SURAT, India, Oct. 9, 2018 /PRNewswire/ — Concept Medical Inc. Raises $60mn For Investigational Device Exemption (IDE) For The World’s First Sirolimus-Coated Balloon. Concept Medical Inc. (CMI) has approached the FDA for an Investigational Device Exemption (IDE) for their Sirolimus-Coated Balloon (DCB). To support this process, they have raised $60mn (for an undisclosed valuation) from cardiologist and […]
Other News
Neovasc Reducer™ Granted Breakthrough Device Designation from FDA
VANCOUVER, Oct. 10, 2018 /PRNewswire/ – Neovasc Inc. (“Neovasc” or the “Company”) (NASDAQ: NVCN) (TSX: NVCN), a leader in the development of minimally invasive transcatheter mitral valve replacement technologies and in the development of minimally invasive devices for the treatment of refractory angina, today announced that the U.S. Food and Drug Administration (the “FDA”) […]
Cagent Vascular Appoints Medical Device Industry Veteran Brian Walsh as Chairman of the Board
WAYNE, Pa.–(BUSINESS WIRE)–Cagent Vascular®, a developer of next generation angioplasty balloons using proprietary serration technology, announces Brian Walsh to serve as Chairman of the Board. Brian Walsh has held several executive level positions in cardiovascular and ophthalmology specialties within the medical device industry. Mr. Walsh currently serves as the President […]
Hemostemix Announces Three Additional Sites Enrolled in Phase II Clinical Trial
CALGARY, Alberta, Oct. 09, 2018 (GLOBE NEWSWIRE) — Hemostemix Inc. (“Hemostemix” or the “Company”) (TSX VENTURE: HEM) is pleased to announce that it has received all approvals from three new medical centers to be clinical trial sites for the Company’s Phase II clinical trial for critical limb ischemia (“CLI”). In […]
Orchestra BioMed™ Adds Three Industry Leaders to Strategic Advisory Board
NEW HOPE, Pa., Oct. 09, 2018 (GLOBE NEWSWIRE) — Orchestra BioMed, Inc. (“Orchestra BioMed” or the “Company”), a biomedical innovation company developing transformational therapeutic devices targeting major medical conditions, announced today the appointment of three new members to its Strategic Advisory Board (“SAB”). Joining the Company’s SAB is Gary J. Pruden, former […]
Bellerophon Provides Update on INOpulse® Phase 2b Clinical Program for Treatment of Pulmonary Hypertension Associated with Interstitial Lung Disease
WARREN, N.J., Oct. 09, 2018 (GLOBE NEWSWIRE) — Bellerophon Therapeutics, Inc. (Nasdaq: BLPH) (“Bellerophon” or the “Company”), a clinical-stage biotherapeutics company, today provided an update on the Company’s ongoing Phase 2b randomized, double-blind, placebo-controlled clinical study (iNO-PF) evaluating INOpulse® in patients with Pulmonary Hypertension associated with Interstitial Lung Disease (PH-ILD), as […]
Hancock Jaffe Enters into Sponsored Research Agreement for CoreoGraft with World-Renowned Texas Heart Institute
IRVINE, Calif., Oct. 09, 2018 (GLOBE NEWSWIRE) — Hancock Jaffe Laboratories, Inc. (Nasdaq: HJLI, HJLIW), a company specializing in medical devices that restore cardiac and vascular health, announced today that it has entered into a Sponsored Research Agreement with Texas Heart Institute for the development of HJLI’s CoreoGraft®, an off […]
Espero BioPharma Appoints Lee Golden, M.D., as Chief Medical Officer
JACKSONVILLE, Fla. and IRVINE, Calif., Oct. 09, 2018 (GLOBE NEWSWIRE) — Espero BioPharma, Inc., (“Espero”, or “the Company”), a pharmaceutical company focused on the development of late-stage drugs to treat forms of cardiovascular disease, today announced the appointment of Lee Golden, M.D. as Chief Medical Officer. In his role as […]
Edwards Announces Start Of U.S. Study Of Self-Expanding Transcatheter Heart Valve
IRVINE, Calif., Oct. 8, 2018 /PRNewswire/ — Edwards Lifesciences Corporation (NYSE: EW), the global leader in patient-focused innovations for structural heart disease and critical care monitoring, today announced that it has commenced the U.S. pivotal trial that will study its self-expanding CENTERA transcatheter valve for severe, symptomatic aortic stenosis (AS) patients at intermediate […]
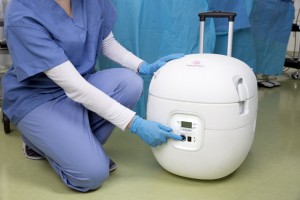
Paragonix Technologies Inc., Announces Presentation of the Paragonix SherpaPak™ Cardiac Transport System at Medline Booths 45 and 46 at the 2018 AATB Annual Meeting held from 9 to 12 October 2018
BRAINTREE, Mass.–(BUSINESS WIRE)–Paragonix Technologies, Inc. will present the Paragonix SherpaPak™ Cardiac Transport System product line at Medline Industries, Inc. Booths 45 and 46 during the 2018 American Association of Tissue Banks (AATB) Annual Meeting held in Dallas, TX, from 9 to 12 October, 2018. The Paragonix SherpaPak™ CTS1,2 is intended for […]