Mid-Summer Launch Anticipated for Next-Generation Oncology Vascular Access Platform MIAMI, May 18, 2026 /PRNewswire/ — PorTal Access, Inc., a medical device company pioneering next-generation vascular access solutions for oncology, today announced the successful completion of its U.S….
Uncategorized
RingConn Gen 3 Smart Ring Opens for Pre-Order: Bringing Vascular Insights to Effortless Everyday Wear
World’s Leading Provider of Cutting-Edge Smart Ring Solutions NEW YORK, May 10, 2026 /PRNewswire/ — RingConn opened Gen 3 pre-orders on May 5th ahead of its official May 29th launch, marking the occasion with a feature on the iconic One Times Square billboard in New York City on the…
Kestra Showcases Accelerated Innovation at HRS 2026
Kestra Medical Technologies booth at HRS 2026
Kestra Medical Technologies will showcase its Cardiac Recovery System® platform at Heart Rhythm 2026 (HRS). At Booth 2119, attendees can explore the platform through an immersive multimedia experience, experience the ASSURE® system firsthand, and review clinical data from the ACE-PAS study.
KIRKLAND, Wash., April 22, 2026 (GLOBE NEWSWIRE) — Kestra Medical Technologies, Ltd. (Nasdaq: KMTS), a leading wearable medical device and digital healthcare company, today announced it will exhibit at Heart Rhythm 2026, the annual meeting of the Heart Rhythm Society (HRS) taking place April 23–26 in Chicago. The company will showcase its Cardiac Recovery System® platform—a modern, integrated approach to managing sudden cardiac arrest (SCA) risk that unites proven therapy, actionable clinical insights, and ongoing patient support to enhance protection and optimize care. Kestra, one of the fastest-growing companies in medical technology, continues to build momentum as clinicians increasingly shift toward more connected approaches to SCA risk management. At HRS, attendees will see why more clinicians are turning to Kestra to improve patient protection and better manage risk during the most vulnerable stages of recovery. The ASSURE® WCD is at the center of the Cardiac Recovery System platform. Insights from the ASSURE WCD Clinical Evaluation Post-Approval Study (ACE-PAS)—the largest prospective study of wearable defibrillators to date—demonstrate robust real-world performance and continue to guide innovation across the platform. At HRS, Kestra is launching a recently FDA-approved ASSURE WCD algorithm update that builds on market-leading clinical performance and applies real-world data to further enhance patient protection and advance care. “At Kestra, innovation is a core principle, so we are excited to further advance the clinical benefits of the ASSURE WCD with this new algorithm. We’re seeing a clear shift in how clinicians approach SCA risk, with a growing emphasis on patient compliance and connected models of care,” said Brian Webster, President and Chief Executive Officer. “HRS is a pivotal moment to engage with prescribers and demonstrate how our clinically informed innovation is driving that momentum—delivering meaningful improvements for clinicians and their patients.” Kestra will be located at Booth 2119, where attendees can experience the ASSURE system firsthand, explore the Cardiac Recovery System platform through an immersive multimedia experience, and review clinical data from ACE-PAS. About Kestra Kestra Medical Technologies, Ltd. is a leading wearable medical device and digital healthcare company focused on transforming patient outcomes in cardiovascular disease using monitoring and therapeutic intervention technologies that are intuitive, intelligent, and connected. For more information, please visit www.kestramedical.com. Forward-Looking StatementsExcept where otherwise noted, the information contained in this press release is as of April 22, 2026. Statements in this press release that express a belief, expectation or intention, as well as those that are not historical fact, are forward-looking statements. Except as required by law, we undertake no obligation to publicly update any forward-looking statements, whether as a result of new information, future events or otherwise. This press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. Given their forward-looking nature, these statements involve substantial risks, uncertainties and potentially inaccurate assumptions, and we cannot ensure that any outcome expressed in these forward-looking statements will be realized in whole or in part. You can identify these statements by the fact that they use future dates or use words such as “will,” “may,” “could,” “likely,” “ongoing,” “anticipate,” “estimate,” “expect,” “project,” “intend,” “plan,” “believe,” “assume,” “target,” “forecast,” “guidance,” “goal,” “objective,” “aim,” “seek,” “potential,” “hope” and other words and terms of similar meaning. Among the factors that could cause actual results