TOLEDO, Ohio, June 4, 2019 /PRNewswire/ — VentureMed Group, Inc., a privately-held medical device innovator in vessel preparation for interventional treatment of peripheral arterial disease (PAD) and stenoses of arteriovenous (AV) fistulas and grafts, announced new data presented at the 20th Annual New Cardiovascular Horizons (NCVH) Annual Conference, May 29-31, in New Orleans, Louisiana. FLEX iDissection Study Results Presentation The study […]
Other News
GORE® CARDIOFORM ASD Occluder Receives FDA Approval for the Treatment of Atrial Septal Defects
FLAGSTAFF, Ariz., June 4, 2019 /PRNewswire/ — W. L. Gore & Associates (Gore) announced the U.S. Food and Drug Administration’s (FDA’s) premarket approval (PMA) of the GORE® CARDIOFORM ASD Occluder for the percutaneous closure of ostium secundum atrial septal defects (ASDs). The FDA approval was supported by data collected from the pivotal stage of the Gore […]
AngioDynamics Completes Sale of NAMIC® Fluid Management Portfolio to Medline Industries, Inc.
LATHAM, N.Y.–(BUSINESS WIRE)–AngioDynamics, Inc. (NASDAQ: ANGO), a leading provider of innovative, minimally invasive medical devices for vascular access, peripheral vascular disease, and oncology, today announced that it closed the previously announced sale of its NAMIC® fluid management portfolio to Medline Industries, Inc. on May 31, 2019 for $167.5 million. AngioDynamics has […]
BioSig Technologies Receives $4.6 Million in Cash Warrant and Option Exercises
Email Print Friendly Share June 03, 2019 07:52 ET | Source: BioSig Technologies, Inc. Santa Monica, CA, June 03, 2019 (GLOBE NEWSWIRE) — BioSig Technologies, Inc. (NASDAQ: BSGM), a medical technology company developing a proprietary biomedical signal processing platform designed to address an unmet technology need for the electrophysiology (EP) marketplace, today announced that it received a […]
Ancora Heart Enrolls First Patient in U.S. Early Feasibility Study of First-of-Its-Kind Investigational Heart Failure Therapy
SANTA CLARA, Calif.–(BUSINESS WIRE)–Ancora Heart, Inc., a company developing a novel therapy to address heart failure, today announced the first patient was enrolled in a U.S. early feasibility study evaluating the AccuCinch® Ventricular Repair System as a treatment for patients with reduced ejection fraction systolic heart failure (HFrEF). The first patient […]
CHF Solutions, Inc. Reports Inducement Grants Under NASDAQ Listing Rule 5635(c)(4)
EDEN PRAIRIE, Minn., May 29, 2019 (GLOBE NEWSWIRE) — CHF Solutions, Inc. (NASDAQ: CHFS), today announced that, on May 23, 2019, the independent directors approved five equity awards under CHF Solution’s New-Hire Equity Incentive Plan, as material inducements to five individuals entering into employment with the Company. The equity awards […]
Safety and efficacy of Philips’ Stellarex .035 low-dose drug-coated balloon demonstrated in clinical trials at three years
Stellarex is the only low-dose [1] drug-coated balloon (DCB) to demonstrate a significant treatment effect and high safety profile through 3 years ILLUMENATE Pivotal trial showcases durable primary patency (maintained blood flow) in the most complex patient pool studied in a DCB randomized clinical trial No significant difference in mortality […]
Medtronic Gains U.S. FDA Clearance for New Catheter System for HIS Bundle Pacing
DUBLIN – May 29, 2019 – Medtronic plc (NYSE:MDT) today announced U.S. Food and Drug Administration (FDA) clearance and commercial launch for the SelectSite(TM) C304-HIS deflectable catheter system for use in procedures involving His-bundle pacing (HBP). The SelectSite C304-HIS deflectable catheter system features a deflectable, out-of-plane curve to reach the bundle […]
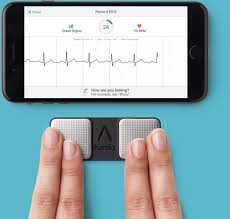
AliveCor and Best Buy Partner to Bring KardiaMobile to Select Stores Nationwide
MOUNTAIN VIEW, Calif., May 29, 2019 /PRNewswire/ — AliveCor, the leader in FDA-cleared consumer electrocardiogram (ECG) technology, and Best Buy Co., Inc. (NYSE: BBY) today announced a strategic partnership, bringing AliveCor’s products to select Best Buy stores across the country. This is the first time AliveCor’s products will be sold in consumer electronics stores […]
Intact Vascular Announces $25 Million Financing with Vensana Capital to Drive Commercialization of the Tack Endovascular System®
WAYNE, Pa.–(BUSINESS WIRE)–Intact Vascular, Inc., a developer of medical devices for minimally invasive peripheral vascular procedures, today announced a $25 million financing with Vensana Capital as the lead investor. Vensana was joined by existing investors including New Enterprise Associates (“NEA”), H.I.G. BioHealth Partners, and Quaker Partners. The Company reported closure […]