of December 31, 2025U.S. Plaque installed base of 489 accounts as of December 31, 2025Aetna began coverage of Heartflow Plaque Analysis, bringing total U.S. covered lives for Plaque to approximately 75% 2026 Annual Guidance Total revenue of $218 million to $222 million (approximately 24% to 26% growth year-over-year)Non-GAAP gross margin of 80% to 81% “Our strong fourth quarter performance concluded a record year for Heartflow,” said John Farquhar, President and CEO of Heartflow. “The accelerating adoption of the Heartflow Platform, combined with our disciplined execution across commercial, innovation, and clinical initiatives, drove 40% fourth quarter and full year revenue growth and record gross margins. We also made significant strides in scaling account activations and driving early physician adoption of Heartflow Plaque Analysis. Our 2026 guidance reflects strong business fundamentals, a solid foundation for growth, and high confidence in consistent execution. With commercial, innovation and clinical catalysts on the horizon, our conviction in the business has never been higher.” Fourth Quarter 2025 Financial ResultsTotal revenue was $49.1 million, a 40% increase year-over-year. U.S. revenue was $44.8 million, a 41% increase year-over-year. International and other revenue was $4.3 million, a 35% increase year-over-year. The year-over-year increase in total global revenue was primarily attributable to an increase in total U.S. FFRCT volume. Gross profit was $39.1 million, compared to $26.3 million in the prior year period. Non-GAAP gross profit was $39.2 million, compared to $26.3 million in the prior year period. Gross margin was 79.5%, compared to 75.0% in the prior year period. Non-GAAP gross margin was 79.9%, compared to 75.3% in the prior year period. The year-over-year gross margin expansion was primarily attributable to an increase in revenue case volume and improved production team productivity driven by AI efficiency initiatives, partially offset by the hiring and training of production team personnel. Total operating expenses were $56.8 million, or 116% of total revenue, compared to $42.3 million, or 121% of total revenue, in the prior year period. Non-GAAP total operating expenses were $51.7 million, or 105% of total revenue, compared to $39.9 million, or 114% of total revenue, in the prior year period. The year-over-year operating expense increase was primarily attributable to increased investment in sales personnel and related expenses, as well as increased investments in technology and clinical research. Net operating loss was $17.8 million, compared to $16.1 million in the prior year period. Non-GAAP net operating loss was $12.5 million, compared to $13.5 million in the prior year period. Net loss was $24.4 million, or ($0.29) net loss per share, compared to $33.0 million, or ($5.59) net loss per share, in the prior year period. Net loss for the fourth quarters of 2025 and 2024 included a noncash charge of $9.3 million and $11.9 million, respectively, resulting from the remeasurement of the fair value of the Company’s common stock warrant liability. As of October 22, 2025, the warrant holder net exercised all warrants in full. Therefore, the fourth quarter of 2025 is the last quarter that movements in the Company’s stock price will trigger a warrant revaluation and result in a noncash charge to net loss. Non-GAAP net loss was $9.8 million, or ($0.12) non-GAAP net loss per share, compared to $18.6 million, or ($3.15) non-GAAP net loss per share, in the prior year period. Adjusted EBITDA was ($11.1) million, compared to ($12.0) million in the prior year period. Full Year 2025 Financial ResultsTotal revenue was $176.0 million, a 40% increase year-over-year. U.S. revenue was $160.6 million, a 41% increase year-over-year. International and other revenue was $15.4 million, a 26% increase year-over-year. The year-over-year increase in total global revenue was primarily attributable to an increase in total U.S. FFRCT volume. Gross profit was $135.2 million, compared to $94.4 million in the prior year period. Non-GAAP gross profit was $135.6 million, compared to $94.8 million in the prior year period. Gross margin was 76.8%, compared to 75.1% in the prior year period. Non-GAAP gross margin was 77.0%, compared to 75.3% in the prior year period. The year-over-year gross margin expansion was primarily attributable to an increase in revenue case