FLAGSTAFF, Ariz.–(BUSINESS WIRE)–W. L. Gore & Associates, Inc. (Gore) announces the first U.S. implant of the GORE® TAG® Conformable Thoracic Stent Graft with ACTIVE CONTROL System. The successful procedure was performed by William Jordan, M.D., chief of the Division of Vascular Surgery and Bradley Leshnower, M.D., cardiothoracic surgeon, at the Emory […]
Other News
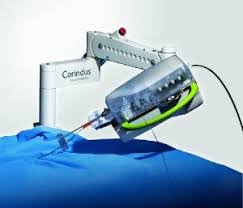
Corindus Announces First Commercial Installation of CorPath® GRX System in South America
WALTHAM, Mass.–(BUSINESS WIRE)–Corindus Vascular Robotics, Inc. (“Corindus” or the “Company”) (NYSE American: CVRS), a leading developer of precision vascular robotics, announced today that Albert Einstein Jewish Hospital (Hospital Israelita Albert Einstein) in São Paulo, Brazil has become the first hospital in the Southern Hemisphere to implement Corindus’ CorPath GRX System. Ranked as the […]
Orchestra BioMed™ Completes $34 Million Financing to Accelerate Product Pipeline Development and Advance Partnerships
Series B-1 Round Led by Perceptive, RTW and Soleus Capital with Strategic Participation from Terumo Corporation Secures $64 Million in 2019 Capital Inflows, Including $30 Million Upfront Payment from Terumo Partnership Financing Structure Provides Up to an Additional $57 Million in Follow-On Investments from Existing Shareholders NEW HOPE, Pa., July […]
Acutus Medical Announces Addition of Scott Huennekens as Executive Chairman of the Board
CARLSBAD, Calif., July 10, 2019 /PRNewswire/ — Acutus Medical today announced the appointment of Scott Huennekensas Executive Chairman of the Board of Directors. Mr. Huennekens has more than 25 years of experience in the medical device industry, specializing in clinical advancement through innovative R&D and technology-based initiatives. He has held a number of executive leadership […]
Opella Ernest, M.D., Appointed President Of American Heart Association’s Metro Chicago Board of Directors
CHICAGO, July 10, 2019 /PRNewswire/ — The American Heart Association today announced Opella Ernest, M.D., has been appointed president of the American Heart Association’s Metro Chicago Board of Directors. Dr. Ernest, senior vice president and chief clinical officer at Health Care Service Corporation (HCSC), previously served as a member on the Metro Chicago […]
Merit Medical Systems to Announce Second Quarter Results on July 25, 2019
SOUTH JORDAN, Utah, July 09, 2019 (GLOBE NEWSWIRE) — Merit Medical Systems, Inc. (NASDAQ: MMSI), a leading manufacturer and marketer of proprietary disposable devices used primarily in cardiology, radiology and endoscopy, announced today that it will release its financial results for the second quarter ended June 30, 2019, after the […]
RAPID Receives Registration Approval in Japan
MENLO PARK, Calif.–(BUSINESS WIRE)–iSchemaView, the worldwide leader in advanced imaging for stroke, has received registration approval under the Japanese Pharmaceutical Affairs Law through third party review by the Japanese Association for the Advancement of Medical Equipment for its RAPID imaging platform. With this news the technology is available to hospitals […]
Resverlogix’s BETonMACE Phase 3 Epigenetics Trial Completes Final Safety Visits
CALGARY, Alberta, July 08, 2019 (GLOBE NEWSWIRE) — Resverlogix Corp. (“Resverlogix” or the “Company”) (TSX:RVX) is pleased to announce today that all patients active in BETonMACE, the Company’s event-based, phase 3 registration trial, recently completed their final, follow-up safety visits, marking another important step towards trial completion. Given the remaining steps […]
Medtronic HeartWare(TM) HVAD(TM) Implanted via Less Invasive Thoracotomy Shows 95 Percent Freedom from Disabling Stroke at Two Years
DUBLIN, July 08, 2019 (GLOBE NEWSWIRE) — Medtronic plc (NYSE:MDT) today announced two-year outcomes from the LATERAL clinical trial evaluating the use of its HeartWare™ HVAD™ System in patients who received the system via a less-invasive, thoracotomy implant approach. The HVAD System is a heart pump, called a left ventricular assist device […]
BIOTRONIK Receives FDA Clearance on Next-Gen Injectable Cardiac Monitor: BIOMONITOR III
LAKE OSWEGO, Ore., July 8, 2019 /PRNewswire/ — BIOTRONIK today announced FDA clearance of the BIOMONITOR III injectable cardiac monitor (ICM). BIOMONITOR III is designed to document suspected arrhythmia or unexplained syncope with increased clarity, enabling fast diagnosis and appropriate treatment. Early detection of arrhythmia through ICMs enables physician intervention before adverse outcomes occur. Atrial fibrillation […]