to differ materially from past results and future plans and projected future results are the following: risks related to our limited operating history and history of net losses; our ability to successfully achieve substantial market adoption of our products; competitive pressures; our ability to adapt our manufacturing and production capacities to evolving patterns of demand, governmental actions and customer trends; product defects or complaints and related liability; our ability to obtain and maintain adequate coverage and reimbursement levels for our products; our ability to comply with changing laws and regulatory requirements and resulting costs; our dependence on a limited number of suppliers; and other risks and uncertainties, including those described under the heading “Risk Factors” in Kestra’s Annual Report on Form 10-K for the fiscal year ended April 30, 2025 filed with the U.S. Securities and Exchange Commission (“SEC”) on July 17, 2025, and in other periodic reports filed by Kestra with the SEC. These filings are available on the Investor Relations section of our website at https://investors.kestramedical.com/ and on the SEC’s website at https://sec.gov/. A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/93288f45-4a54-4de1-b66e-207047603f80 CONTACT: Media contact
Rhiannon Pickus
rhiannon.pickus@kestramedical.com
Investor contact
Neil Bhalodkar
neil.bhalodkar@kestramedical.com
UltraGreen Cardiac Technologies launches Partnership with LifeSignals to Expand Access to Advanced Wearable Cardiac Monitoring Biosensors Globally
NEW YORK, April 21, 2026 /PRNewswire/ — UltraGreen Cardiac Technologies, a business unit of UltraGreen.ai (“UltraGreen” or the “Company”), a global leader in digital health solutions, today announced a strategic partnership with LifeSignals, a pioneer in wearable biosensor technology, to…
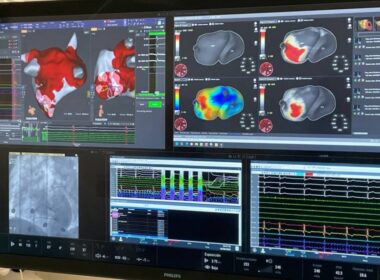
Corify Care Validates 4-D Global Cardiac Mapping for Complex Atrial and Ventricular Arrhythmias
New publications in Heart Rhythm and Europace demonstrate how single-beat mapping provides instant clinical clarity, optimizing ablation strategies and improving patient safety. MADRID, April 21, 2026 /PRNewswire/ — Corify Care today announced a major clinical milestone with the…
EHRA 2026 Studies Reveal Why False Positives Persist in AI-Equipped Implantable Cardiac Monitors
Research identifies guideline-based interpretation gaps and signal-detection issues behind non-actionable alerts and shows how an additional cloud-based AI layer can significantly reduce clinician review burden while maintaining high sensitivity April 14, 2026 08:00 ET | Source: Implicity CAMBRIDGE, Mass., April 14, 2026 (GLOBE NEWSWIRE) — Implicity, a leader in remote patient monitoring […]
Sirtex Medical’s DOORwaY90 Study Demonstrates 100% Local Tumor Control with SIR-Spheres®, Setting a New Benchmark in Y-90 for HCC
The DOORwaY90 study met its prespecified co-primary endpoints, achieving a best overall response rate (ORR) of 99% WOBURN, Mass., April 13, 2026 /PRNewswire/ — Sirtex Medical (“Sirtex”), a leading manufacturer of interventional oncology and embolization solutions, today announced…
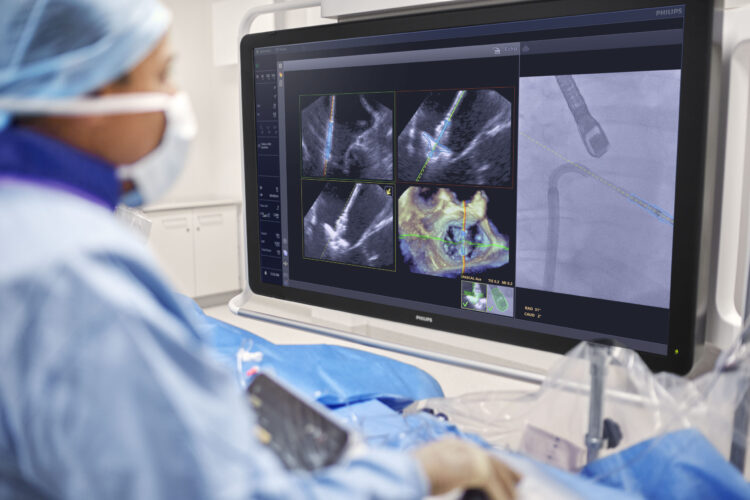
FDA clears Philips AI solution that provides real-time guidance during complex minimally invasive heart valve repair
Physician using DeviceGuide with EchoNavigator 5
Physician using DeviceGuide with EchoNavigator 5
Philips DeviceGuide with EchoNavigator 5 Screen
Philips DeviceGuide with EchoNavigator 5 Screen
DeviceGuide with EchoNavigator 5
DeviceGuide with EchoNavigator 5
March 26, 2026 Philips DeviceGuide uses AI to track and visualize mitral valve repair devices [1] in real time during minimally invasive procedures, supporting treatment [2]DeviceGuide integrates with Philips Azurion image-guided therapy platform that combines imaging, real-time data and intelligent software within a unified workflow to support more consistent, efficient and confidently guided minimally invasive procedures Amsterdam, the Netherlands – Royal Philips (NYSE: PHG, AEX: PHIA), a global leader in health technology, today announced it has received U.S. Food and Drug Administration (FDA) 510(k) clearance for EchoNavigator R5.0 with DeviceGuide, an AI-powered software solution that assists physicians during one of interventional cardiology’s most technically demanding procedures – repairing leaking mitral valves through a minimally invasive approach. DeviceGuide was developed in close collaboration with Edwards Lifesciences, the global leader in structural heart innovation, aligning Philips’ imaging and AI expertise with Edwards’ expertise in valvular heart therapy development. Together, the companies have innovated image guidance of the mitral transcatheter edge-to-edge repair (M-TEER) workflow to make these complex minimally