volume and improved production team productivity driven by AI efficiency initiatives, partially offset by the hiring and training of production team personnel. Total operating expenses were $199.3 million, or 113% of total revenue, compared to $155.7 million, or 124% of total revenue, in the prior year period. Non-GAAP total operating expenses were $185.7 million, or 105% of total revenue, compared to $145.8 million, or 116% of total revenue, in the prior year period. The year-over-year operating expense increase was primarily attributable to increased investment in personnel and related expenses, as well as increased investments in technology and clinical research. Net operating loss was $64.1 million, compared to $61.2 million in the prior year period. Non-GAAP net operating loss was $50.1 million, compared to $51.0 million in the prior year period. Net loss was $116.8 million, or ($3.17) net loss per share, compared to $96.4 million, or ($17.98) net loss per share, in the prior year period. Non-GAAP net loss was $59.9 million, or ($1.62) non-GAAP net loss per share, compared to $69.6 million, or ($12.98) non-GAAP net loss per share, in the prior year period. Adjusted EBITDA was ($44.7) million, compared to ($45.7) million in the prior year period. Cash, cash equivalents and investments totaled $280.2 million as of December 31, 2025. For additional information regarding non-GAAP financial measures, see “Use of Non-GAAP Measures,” “Heartflow GAAP to Non-GAAP Reconciliations” and “Reconciliation of GAAP Net Loss to Adjusted EBITDA” below. Webcast and Conference Call DetailsHeartflow will host a conference call today, March 18, 2026, at 1:30 p.m. PT / 4:30 p.m. ET to discuss its fourth quarter and full year 2025 financial results. Those interested in listening to the conference call should register online using this link. Once registered, participants will receive dial-in numbers and a unique PIN to join the call. Participants are encouraged to register more than 15 minutes prior to the start of the call. A live and archived webcast of the event will also be available on the “Investor Relations” section of the Heartflow website at https://ir.heartflow.com. The archived version will be available for 12 months following completion of the live call. About Heartflow’s Technology and ResearchHeartflow’s technology is redefining precision cardiovascular care through clinically-proven AI and the world’s largest coronary imaging dataset. Heartflow has been adopted by more than 1,800 institutions globally and continues to strengthen its commercial presence to make this cutting-edge solution more widely available to an increasingly diverse patient population. Backed by American College of Cardiology and American Heart Association (ACC/AHA) guidelines and supported by more than 600 peer-reviewed publications, Heartflow has redefined how clinicians manage care for over 600,000 patients worldwide.1 Key benefits include: Proprietary data pipeline: Built from more than 160 million annotated CTA images, Heartflow’s data foundation powers advanced AI models that deliver highly accurate, reproducible insights across diverse patient populations.Extensive clinical and real-world validation: Heartflow’s AI-driven solutions have been validated through clinical evidence in over 200 studies assessing over 365,000 patients. Proven in real-world practice with reproducibility and accuracy, Heartflow’s coronary CTA image acceptance rates exceed 97%.Seamless clinical integration via upgraded workflow: Heartflow delivers final quality-reviewed analyses instantly upon order, enabling clinicians to move from diagnosis to decision without delay.Quality system, global security and patient-data integrity compliance: Heartflow meets or exceeds leading international standards, including HITRUST, SOC 2 Type 2, ISO 13485, and ISO 27001. About Heartflow, Inc.Heartflow is transforming coronary artery disease from the world’s leading cause of death into a condition that can be detected early, diagnosed accurately, and managed for life. The Heartflow One platform uses AI to turn coronary CTA images into personalized 3D models of the heart, providing clinically meaningful, actionable insights into plaque location, volume, and composition and its effect on blood flow — all without invasive procedures. Discover how we’re shaping the future of cardiovascular care at heartflow.com. Use of Non-GAAP