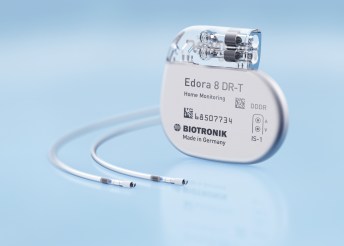
LAKE OSWEGO, Ore., June 7, 2017 /PRNewswire/ — BIOTRONIK today announced the availability of the Edora line of devices, the company’s first available pacemaker series featuring BIOTRONIK’s MRI AutoDetect technology. Edora SR-T is the smallest MR conditional pacemaker with automated MRI detection capability available in the US with a volume of […]
Other News
Aegis Medical Innovations Announces FDA Approval of Clinical Trial
VANCOUVER, British Columbia–(BUSINESS WIRE)–Aegis Medical Innovations Inc. (Aegis) today announced that it has received Investigational Device Exemption approval from the U.S. FDA to initiate a clinical trial in the U.S. for its medical device called the Sierra Ligation System (Sierra). Aegis developed the Sierra technology in partnership with the Mayo […]
Biosense Webster Launches CARTO® 3 System CARTO VISITAG™ Module with Ablation Index, designed to help Electrophysiologists standardize and simplify the treatment of Atrial Fibrillation
DIAMOND BAR, California, March 22, 2017 /PRNewswire/ — Biosense Webster, a Division of Johnson & Johnson Medical NV/SA, a worldwide leader in the diagnosis and treatment of cardiac arrhythmias, announced today the launch of the CARTO VISITAG™ Module with Ablation Index, a new technology providing visual indication based on the […]
New Meta-Analysis Presented at American College of Cardiology Strongly Supports RenalGuard(R) Use in Cardiovascular Interventional Procedures
MILFORD, MA — (Marketwired) — 03/22/17 — A new meta-analysis presented this week at the American College of Cardiology further supports the ability of RenalGuard Therapy® to significantly reduce the incidence of acute kidney injury associated with cardiovascular interventional procedures compared to other methods of urine volume expansion. Moreover, findings […]
Arrhythmia Research Technology, Inc. Reports 2016 Fourth Quarter and Year End Results
FITCHBURG, Mass., March 22, 2017 (GLOBE NEWSWIRE) — Arrhythmia Research Technology, Inc. (NYSE MKT:HRT) (the “Company”), through its wholly-owned subsidiary, Micron Products, Inc., a diversified contract manufacturing organization that produces highly-engineered, innovative medical device components requiring precision machining and injection molding, announced results for its fourth quarter and year ended […]
Intact Vascular Announces Completion of Enrollment in the Tack Optimized Balloon Angioplasty II (TOBA II) Clinical Trial
WAYNE, Pa.–(BUSINESS WIRE)– Intact Vascular, Inc., a developer of medical devices for minimally invasive peripheral vascular procedures, today announced that its Tack Optimized Balloon Angioplasty II (TOBA II) clinical trial has completed enrollment. A total of 210 patients have been enrolled in TOBA II at 33 sites in the U.S. […]
Texas Cardiac Arrhythmia Institute at St. David’s Medical Center First in Texas and Among First in U.S. to Use FlexAbility™ Ablation Catheter, Sensor Enabled™
AUSTIN, Texas, March 21, 2017 /PRNewswire/ — On March 1, 2017, the Texas Cardiac Arrhythmia Institute (TCAI) at St. David’s Medical Center was the first in Texas and among the first facilities in the U.S. to use the FlexAbility™ Ablation Catheter, Sensor Enabled™, designed to improve versatility and precision during […]
Tryton Medical Announces First U.S. Commercial Case with Tryton Side Branch Stent
DURHAM, N.C.–(BUSINESS WIRE)– Tryton Medical, Inc., the leading developer of stents designed to treat coronary bifurcation lesions, today announced that the first U.S. commercial case using the Tryton Side Branch Stent to treat a coronary bifurcation lesion involving a large side branch (appropriate for a ≥2.5mm stent) was completed at […]
QT Vascular Scores $14.3 Million Capital
QT Vascular Secures Up To S$20 Million Capital Commitment GEM Global SINGAPORE, March 21, 2017 /PRNewswire/ — QT Vascular Ltd., (the “Company” or “QT Vascular“, and together with its subsidiaries, the “Group“) announced that it has signed an agreement (“Agreement“) with GEM Global Yield Fund LLC SCS (“GEM Global“), the New York based […]
BioStable Science & Engineering Wins FDA Nod for Its HAART 300 Aortic Annuloplasty Device
BioStable Science & Engineering Announces FDA Clearance of the HAART™ 300 Aortic Annuloplasty Device AUSTIN, Texas–(BUSINESS WIRE)–BioStable Science & Engineering, Inc. announced today it has received FDA market clearance for the HAART 300 Aortic Annuloplasty Device, the first commercially available internal annuloplasty device designed for aortic valve repair. BioStable expects […]