WINNIPEG, April 24, 2018 /PRNewswire/ – Medicure Inc. (“Medicure”) (TSXV:MPH, OTC:MCUJF), a cardiovascular pharmaceutical company, will release financial results for the year ended December 31, 2017 after market close on Monday, April 30, 2018. Medicure will hold a conference call and webcast regarding the results on Tuesday, May 1, 2018at 7:30 AM Central Time (8:30 AM Eastern Time). […]
Other News
BD Board Declares Dividends
FRANKLIN LAKES, N.J., April 23, 2018 /PRNewswire/ — The Board of Directors of BD (Becton, Dickinson and Company) (NYSE: BDX) has declared a quarterly dividend of $0.75 per common share, payable on June 29, 2018 to holders of record on June 8, 2018. The indicated annual dividend rate is $3.00 per share. In addition, the Board of Directors has declared […]
Bardy Diagnostics™ Announces Appointment of Alan Levy to Board of Directors
SEATTLE, April 24, 2018 /PRNewswire/ — Bardy Diagnostics, Inc., (“BardyDx”), a leading provider of ambulatory cardiac monitoring technologies and custom data solutions, including the Carnation Ambulatory Monitor (“CAM™”), the world’s only P-wave centric™ ambulatory cardiac monitor and arrhythmia detection device, announced today the appointment of Alan J. Levy, PhD to its Board of […]
Arkis BioSciences® Brings Endexo® Technology To Neurosurgical Use With Its New CerebroFlo™ EVD Catheter
KNOXVILLE, Tenn., April 24, 2018 /PRNewswire/ — Arkis BioSciences®, a leading innovator of next generation neurosurgical catheters and minimally invasive instrumentation, announced today that it is receiving promising clinical feedback for its new CerebroFlo™ External Ventricular Drainage (EVD) brain catheter. The company introduced the new catheter, incorporating the Endexo® molecule, into clinical use for […]
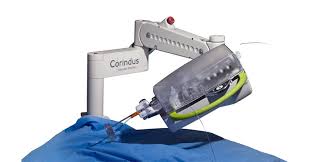
Corindus Presents “Optimizing Care with CorPath GRX: Hot Topics in Robotic Interventions” at SCAI 2018 Dinner Symposium on April 26
WALTHAM, Mass.–(BUSINESS WIRE)–Corindus Vascular Robotics, Inc. (NYSE American: CVRS), a leading developer of precision vascular robotics, announced today that it will sponsor a dinner event during the Society for Cardiovascular Angiography and Interventions (SCAI) 2018 Scientific Sessions at the Manchester Grand Hyatt in San Diego, California. Vascular robotics will be […]
Surmodics Announces FDA Clearance of a New .018” Low-Profile PTA Balloon Dilation Catheter
EDEN PRAIRIE, Minn.–(BUSINESS WIRE)–Surmodics, Inc. (NASDAQ: SRDX), a leading provider of medical device and in vitro diagnostic technologies to the healthcare industry, announced it received U.S. Food and Drug Administration (FDA) 510(k) clearance for its .018” Low-Profile percutaneous transluminal angioplasty (PTA) balloon dilation catheter, designed and indicated for a broad […]
The global microelectronic medical implants market size is anticipated to reach USD 57.12 billion by 2025
LONDON, April 23, 2018 /PRNewswire/ — Microelectronic Medical Implants Market Analysis Report by Product (Pacemakers & Defibrillators, Neurostimulators, Implantable Drug Pumps, Spinal Fusion Stimulators), by Technology, And Segment Forecasts, 2018 – 2025 Download the full report: https://www.reportbuyer.com/product/5360535 The global microelectronic medical implants market size is anticipated to reach USD 57.12 billion by 2025, according to […]
VGS Initiates U.S. Pivotal Trial to Evaluate the Use of Its VEST Technology for Bypass Surgery
TEL AVIV, Israel, April 23, 2018 /PRNewswire/ — Vascular Graft Solutions Ltd (VGS) announces on enrollment of the first 20 patients into the VEST US pivotal trial that will evaluate the safety and effectiveness of the VEST, a novel external support device for treatment of saphenous vein graft disease after coronary artery […]
ARCA biopharma and Medtronic to Collaborate on Atrial Fibrillation Clinical Trial for GencaroTM
BROOMFIELD, Colo.–(BUSINESS WIRE)–ARCA biopharma, Inc. (Nasdaq: ABIO), a biopharmaceutical company developing genetically-targeted therapies for cardiovascular diseases, today announced that it has entered into an agreement with Medtronic, Inc. (NYSE:MDT), a leader in medical technologies to improve the treatment of chronic diseases, including cardiac rhythm disorders, to collaborate on ARCA’s proposed […]
Medtronic IN.PACT(TM) Admiral(TM) Drug Coated Balloon Receives FDA Approval to Treat Long SFA Lesions
DUBLIN – April 23, 2018 – Medtronic plc (NYSE: MDT) today announced that it has received U.S. Food and Drug Administration (FDA) approval for the IN.PACT(TM) Admiral(TM) Drug-Coated Balloon (DCB) to treat long superficial femoral artery (SFA) lesions up to 360mm in patients with peripheral artery disease (PAD). Approval was based […]