MeasuresTo supplement its consolidated financial statements prepared in accordance with U.S. generally accepted accounting principles (GAAP), the Company discloses non-GAAP gross profit and non-GAAP gross margin, non-GAAP total operating expenses, non-GAAP research and development expense, non-GAAP selling, general and administrative expense, non-GAAP net operating loss, non-GAAP net loss, non-GAAP net loss per share, basic and diluted, and Adjusted EBITDA (collectively, the “Non-GAAP Measures”) in this press release. As used by the Company, these measures are adjusted to exclude stock-based compensation expense from the comparable GAAP financial measure. Non-GAAP net loss and non-GAAP net loss per share, basic and diluted, are also adjusted for change in fair value of common stock warrant liability, change in fair value of derivative liability and loss on extinguishment of debt. In addition, Adjusted EBITDA is calculated by adding back to net loss or excluding, as appropriate, interest income and expense, provision for income taxes, and charges for depreciation and amortization and is further adjusted by adding back in or excluding, stock-based compensation and, as appropriate, other income and expense items that are not reflective of the Company’s underlying continuing operating performance. Reconciliations of the Non-GAAP Measures to their most directly comparable GAAP financial measures are provided in the financial statement tables included at the end of this press release, and investors are encouraged to review the reconciliations. The Company believes the presentation of the Non-GAAP Measures, when shown in conjunction with the corresponding GAAP measures, provides useful information to investors as it provides visibility to the Company’s underlying continuing operating performance from period to period by excluding the impact of stock-based compensation and certain other items that are not reflective of the Company’s ongoing operations. Because of the variety of equity awards used by companies, the varying methodologies for determining stock-based compensation expense, the subjective assumptions used in those determinations, and the volatility in valuations that can be driven by market conditions outside the Company’s control, we believe excluding stock-based compensation expense enhances the ability of management and investors to understand and assess the underlying performance of our business over time and compare it against our peers, a majority of whom also exclude stock-based compensation expense from their non-GAAP results. With respect to the presentation of Adjusted EBITDA, the Company believes it is a useful measure to evaluate the Company’s operating performance and it is used by the Company to evaluate ongoing operations and for planning and forecasting purposes. Adjusted EBITDA is also a measure frequently used by analysts, investors and other interested parties to evaluate companies in our same industry. The Company’s definition of the Non-GAAP Measures may differ from similarly titled measures used by others. The Non-GAAP Measures should be considered only as a supplement to, and not as a substitute for, or superior to, their most directly comparable GAAP financial measures. Because the Non-GAAP Measures exclude the effect of items that increase or decrease the Company’s reported results of operations, management strongly encourages investors to review the reconciliations to the most comparable GAAP financial measures at the end of this press release and, when they become available, the Company’s consolidated financial statements and publicly filed Securities and Exchange Commission (“SEC”) reports in their entirety. The Company is not able to provide a reconciliation without unreasonable efforts of its forward-looking guidance related to non-GAAP gross margin to the most directly comparable GAAP financial measure due to the unknown effect of stock-based compensation that is material to the comparable GAAP financial measure. Forward-Looking StatementsThis press release contains express or implied forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. All statements other than statements of historical facts contained in this press release, including statements regarding our strategy, expected market growth and financial guidance, are forward-looking statements. These forward-looking statements are based on management’s current