RVINE, Calif.–(BUSINESS WIRE)–Endologix, Inc. (the “Company”) (NASDAQ: ELGX), a developer and marketer of innovative treatments for aortic disorders, today announced financial results for the first quarter ended March 31, 2019. “Our first quarter financial and operational performance provides us with a solid foundation to accomplish our goals for the rest of […]
Other News
New Data Emphasizing LivaNova Perceval Valve Durability to be Presented at the American Association for Thoracic Surgery Meeting
LONDON–(BUSINESS WIRE)–LivaNova PLC (NASDAQ:LIVN), a market-leading medical technology company, today announced that new clinical data for its sutureless surgical aortic valve, Perceval®, will be unveiled at this year’s American Association for Thoracic Surgery (AATS) meeting. On May 4, Prof. Bart Meuris from Leuven University Hospital (BE) will present the data […]
Hemostemix Announces Convertible Debenture Financing
CALGARY, Alberta, May 03, 2019 (GLOBE NEWSWIRE) — Hemostemix Inc. (“Hemostemix” or the “Company”) (TSX VENTURE: HEM; OTCQB: HMTXF) a biotechnology company focused on developing and commercializing innovative blood-derived stem cell therapies for medical conditions not adequately addressed by current treatments, is pleased to announce that it intends to complete, […]
Endonovo Therapeutics Hires David Clark as Vice President of Sales
Los Angeles, May 03, 2019 (GLOBE NEWSWIRE) — Endonovo Therapeutics, Inc. (OTCQB: ENDV), today announced veteran sales leader David Clark joined as Endonovo’s Vice President of Sales. In his new position, Mr. Clark will lead the U.S. Sales Team, distributors and strategic partners through the next phase of growth. He’ll […]
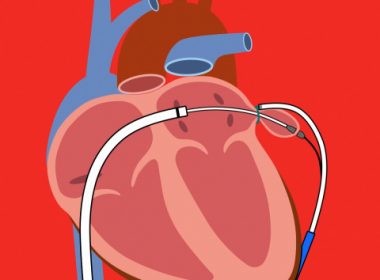
SentreHEART Receives CE Mark for LARIAT-RS™ 50mm and Expanded Indication for Left Atrial Appendage Exclusion
REDWOOD CITY, Calif.–(BUSINESS WIRE)–SentreHEART, Inc., announced it has received CE Mark for the LARIAT-RS™ 50mm as well as expanded labeling for the LARIAT-RS for left atrial appendage (LAA) exclusion. The new 50mm device along with the 45mm LARIAT-RS is now available for sale in Europe. The LARIAT-RS (LARIAT) device is […]
Abiomed Announced Q4 FY 2019 Revenue of $207 Million and 31.6% Operating Margin
DANVERS, Mass.–(BUSINESS WIRE)–Abiomed, Inc. (NASDAQ: ABMD), a leading provider of breakthrough heart recovery and support technologies, today reported fourth quarter fiscal 2019 revenue of $207.1 million, an increase of 19% compared to revenue of $174.4 million for the same period of fiscal 2018. For fiscal year 2019, total revenue was $769.4 […]
Cardiovascular Systems, Inc. Reports Fiscal 2019 Third-Quarter Financial Results
ST. PAUL, Minn.–(BUSINESS WIRE)–Cardiovascular Systems, Inc. (CSI®) (NASDAQ: CSII), a medical device company developing and commercializing innovative interventional treatment systems for patients with peripheral and coronary artery disease, today reported financial results for its fiscal third quarter, ended March 31, 2019. CSI’s third-quarter revenues were $63.3 million, an increase of […]
Surmodics Reports Second Quarter Fiscal 2019 Results
EDEN PRAIRIE, Minn.–(BUSINESS WIRE)–Surmodics, Inc. (Nasdaq: SRDX), a leading provider of medical device and in vitro diagnostic technologies to the healthcare industry, today announced results for its fiscal 2019 second quarter ended March 31, 2019, and updated its financial outlook for fiscal 2019. Summary of Second Quarter and Recent Highlights […]
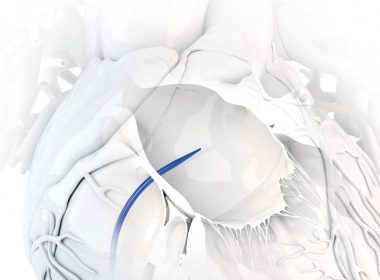
First-of-its-Kind Large Access Dilator Launches in U.S., Following Cleveland Clinic Case
TORONTO, May 2, 2019 /PRNewswire/ – Baylis Medical announced today the U.S. launch and first clinical use of its ExpanSure™ Transseptal Dilation System, used in a left-atrial appendage (LAA) closure case performed at the Cleveland Clinic, in Cleveland, Ohio. The ExpanSure dilator combines a sheath and dilator into a single tool, and facilitates larger […]
Concept Medical Inc. Granted ‘Breakthrough Device Designation’ From FDA for Its MagicTouch Sirolimus Coated Balloon
TAMPA, Florida, May 2, 2019 /PRNewswire/ — Concept Medical Inc. (CMI) has been granted “Breakthrough Device Designation” from the U.S. Food and Drug Administration (FDA) for MagicTouch, its Sirolimus drug-coated balloon (DCB) catheter, for the treatment of coronary in-stent restenosis (ISR). In-stent restenosis (ISR) is the gradual re-narrowing of a stented coronary artery lesion, due to […]