KFAR-SABA, Israel & ALISO VIEJO, Calif.–(BUSINESS WIRE)–CathWorks announced the completion of a $30 million Series C financing round led by Deerfield Management. Jim Corbett, CathWorks CEO, stated, “We are delighted to have such a notable investor leading this round and for the continued full participation of our existing syndicate of […]
Other News
REVA Announces Reduction in Force
SAN DIEGO, Feb. 04, 2019 (GLOBE NEWSWIRE) — REVA Medical, Inc. (ASX: RVA) (“REVA” or the “Company”), a leader in bioresorbable polymer technologies for vascular applications, announced it is realigning the organization to align with current business conditions. Effective February 8, 2019, REVA will reduce its San Diego-based personnel to […]
BioSig Appoints John Kowalski to Lead Commercialization of PURE EP
Santa Monica, CA, Feb. 05, 2019 (GLOBE NEWSWIRE) — BioSig Technologies, Inc. (NASDAQ: BSGM), a medical device company developing a proprietary biomedical signal processing platform designed to address an unmet technology need for the electrophysiology (EP) marketplace, today announced that the Company has appointed Mr. John Kowalski as Vice President of Sales. Mr. […]
Kaneka invests in a US-based medical device company
TOKYO, Feb. 4, 2019 /PRNewswire/ — Kaneka Corporation (Tokyo, Japan, President: Mamoru Kadokura) has entered into an investment and strategic agreement with an undisclosed US-based medical device company (“Company”). According to this agreement, Kaneka has made an investment in 18.5% shares of the Company and plans to start sales as the exclusive distributor […]
American College of Cardiology Acquires MedAxiom
WASHINGTON, Feb. 4, 2019 /PRNewswire/ — The American College of Cardiology Foundation has acquired MedAxiom, a cardiovascular membership, services and consulting firm, adding the company’s recognized leadership in cardiovascular operations, performance improvement, and business acumen to ACC’s world class science, education, registry, quality improvement and advocacy work. Through this acquisition, the ACC will expand […]
Teleflex Announces U.S. FDA Premarket Approval of MANTA™ Vascular Closure Device
WAYNE, Pa.–(BUSINESS WIRE)–Teleflex Incorporated (NYSE: TFX), a leading global provider of medical technologies for critical care and surgery, has announced that it received premarket approval (PMA) from the U.S. Food and Drug Administration for the MANTA™ Vascular Closure Device – the first commercially available biomechanical vascular closure device designed specifically […]
BD Announces Results for 2019 First Fiscal Quarter; Reaffirms Fiscal 2019 Guidance
FRANKLIN LAKES, N.J., Feb. 5, 2019 /PRNewswire/ — BD (Becton, Dickinson and Company) (NYSE: BDX), a leading global medical technology company, today reported quarterly revenues of $4.160 billion for the first fiscal quarter ended December 31, 2018. This represents an increase of 35.1 percent from the prior-year period, which is primarily due to the acquisition of […]
AtriCure to Announce Fourth Quarter and Full Year 2018 Financial Results
MASON, Ohio–(BUSINESS WIRE)–AtriCure, Inc. (Nasdaq: ATRC), a leading innovator in treatments for atrial fibrillation (Afib) and left atrial appendage (LAA) management, today announced that it will release its fourth quarter and full year 2018 financial results on Thursday, February 28, 2019. AtriCure will host a conference call at 4:30 p.m. Eastern […]
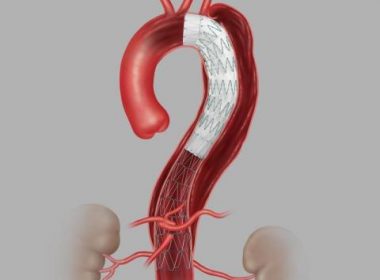
Cook Medical Receives U.S. FDA Approval for Aortic Dissection Device
BLOOMINGTON, Ind.–(BUSINESS WIRE)–Today, Cook Medical announced its recent approval from the U.S. FDA for its Zenith Dissection Endovascular System. The system, consisting of a proximal stent-graft component and a distal bare stent component, provides physicians a less invasive alternative to open surgery for repair of Type B dissections of the […]
First Atrial Fibrillation Patient Treated In Biosense Webster U.S. IDE Study Evaluating High Power, Short Duration Ablation Catheter
IRVINE, Calif., Feb. 4, 2019 /PRNewswire/ — Johnson & Johnson Medical Devices Companies* announced today that Biosense Webster, Inc., a worldwide leader in the diagnosis and treatment of heart arrhythmias, has enrolled and treated the first patient in its U.S. Investigational Device Exemption (IDE) study** which evaluates the company’s QDOT MICRO Radiofrequency […]