BURLINGTON, Mass., Sept. 07, 2018 (GLOBE NEWSWIRE) — LeMaitre Vascular, Inc. (Nasdaq:LMAT) announced today that it will be presenting at four conferences in September. David Roberts, President, will participate in the Lake Street Best Ideas Growth Conference on Thursday, September 13, 2018 at the Parker Hotel in New York City. […]
Other News
New Report from Vizient Evaluates “MRI-compatible” Cardiac Rhythm Devices
IRVING, Texas–(BUSINESS WIRE)–Vizient, Inc. today released its report “The Beat Goes On: Weighing Clinical Benefits, Patient Selection and Healthcare Economics of MRI-Compatible Cardiac Rhythm Devices”, which discusses the financial impact to hospitals resulting from the sudden and dramatic increase in the use of MRI-compatible CRM devices over the last 18 months. Magnetic resonance imaging (MRI)-compatible […]
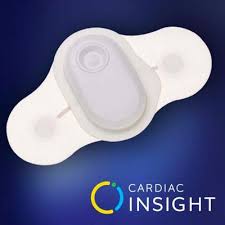
Cardiac Insight’s Wearable ECG Sensor, Cardea SOLO™, Continues to Attract Premier Cardiology Groups Across the U.S.; Announces Baltimore Heart Associates and Regional Cardiac Arrhythmia as Customers
KIRKLAND, Wash.–(BUSINESS WIRE)–Cardiac Insight, Inc., a leading U.S. developer of wearable biosensors and diagnostic software systems featuring proprietary automated data analysis algorithms, announced today that its wearable ECG Sensor, Cardea SOLO, continues to attract high-volume cardiology group practices across the U.S. The company unveiled Baltimore Heart Associates of Maryland (www.baltimoreheart.com) […]
InnovHeart Elects Keith D. Dawkins MD as Chairman of the Board
MILAN, Italy, Sept. 6, 2018 /PRNewswire/ — InnovHeart s.r.l., a privately-held, venture-backed developer of transcatheter-delivered mitral valve replacement systems (TMVR) for the treatment of mitral valve disease, announced today that it has appointed veteran medical device executive Keith D. Dawkins, M.D., as Chairman of its Board of Directors. Dr. Dawkins is transitioning from his […]
Leviticus Cardio Announces Successful Completion of Third Chronic Animal Study
PETAH TIKVA, Israel, Sept. 6 2018 /PRNewswire/ — Leviticus Cardio, inventors of the versatile transcutaneous Coplanar Energy Transfer (CET) system for use with implanted left ventricular assist devices (LVADs), announces the successful completion of a 90-day preclinical chronic animal study to evaluate its CET technology in combination with a commercial heart pump. The […]
Gore Completes Patient Enrollment in U.S. Pivotal Clinical Study of GORE® CARDIOFORM ASD Occluder
FLAGSTAFF, Ariz.–(BUSINESS WIRE)–W. L. Gore & Associates, Inc. (Gore) has completed enrollment for the pivotal phase of the Gore ASSURED Clinical Study. This investigational device exemption (IDE) trial is researching the new GORE®CARDIOFORM ASD Occluder for the interventional closure of Atrial Septal Defects (ASDs), sized 8 to 35 mm. The […]
ULTRACONNECT from A&D Medical: The Power of Connected Health is Now Available
SAN JOSE, Calif., Sept. 5, 2018 /PRNewswire/ — A&D Medical, a global leader in connected health and biometric measurement devices and services, today announced its line of ULTRACONNECT blood pressure monitors is available nationwide, giving patients who are managing hypertension a new tool to better manage their heart health – with just a twist […]
Biotricity Developing ECG Patch for Anticipated Q1 2019 Release
REDWOOD CITY, Calif., Sept. 05, 2018 (GLOBE NEWSWIRE) — Biotricity Inc. (OTCQB: BTCY), a medical diagnostic and consumer healthcare technology company, today announced that it is developing “Biopatch,” an ECG patch which it hopes to release in Q1 2019. An extension of the company’s award-winning Bioflux device, Biopatch offers an alternative to the 3-lead system […]
SentreHEART Announces 400th Patient Enrollment in the Landmark aMAZE Trial
REDWOOD CITY, Calif.–(BUSINESS WIRE)–SentreHEART, Inc., manufacturer of the LARIAT® Suture Delivery Device (LARIAT) for percutaneous left atrial appendage (LAA) closure announces enrollment of the 400th patient in the prospective, multi-center, randomized controlled Trial known as the aMAZE Trial. The Trial is a superiority design and intends to demonstrate the LARIAT procedure […]
Vascular Dynamics Announces Initial Enrollment in Rigorously Designed Hypertension Trial
MOUNTAIN VIEW, Calif.–(BUSINESS WIRE)–Vascular Dynamics, Inc. (VDI), a privately held medical device company focused on innovative, minimally invasive device-based solutions for cardiovascular conditions, today announced enrollment of the first patients in an FDA-approved best-in-class pivotal clinical trial. The CALM-2 study is designed to establish safety and efficacy of the novel […]