invasive heart valve repair procedures more intuitive and streamlined. DeviceGuide will be showcased at the American College of Cardiology (ACC) 2026 meeting in New Orleans, one of the world’s leading forums for cardiovascular innovation. Transforming treatment for a common heart conditionA leaking mitral valve, known as mitral regurgitation (MR), affects more than 35 million adults worldwide and over 2 million in the U.S. [3, 4]. Minimally invasive transcatheter techniques such as M-TEER offer an alternative to open heart surgery in selective patient populations. During M-TEER procedures, physicians make a small skin incision to access a vein which allows the introduction of a catheter to the heart to deliver a repair device to the diseased mitral valve. Because the mitral valve is inherently a complex and heterogeneous structure, transcatheter repair of the valve requires experienced physicians positioning the device delivery system, and physicians guiding the placement of the device. In guiding and positioning the repair device, the intraprocedural heart team must interpret both X-ray and ultrasound images on multiple screens, communicate and coordinate movements between two operators, and make precise adjustments to grasp the moving valve leaflets and then confirm the result in real time. The process demands accuracy, coordination, and experience from the entire team – and this is where DeviceGuide can help with navigation guidance. How AI assists inside the beating heartBuilt on Philips’ EchoNavigator echo-fluoro fusion technology, which combines live echocardiography images from Philips EPIQ CVxi cardiovascular platform with live X-ray images from Philips Azurion image-guided therapy system, DeviceGuide brings real-time AI guidance directly into the procedure room. The software’s AI algorithm automatically tracks and visualizes the Edwards PASCAL Ace mitral valve repair device, combining live ultrasound and X-ray images into a single, integrated view. This helps clinicians navigate and position the device with greater clarity and confidence. “The AI software serves as an assistive tool; the physician always remains in control. This isn’t about replacing expertise – it’s about amplifying it,” explains Dr. Atul Gupta, Chief Medical Officer Diagnosis & Treatment at Philips. “By embedding AI into the procedure, DeviceGuide gives physicians an extra pair of eyes, helping them treat more patients safely and confidently.” Collaboration with clinical partnersIn developing DeviceGuide, Philips and Edwards worked closely with investigational sites in Europe and the U.S., including a team at the Structural Heart and Valve Center at NewYork-Presbyterian/Columbia University Irving Medical Center, led by interventional cardiologist Susheel Kumar Kodali, MD, director, and Rebecca T. Hahn, MD, director of interventional echocardiography.“In helping to guide mitral repair procedures, one of my roles as an echocardiographer is to help the interventional cardiologist understand the complex anatomy of the valve which will determine the orientation, trajectory and position of the repair device relative to the target and the surrounding structures,” said Dr. Hahn. “Since AI auto-aligns imaging to the device in real time and continuously informs the interventionalist about the location of the device in space on the imaging screen, it minimizes unnecessary repositioning of the imaging window, streamlines procedural guidance and may improve the precision of device implantation.” “DeviceGuide provides me with a visual overlay, trajectory line and orientation line of the therapy device in both live 3D echo and fluoroscopic images during mitral valve repair procedures,” said Dr. Kodali. “Having a single, intuitive presentation of real-time target, orientation and auto device-aligned views simplifies this procedure and improves team communication.” Supporting teams in real time“Structural heart procedures are among the fastest-growing areas in cardiology, and also among the most complex,” said Mark Stoffels, Business Leader, Image Guided Therapy Systems at Philips. “By assisting physicians with real-time visualization and navigation inside the beating heart, DeviceGuide helps them manage that complexity, and perform procedures more confidently, with the ultimate goal to treat more patients effectively. It fits seamlessly into cath-lab workflows and gives the entire team a shared view of the procedure, improving coordination and confidence.” Part of Philips’ connected cardiology ecosystemDeviceGuide extends Philips’ connected cardiology portfolio that helps physicians care for heart patients from diagnosis through recovery. It also represents a step toward Philips’ vision of the AI-powered cath lab of the future where imaging, devices, and real-time data are intelligently connected to