expectations and are subject to risks and uncertainties that may cause actual results to differ materially from those expressed or implied in the forward-looking statements, including, but not limited to: we may not be able to achieve or sustain profitability; our dependence on the success of our one product, Heartflow FFRCT Analysis; healthcare providers may be unwilling to change their standard practice regarding the evaluation of coronary artery disease; adoption of the Heartflow Platform by healthcare providers may be negatively impacted if third-party payors, including government payors, do not cover or provide adequate reimbursement; the concentration of our customer base; the significant competition we face in an environment of rapid technological change; the commercialization of Heartflow Plaque Analysis is nascent; risks associated with our use and development of AI models; risks related to failing to properly manage our future growth; disruption by catastrophic events; risks associated with our dependence on our information technology systems; security breaches that we cannot anticipate or successfully defend; extensive regulatory requirements we face to bring our products to market; and third parties could develop and commercial technology and products similar or identical to ours. For a more extensive description of these and other risks and uncertainties that could materially affect our results, you should read our filings with the SEC, including our Annual Report on Form 10-K and Quarterly Reports on Form 10-Q, as such filings may be amended, supplemented or superseded from time to time by other reports Heartflow files with the SEC. You should not place undue reliance on the forward-looking statements in this press release, which speak only as of the date hereof, and we undertake no obligation to update the forward-looking statements to reflect events or circumstances after the date of this press release or to reflect new information or the occurrence of unanticipated events, except as required by law. Investor ContactNick Laudiconlaudico@heartflow.com Media ContactElliot Levyelevy@heartflow.com Heartflow, Inc.Consolidated Statements of Operations(unaudited, in thousands, except share and per share data) Three Months Ended Year Ended December 31, December 31, 2025 2024 2025 2024 Revenue $49,130 $34,977 $176,034 $125,808 Cost of revenue 10,067 8,727 40,837 31,359 Gross profit 39,063 26,250 135,197 94,449 Operating Expenses: Research and development 18,665 12,279 64,918 43,517 Selling, general and administrative 38,148 30,029 134,345 112,154 Total operating expenses 56,813 42,308 199,263 155,671 Loss from operations (17,750) (16,058) (64,066) (61,222)Interest income 2,635 592 5,538 4,066 Interest expense (8) (5,152) (15,173) (22,768)Change in fair value of common stock warrant liability (9,308) (11,905) (43,894) (16,395)Change in fair value of derivative liability – – 7,311 (222)Loss on extinguishment of debt – – (6,360) – Other income (expense), net (129) (447) (223) 168 Loss before provision for income taxes (24,560) (32,970) (116,867) (96,373)(Provision for) benefit from income taxes 165 (5) 76 (53)Net loss $(24,395) $(32,975) $(116,791) $(96,426)Comprehensive loss: Net loss $(24,395) $(32,975) $(116,791) $(96,426)Other comprehensive income (loss): Foreign currency translation gain (loss) (69) 233 191 (271)Unrealized gain on investments, net 156 – 156 – Total other comprehensive income (loss) 87 233 347 (271)Total comprehensive loss $(24,308) $(32,742) $(116,444) $(96,697) Net loss per share, basic and diluted $(0.29) $(5.59) $(3.17) $(17.98)Weighted-average shares used to compute net loss per share, basic and diluted 84,828,694 5,894,840 36,853,867 5,363,435 Heartflow, Inc.Consolidated Balance Sheets(unaudited, in thousands) December 31, 2025 2024Assets Current assets Cash and cash equivalents $44,776 $51,367 Short-term investments 132,010 – Accounts receivable, net 29,343 24,639 Restricted cash, current – 150 Prepaid expenses and other current assets 14,075 6,132 Total current assets 220,204 82,288 Long-term investments 103,365 – Property and equipment, net 8,587 8,920 Operating lease right-of-use assets 17,488 18,805 Restricted cash, non-current 4,709 4,325 Other non-current assets 5,099 4,366 Total assets $359,452 $118,704 Liabilities, redeemable convertible preferred stock and stockholders’ equity (deficit) Current liabilities Accounts