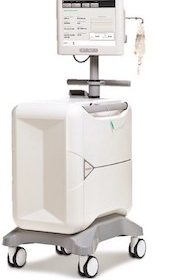
CHELMSFORD, Mass.–(BUSINESS WIRE)–ZOLL® Medical Corporation, an Asahi Kasei Group company that manufactures medical devices and related software solutions, announced the launch of and first patient prescription for the ZOLL µCor™ (pronounced “Micro Core”) Heart Failure and Arrhythmia Management System (HFAMS), designed to help clinicians improve outcomes and reduce hospitalizations for […]
Other News
Endologix Announces Reinstatement of CE Mark for its Nellix EndoVascular Aneurysm Sealing System
IRVINE, Calif.–(BUSINESS WIRE)–Endologix® Inc. (Nasdaq: ELGX), a developer and marketer of innovative treatments for aortic disorders, announced today that the EC Certificate of Conformity (CE Mark) for the Nellix EndoVascular Aneurysm Sealing System (Nellix System) has been reinstated by GMED, the EU Notified Body for the Nellix System. The reinstatement […]
Precision Wellness Validates Performance of Predictive Risk Models to Power Precision Health
PALO ALTO, Calif.–(BUSINESS WIRE)–Precision Wellness, Inc. a Silicon Valley-based company at the intersection of next-generation AI, big data analytics and the latest medical science, today announced the validation of its cardiometabolic disease (CMD) predictive risk engine. The study was performed as part of a sponsored research with Stanford University to evaluate 416,936 […]
Jubilant DraxImage Inc. Receives CE Mark Certificate for Its Medical Device RUBY Rubidium Elution System™ (RbES) and Proprietary RUBY™ Consumable Accessories
MONTREAL, June 7, 2019 /PRNewswire/ — Jubilant DraxImage Inc. (“DraxImage”), a wholly owned subsidiary of Jubilant Pharma Limited, is pleased to announce the receipt of the CE Mark certificate for its state-of-the-art RUBY Rubidium Elution System (RbES) and the proprietary RUBY consumable accessories dedicated for use with their RUBY-FILL®(Rubidium Rb82 Generator). This certificate allows the […]
Itamar Medical’s WatchPAT™ One, the First and Only Fully Disposable Home Sleep Apnea Test Receives FDA 510(k) Clearance
New system expands WatchPAT access to additional sleep centers by reducing infrastructure needs and eliminating capital requirements; also reduces infection risk for in-patient sleep apnea testing CAESAREA, Israel, June 06, 2019 (GLOBE NEWSWIRE) — Itamar Medical Ltd. (Nasdaq:ITMR) (TASE:ITMR), a company that develops, manufactures and markets non-invasive diagnostic medical devices […]
BIOXYTRAN’S MDX VIEWER DEMONSTRATES EARLY WARNING SIGNS OF BRAIN HEALTH DURING CARDIOVASCULAR SURGERIES
MDX Viewer use during open heart surgery is an essential vital sign that could mitigate the risk of brain damage caused by prolonged oxygen deprivation during perioperative cardiovascular procedures and validates tissue oxygenation in the brain for stoke patients BOSTON, MASSACHUSETTS, June 06, 2019 (GLOBE NEWSWIRE) — BIOXYTRAN, INC. (OTC: […]
Quantum Genomics Enrolls First Patient in QUORUM Phase IIb Study of Firibastat in Heart Failure Patients
PARIS and NEW YORK, June 06, 2019 (GLOBE NEWSWIRE) — Quantum Genomics (Euronext Growth – FR0011648971 – ALQGC), a biopharmaceutical company specializing in the development of a new drug class that directly targets the brain to treat resistant hypertension and heart failure, announces the enrollment of the first patient in its QUORUM […]
BioSig Allowed a U.S. Patent for PURE EP™ Simulator
Santa Monica, CA, June 06, 2019 (GLOBE NEWSWIRE) — BioSig Technologies, Inc. (NASDAQ: BSGM), a medical device company developing a proprietary biomedical signal processing platform designed to address an unmet technology need for the electrophysiology (EP) marketplace, today announced that the U.S. Patent & Trademark Office allowed a U.S. patent application covering its […]
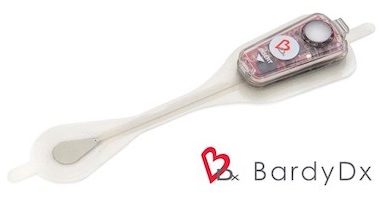
Bardy Diagnostics™ Selected as Winner of the MedTech Breakthrough Award For Best New Diagnostic Technology
SEATTLE, June 6, 2019 /PRNewswire/ — Bardy Diagnostics, Inc., (“BardyDx”), a leading provider of ambulatory cardiac monitoring technologies and custom data solutions, announced that MedTech Breakthrough, an independent organization that recognizes the top companies and solutions in the global health and medical technology market, has recognized the Carnation Ambulatory Monitor (CAM™) with the […]
Texas Cardiac Arrhythmia Institute at St. David’s Medical Center opening new Electrophysiology Center
AUSTIN, Texas, June 4, 2019 /PRNewswire/ — The Texas Cardiac Arrhythmia Institute (TCAI) at St. David’s Medical Center is opening a new, state-of-the-art Electrophysiology Center dedicated to treating patients with cardiac arrhythmias. The new Electrophysiology Center is set to open at the end of the month. “This expansion at the Texas Cardiac Arrhythmia Institute […]