reduce procedural complexity and improve consistency. In hospitals, Philips systems already bring together ultrasound, X-ray, and real-time data into a unified workflow to guide minimally invasive procedures. Building on this foundation, intelligent software solutions such as DeviceGuide help care teams navigate complex structural heart interventions with greater clarity and coordination, serving as an assistive “copilot” in the procedure room.After treatment, Philips connected monitoring tools extend care beyond the cath lab, helping care teams detect complications early and support recovery. Together, these innovations create a connected cardiology ecosystem that helps care teams diagnose earlier, treat less invasively, improve procedural efficiency, and support recovery – improving outcomes and experiences for heart patients everywhere. AvailabilityDeviceGuide enabled by EchoNavigator is FDA 510(k) cleared in the United States. Commercial availability is subject to market release and applicable regulatory requirements. DeviceGuide is currently intended for use with the Edwards PASCAL Ace Mitral Valve Repair System. Availability outside the U.S. varies by country and regulatory status. A recent first-in-human publication in JACC: Case Reports describes early clinical experience using DeviceGuide for AI-supported imaging guidance during mitral transcatheter edge-to-edge repair procedures. Read more on how Philips DeviceGuide works here. Philips DeviceGuide enabled by EchoNavigator R5 is not available for sale or use in all countries. Its availability is subject to local regulatory clearance and market release. Please contact your Philips representative for details on product availability in your region. Dr. Hahn reports Institutional funding to Columbia University and/or the Cardiovascular Research Foundation from Philips North America LLC, and from Edwards Lifesciences Corporation.Dr. Kodali discloses consulting payments from Philips North America LLC and reports Institutional funding to Columbia University and/or the Cardiovascular Research Foundation from Edwards Lifesciences Corporation. REFERENCES:[1] DeviceGuide is currently intended for use only with the Mitral TEER Therapy Device (Edwards PASCAL Ace); Hahn RT, Biaggi P, Corti R et al. Mitral Transcatheter Edge-to-Edge Repair Using Novel Augmented Imaging Software. JACC: Case Reports 2025;30:106160.[2] DeviceGuide assists physicians in visualizing and navigating the repair device within the heart. It supports, but does not perform, the therapeutic procedure itself; Biaggi P, Corti R, Gaemperli O et al. Artificial intelligence based fusion imaging streamlining mitral transcatheter edge-to-edge repair. European Heart Journal – Imaging Methods and Practice 2026;4.[3] MDPI[4] CDC For further information, please contact: Joost MalthaPhilips Global External RelationsTel.: +31 6 1055816E-mail: joost.maltha@philips.com Avi DinesPhilips North AmericaTel: +1 781 690 3814E-mail: avi.dines@philips.com About Royal Philips Royal Philips (NYSE: PHG, AEX: PHIA) is a leading health technology company focused on improving people’s health and well-being through meaningful innovation. Philips’ patient- and people-centric innovation leverages advanced technology and deep clinical and consumer insights to deliver personal health solutions for consumers and professional health solutions for healthcare providers and their patients in the hospital and the home. Headquartered in the Netherlands, the company is a leader in diagnostic imaging, ultrasound, image-guided therapy, monitoring and enterprise informatics, as well as in personal health. Philips generated 2025 sales of EUR 18 billion and employs approximately 64,800 employees with sales and services in more than 100 countries. News about Philips can be found at www.philips.com/newscenter.
Attachments
Physician using DeviceGuide with EchoNavigator 5
Philips DeviceGuide with EchoNavigator 5 Screen
DeviceGuide with EchoNavigator 5
XCath Makes Medical History With the World’s First Remote Robotic Intervention in a Stroke Patient
HOUSTON & PANAMA CITY, Panama–(BUSINESS WIRE)–XCath announced successful completion of world’s first telerobotic mechanical thrombectomy for stroke using the XCath Iris Surgical Robotic System.
AngioDynamics to Report Fiscal 2026 Third Quarter Results on April 2, 2026
LATHAM, N.Y.–(BUSINESS WIRE)–AngioDynamics, Inc. (NASDAQ: ANGO), a leading and transformative medical technology company focused on restoring healthy blood flow in the body’s vascular system, expanding cancer treatment options and improving patient quality of life, today announced that it will report financial results for the third quarter of fiscal year 2026 before the market open on Thursday, April 2, 2026. The Company’s management will host a conference call at 8:00 am ET the same day to d