payable $3,169 $2,870 Accrued expenses and other current liabilities 33,279 25,319 Operating lease liabilities, current portion 5,922 5,416 Total current liabilities 42,370 33,605 Term loan – 136,431 Common stock warrant liability – 20,835 Operating lease liabilities, non-current portion 16,132 18,537 Other non-current liabilities 303 214 Total liabilities 58,805 209,622 Redeemable convertible preferred stock issuable in series, $0.001 par value – 768,566 Stockholders’ equity (deficit) Preferred stock, $0.001 par value – – Common stock, $0.001 par value 85 6 Additional paid-in capital 1,388,737 112,241 Accumulated other comprehensive income (425) (772)Accumulated deficit (1,087,750) (970,959)Total stockholders’ equity (deficit) 300,647 (859,484) Total liabilities, redeemable convertible preferred stock and stockholders’ equity (deficit)$359,452 $118,704 Heartflow, Inc.GAAP to Non-GAAP Reconciliations(unaudited, in thousands except per share amounts and percentage data) Three Months Ended December 31, 2025 Three Months Ended December 31, 2024 GAAP Adjustments Non-GAAP GAAP Adjustments Non-GAAP Gross profit $39,063 $184 (a)$39,247 $26,250 $76 (a)$26,326 Gross margin 79.5% 0.4% 79.9% 75.0% 0.2% 75.3% Operating Expenses: Research and development $18,665 $(1,547)(a)$17,118 $12,279 $(585)(a)$11,694 Selling, general and administrative $38,148 $(3,529)(a)$34,619 $30,029 $(1,853)(a)$28,176 Total operating expenses $56,813 $(5,076) $51,737 $42,308 $(2,438) $39,870 Loss from operations $(17,750) $5,260 $(12,490) $(16,058) $2,514 $(13,544) Net loss $(24,395) $14,568 (b)$(9,827) $(32,975) $14,419 (c)$(18,556)Net loss per share, basic and diluted $(0.29) $0.17 $(0.12) $(5.59) $2.44 $(3.15) (a) Represents adjustments related to stock-based compensation expense(b) Represents adjustments for: (i) stock-based compensation expense of $5.3 million; and (ii) change in fair value of common stock warrant liability of $9.3 million(c) Represents adjustments for: (i) stock-based compensation expense of $2.5 million; and (ii) change in fair value of common stock warrant liability of $11.9 million Heartflow, Inc.GAAP to Non-GAAP Reconciliations(unaudited, in thousands except per share amounts and percentage data) Year Ended December 31, 2025 Year Ended December 31, 2024 GAAP Adjustments Non-GAAP GAAP Adjustments Non-GAAP Gross profit $135,197 $413 (a)$135,610 $94,449 $307 (a)$94,756 Gross margin 76.8% 0.2% 77.0% 75.1% 0.2% 75.3% Operating Expenses: Research and development $64,918 $(3,434)(a)$61,484 $43,517 $(2,151)(a)$41,366 Selling, general and administrative $134,345 $(10,118)(a)$124,227 $112,154 $(7,755)(a)$104,399 Total operating expenses $199,263 $(13,552) $185,711 $155,671 $(9,906) $145,765 Loss from operations $(64,066) $13,965 $(50,101) $(61,222) $10,213 $(51,009) Net loss $(116,791) $56,908 (b)$(59,883) $(96,426) $26,830 (c)$(69,596)Net loss per share, basic and diluted $(3.17) $1.55 $(1.62) $(17.98) $5.00 $(12.98) (a) Represents adjustments related to stock-based compensation expense(b) Represents adjustments for: (i) stock-based compensation expense of $14.0 million; (ii) change in fair value of common stock warrant liability of $43.9 million; (iii) change in fair value of derivative liability of $7.3 million; and (iv) loss on extinguishment of debt of $6.4 million(c) Represents adjustments for: (i) stock-based compensation expense of $10.2 million; (ii) change in fair value of common stock warrant liability of $16.4 million; and (iii) change in fair value of derivative liability of $0.2 million Heartflow, Inc.Reconciliation of GAAP Net Loss to Adjusted EBITDA(unaudited, in thousands) Three Months Ended Year Ended December 31, December 31, 2025 2024 2025 2024 GAAP net loss $(24,395) $(32,975) $(116,791) $(96,426)Non-GAAP adjustments: Interest (income) expense, net (2,627) 4,560 9,635 18,702 Change in fair value of common stock warrant liability 9,308 11,905 43,894 16,395 Change in fair value of derivative liability – – (7,311) 222 Loss on extinguishment of debt – – 6,360 – Other (income) expense, net 129 447 223 (168)Provision for (benefit from) income taxes (165) 5 (76) 53 Depreciation and amortization 1,371 1,591 5,440 5,358 Stock-based compensation expense 5,260 2,514 13,965 10,213 Adjusted EBITDA $(11,119) $(11,953) $(44,661) $(45,651) 1 Gulati, et al. 2021 AHA/ACC/ASE/CHEST/SAEM/SCCT/SCMR Guideline for the Evaluation & Diagnosis of Chest Pain. J Am Coll Cardiol.
