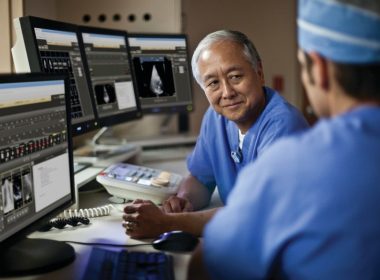
Comprehensive informatics and imaging solution features enhanced security protocols to meet industry-leading standards Improved interoperability and standardization increases scalability across the hospital enterprise to help improve efficiency and quality AMSTERDAM and LAS VEGAS, March 5, 2018 /PRNewswire/ — Royal Philips (NYSE: PHG, AEX: PHIA), a global leader in health technology, today announced the launch of IntelliSpace Cardiovascular 3.1, […]
Other News
Corindus Receives FDA Clearance for First Automated Robotic Movement in technIQ™ Series for CorPath GRX Platform
WALTHAM, Mass.–(BUSINESS WIRE)– Corindus Vascular Robotics Inc. (NYSE American: CVRS), a leading developer of precision vascular robotics, announced today that it received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for the first automated robotic movement designed for the CorPath GRX platform. The proprietary software feature, named “Rotate on Retract” […]
Nihon Kohden Launches Aireeg® WEE-1200 Wireless EEG System at HIMSS18
IRVINE, Calif.–(BUSINESS WIRE)– Nihon Kohden Corporation, a U.S. market leader in precision medical products and services, has launched its aireeg®WEE-1200 wireless electroencephalogram (EEG) system designed specifically for patient comfort and easy access to real-time patient data during long-term epilepsy monitoring, intensive care, and routine EEG environments. The smart, safe and secure […]
Avinger Regains Compliance with Nasdaq Listing Requirements
REDWOOD CITY, Calif., March 02, 2018 (GLOBE NEWSWIRE) — Avinger, Inc. (Nasdaq:AVGR), a leading developer of innovative treatments for peripheral artery disease (PAD), today announced it has regained compliance with Nasdaq listing requirements. In a letter dated March 1, 2018, Nasdaq informed the Company that it has achieved compliance with […]
Jason Field to Succeed Terri Kelly as President and CEO of W. L. Gore & Associates
NEWARK, Del.–(BUSINESS WIRE)–W. L. Gore & Associates today announced that Jason Field, global sales and marketing leader for Gore Medical Products Division, will succeed Terri Kelly in the role of President and CEO effective April 1, 2018. Terri Kelly will continue to serve on Gore’s Board of Directors through July 2018. […]
Cardiovascular Systems, Inc. and OrbusNeich Announce FDA Clearance of the First and Only 1.0mm Coronary Balloon in the U.S.
ST. PAUL, Minn.–(BUSINESS WIRE)–Cardiovascular Systems, Inc. (CSI®) (NASDAQ: CSII), a medical device company developing and commercializing innovative interventional treatment systems for patients with peripheral and coronary artery disease, today announced that the U.S. Food and Drug Administration (FDA) has granted 510(k) clearance for the OrbusNeich® 1.0mm Sapphire® II PRO coronary balloon. OrbusNeich’s […]
Safety and Effectiveness of BioTrace Medical’s Tempo® Lead Further Demonstrated in Multi-Center Analysis of More Than 200 Cardiac Procedures
MENLO PARK, Calif. & ORLANDO, Fla.–(BUSINESS WIRE)–BioTrace Medical, Inc., the leader in innovative temporary pacing technology, today announced positive safety and effectiveness data from a physician-sponsored U.S. study of the company’s Tempo® Temporary Pacing Lead. Results from a multi-center retrospective analysis of 224 U.S. interventional cardiac procedures, including transcatheter aortic valve […]
Results of Landmark Wearable Defibrillator Trial to Be Presented as ACC.18 Late-Breaker
CHELMSFORD, Mass.–(BUSINESS WIRE)–ZOLL® Medical Corporation, an Asahi Kasei Group Company that manufactures medical devices and related software solutions, announced today that findings from the “Vest Prevention of Early Sudden Death Trial” (VEST Trial) on use of the ZOLL LifeVest® wearable cardioverter defibrillator (WCD) will be presented as a Late-Breaking Clinical Trial at the American […]
4C Medical’s Novel Mitral Regurgitation Therapy to be Highlighted at CRT 2018 Meeting
BROOKLYN PARK, Minn., March 1, 2018 /PRNewswire/ — 4C Medical Technologies, Inc. (4C Medical), a developer of minimally invasive technologies for structural heart disease, today announced that its medical device for mitral regurgitation (MR) will be highlighted in two presentations at the Cardiovascular Research Technologies (CRT) meeting held March 3-6, 2018 in Washington, DC. 4C Medical […]
New Biocompatible Embolic Device to Control Blood Flow & Treat Tumors Presented at the Leipzig Interventional Congress (LINC)
GPX is a biocompatible gel-particle embolic device that transforms from low-viscosity particles in a syringe to a solid particle after injection into a vessel LEIPZIG, GERMANY, February 01, 2018 /24-7PressRelease/ — Two scientific abstracts of a new vascular embolic device were presented this week at the 2018 Leipzig Interventional Congress […]