HEARTio Announces Closing of $4.25 Million Financing for its AI Algorithm to Detect Warning Signs of Heart Attacks
Pittsburgh, PA — March 16, 2025— Heart Input Output Inc. (“HEARTio”), an artificial intelligence healthcare diagnostics company, today announced the closing of $4.25 Million in new seed financing with participation from Intelligence Ventures, Audacious Capital, VU Venture Partners, LifeX (Pittsburgh), Bessel, and others. These funds will enable the launch of […]
Field Medical Names Mark A. Turco, MD, as Chief Executive Officer
CARDIFF-BY-THE-SEA, Calif., March 16, 2026 /PRNewswire/ — Field Medical, Inc., a clinical-stage medical technology company advancing second-generation pulsed field ablation (PFA) solutions for complex ventricular and atrial arrhythmias, today announced the appointment of Mark A. Turco,…
Lohman Technologies Appoints Two Industry Leaders to Drive AI-Enabled Remote Cardiac Innovation
Board expansion accelerates commercialization pipeline of explainable AI and scalable RPM partnerships. MILWAUKEE, Feb. 26, 2026 /PRNewswire/ — Lohman Technologies announced today that Dr. Daniel DeBehnke and Peter E. Aftosmes have joined its Board of Directors as the company accelerates…
Medtronic reports strong third quarter fiscal 2026 results with highest enterprise revenue growth in 10 quarters
Cardiovascular portfolio up 11% year-over-year; Cardiac Ablation Solutions grew 80% on strength of pulsed field ablation portfolio GALWAY, Ireland, Feb. 17, 2026 /PRNewswire/ — Medtronic plc (NYSE: MDT), a global leader in healthcare technology, today announced financial results for its…
BioCardia Announces Late Breaking Echocardiography Results from the CardiAMP HF Trial to be Presented at Technology and Heart Failure Therapeutics (THT)
SUNNYVALE, Calif., Feb. 03, 2026 (GLOBE NEWSWIRE) — BioCardia®, Inc. [NASDAQ: BCDA], a developer of cellular and cell-derived therapeutics for the treatment of cardiovascular and pulmonary diseases, today announced echocardiography data for the CardiAMP Cell Therapy for the treatment of heart failure has been accepted for Late Breaking Clinical Trial Oral Presentation at the Technology and Heart Failure Therapeutics (THT) Meeting, which takes place March 2-4 in Boston, Massachusetts. On behalf of the CardiAMP HF investigators, the presentation will be made by Dr. Amish Raval, M.D., Professor of Medicine at UW School of Medicine and Public Health and CardiAMP HF Trial National Co-Principal Investigator. Presentation Title: “Autologous Cell Therapy May Curb Pathological Ventricular Remodeling in Chronic Ischemic HFrEF Patients Selected for Favorable Cell Characteristics – Late Breaking Echocardiography Results from the CardiAMP HF trial.” Presentation Session: THT Late Breaking Clinical Trial Oral Presentation on March 2 at 2pm EST. About CardiAMP Autologous Cell Therapy Granted FDA Breakthrough designation, CardiAMP Cell Therapy uses a patient’s own bone marrow cells delivered to the heart in a minimally invasive, catheter-based procedure intended to increase capillary density and reduce tissue fibrosis of myocardial tissue to address microvascular dysfunction. Clinical development of the CardiAMP Cell Therapy for heart failure is supported by the Maryland Stem Cell Research Fund and is reimbursed by Centers for Medicare and Medicaid Services (CMS). CAUTION – Limited by United States law to investigational use. About BioCardia® BioCardia, Inc., headquartered in Sunnyvale, California, is a global leader in cellular and cell-derived therapeutics for the treatment of cardiovascular and pulmonary disease. CardiAMP® autologous and CardiALLO™ allogeneic cell therapies are the Company’s biotherapeutic platforms with three cardiac clinical stage product candidates in development. These therapies are enabled by its Helix™ biotherapeutic delivery and Morph® vascular navigation product platforms, and soon the Heart3D™ fusion imaging platform. BioCardia selectively partners on biotherapeutic delivery with peers developing important biologic therapies. For more information visit www.biocardia.com. Forward Looking Statements This press release contains forward-looking statements that are subject to risks and uncertainties. Forward-looking statements include, among other things, the presentation of data and future conferences, the protection of the Company’s intellectual property estate, and references to the Company’s investigational product candidates. These forward-looking statements are made as of the date of this press release, and BioCardia assumes no obligation to update the forward-looking statements. We may use terms such as “believes,” “estimates,” “anticipates,” “expects,” “plans,” “intends,” “may,” “could,” “might,” “will,” “should,” “approximately” or other words that convey the uncertainty of future events or outcomes to identify these forward-looking statements. Although we believe that we have a reasonable basis for each forward-looking statement contained herein, we caution you that forward-looking statements are not guarantees of future performance and that our actual results may differ materially from the forward-looking statements contained in this press release. As a result of these factors, we cannot assure you that the forward-looking statements in this press release will prove to be accurate. Additional factors that could materially affect actual results can be found in BioCardia’s Form 10-K filed with the Securities and Exchange Commission on March 26, 2025, under the caption titled “Risk Factors,” and in our subsequently filed Quarterly Reports on Form 10-Q. BioCardia expressly disclaims any intent or obligation to update these forward-looking statements, except as required by law. Media Contact: Miranda Peto, Investor RelationsEmail: mpeto@BioCardia.comPhone: 650-226-0120 Investor Contact: David McClung, Chief Financial OfficerEmail: investors@BioCardia.comPhone: 650-226-0120
Instylla Initiates Commercial Launch with First Use of the Embrace™ Hydrogel Embolic System
Milestone marks the commercial use of the first and only liquid embolic approved for hypervascular tumor embolization supported by a prospective, randomized, controlled clinical trial BEDFORD, Mass., Jan. 28, 2026 /PRNewswire/ — Instylla, Inc., a privately held company developing novel…
Milestone Pharmaceuticals to Present New Analysis from Investigational Etripamil Nasal Spray Clinical Trials for PSVT at the American Heart Association Scientific Sessions 2025
Data show consistent efficacy, safety, and tolerability findings across multiple clinical trials that evaluated self-administered etripamilMONTREAL and CHARLOTTE, N.C., Nov. 03, 2025 (GLOBE NEWSWIRE) — Milestone® Pharmaceuticals Inc. (Nasdaq: MIST), a biopharmaceutical company focused on the development and commercialization of innovative cardiovascular medicines, today announced that new data analyses surrounding its lead investigational product for the management of paroxysmal supraventricular tachycardia (PSVT), etripamil nasal spray, consistently showed efficacy and a favorable safety profile across multiple studies comprising the global development program. The poster, “Combined Efficacy, Safety, and Test Dose Tolerability of Etripamil for Acute Paroxysmal Supraventricular Tachycardia (PSVT) Across Multiple Clinical Trials,” will be presented on Monday, November 10 (10:30–11:30 AM CST) at the American Heart Association Scientific Sessions 2025, in New Orleans, La. The data will be presented by James Ip, M.D., Professor of Medicine and Director of Cardiac Pacing and Implantable Devices, Division of Cardiology, Department of Medicine, New York Weill Cornell-Presbyterian Hospital. Key Findings: Analysis of data from 622 unique enrolled patients across multiple trials showed consistent efficacy of etripamil across multiple study phases, types of trial design, and geographic regions, with etripamil treatment showing similarly greater conversion rates for symptomatic PSVT episodes compared with placebo arms.Across studies, the median time to conversion in patients self-administering etripamil was 18.5 minutes (95% CI: 15.7 to 21.0 minutes).The Kaplan-Meier estimate, from the pooled data for conversion of PSVT to sinus rhythm (SR) by 30 minutes of drug administration, was 59.6% (range: 53.6% to 64.3%). By 60 minutes post-administration, etripamil conversion rates rose to 63.2% to 75.1% across studies.Safety data from over 1,050 etripamil-treated patients were favorable, consistently shown, and characterized by predominantly mild, transient, and nasal-site localized adverse events.The low rate of test-dose failures (1.4%) among over 1,100 patients administered etripamil in SR further indicates the consistent tolerability of etripamil. “These results show the consistent efficacy, with onset soon after self-administration, and safety of etripamil nasal spray across a wide range of studies and patient populations,” said David Bharucha, M.D., PhD, FACC, Chief Medical Officer of Milestone. “The potential for patients to self-administer etripamil and achieve rapid termination of symptomatic PSVT episodes would represent a meaningful advance in the management of PSVT, a condition that frequently leads to emergency room visits and causes significant burden for patients.” Study Methods This analysis involved a systematic review of randomized-controlled and open-label trials evaluating etripamil in adult patients with documented PSVT. Studies were selected based on their focus on self-administered etripamil for acute termination of symptomatic PSVT episodes, with inclusion criteria encompassing patients aged 18 years or older with a history of PSVT confirmed by electrocardiogram (ECG). These studies included: Phase 2: NODE-1; Phase 3: NODE-301 (Parts 1 and 2 [RAPID] and its extensions); and NODE-302 and NODE-303 (open-label extensions). A New Drug Application (NDA) for CARDAMYST is currently being evaluated by the U.S. Food & Drug Administration (FDA) which has set a new Prescription Drug User Fee Act (PDFUA) target date of December 13, 2025. About Etripamil Etripamil is Milestone’s lead investigational product. It is a novel calcium channel blocker nasal spray under clinical development for frequent and often highly symptomatic episodes of PSVT and AFib-RVR. It is designed as a self-administered rapid response therapy for patients, thereby bypassing the need for immediate medical oversight. If approved, etripamil is intended to provide health care providers with a new treatment option to enable on-demand care and patient self-management. This portable, self-administered treatment may provide patients with active management and a greater sense of control over their condition. CARDAMYST™, the conditionally approved brand name for etripamil nasal spray, is well studied with a robust clinical trial program that includes a completed Phase 3 clinical-stage program for the treatment of PSVT and Phase 2 trial for the treatment of patients with AFib-RVR. About Milestone Pharmaceuticals Milestone Pharmaceuticals Inc. (Nasdaq: MIST) is a biopharmaceutical company developing and commercializing innovative cardiovascular medicines to benefit people living with certain heart conditions. Milestone recently submitted a New Drug Application (NDA) to the U.S. Food and Drug Administration (FDA) for etripamil for the treatment of an abnormal heart rhythm, paroxysmal supraventricular tachycardia or PSVT. Forward-Looking Statements This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. Words such as “believe,” “continue,” “could,” “demonstrate,” “designed,” “develop,” “estimate,” “expect,” “may,” “pending,” “plan,” “potential,” “progress,” “will”, “intend” and similar expressions (as well as other words or expressions referencing future events, conditions, or circumstances) are intended to identify forward-looking statements. These forward-looking statements are based on Milestone’s expectations and assumptions as of the date of this press release. Each of these forward-looking statements involves risks and uncertainties. Actual results may differ materially from these forward-looking statements. Forward-looking statements contained in this press release include statements regarding: expectations in regards to etripamil’s efficacy; CARDAMYST’s potential as a novel treatment option to help patients with PSVT; the timing of the PDUFA date; and other statements not related to historical facts. Important factors that could cause actual results to differ materially from those in the forward-looking statements include, but are not limited to, whether our future interactions with the FDA will have satisfactory outcomes; whether and when, if at all, our NDA for etripamil will be approved by the FDA; uncertainties related to the timing of initiation, enrollment, completion, evaluation and results of our clinical trials; risks and uncertainty related to the complexity inherent in cleaning, verifying and analyzing trial data; and whether the clinical trials will validate the safety and efficacy of etripamil for PSVT or other indications, among others, general economic, political, and market conditions, including deteriorating market conditions due to investor concerns regarding inflation, international tariffs, Russian hostilities in Ukraine and ongoing disputes in Israel and Gaza and overall fluctuations in the financial markets in the United States and abroad, risks related to pandemics and public health emergencies, and risks related the sufficiency of Milestone’s capital resources and its ability to raise additional capital in the current economic climate. These and other risks are set forth in Milestone’s filings with the U.S. Securities and Exchange Commission (SEC), including in its annual report on Form 10-K for the year ended December 31, 2025 and its quarterly report on Form 10-Q for the quarter ended June 30, 2025, in each case under the caption “Risk Factors,” as such discussions may be updated from time to time by subsequent filings Milestone may make with the SEC. Except as required by law, Milestone assumes no obligation to update any forward-looking statements contained herein to reflect any change in expectations, even as new information becomes available. Investor Relations Kevin Gardner, kgardner@lifesciadvisors.com