Study to Enroll Subjects at up to 35 Sites Across the U.S. and Europe DUBLIN – January 26, 2018 – Medtronic plc (NYSE: MDT) today announced the initiation of its investigational device exemption (IDE) study for the Abre(TM) venous self-expanding stent system. The ABRE IDE Study will evaluate the safety and […]
Other News
CORRECTING and REPLACING First Patient Enrolled in Tryton Post Approval Study
DURHAM, N.C.–(BUSINESS WIRE)–Please replace the release dated January 24, 2018, with the following corrected version due to multiple revisions. The corrected release reads: FIRST PATIENT ENROLLED IN TRYTON POST APPROVAL STUDY The only FDA approved device to treat coronary bifurcations Tryton Medical, Inc., the leading developer of stents designed to […]
Vascular Insights® Announces New Chief Executive Officer
QUINCY, Mass., Jan. 25, 2018 /PRNewswire/ — Vascular Insights® announced that its Board of Directors has appointed James P. Ott as President and CEO. Jim brings to the position greater than 20 years of experience as President for companies in the manufacturing and medical device industries. He has held numerous leadership positions that have focused on […]
Stryker’s WEAVE Trial Demonstrates Positive Results of Endovascular Treatment for Patients With Intracranial Atherosclerotic Disease
KALAMAZOO, Michigan, USA, Jan. 26, 2018 /PRNewswire/ — Results from the Wingspan® StEnt System Post MArket SurVEillance Study (WEAVE™ Trial) were presented yesterday at the International Stroke Conference, providing compelling evidence that more patients suffering from intracranial atherosclerotic disease (ICAD) may benefit from endovascular treatment with the Wingspan Stent System. Results from […]
INDEPENDENT STUDY REAFFIRMS PENUMBRA ASPIRATION AS FRONTLINE THROMBECTOMY APPROACH FOR ACUTE ISCHEMIC STROKE
Results of COMPASS Trial Presented at International Stroke Conference LOS ANGELES – Jan. 25, 2018 – Penumbra, Inc. (NYSE: PEN), a global healthcare company focused on innovative therapies, today announced the presentation of the results of the COMPASS Trial, an independent, prospective, multi-center randomized trial that showed the use of […]
NEW STUDY SHOWS USE OF EMBOTRAP® DEVICE CLOT RETRIEVER DURING TREATMENT OF STROKE YIELDS POSITIVE RATES OF REVASCULARIZATION
ARISE II Clinical Trial Results Presented at International Stroke Conference LOS ANGELES – Jan. 25, 2018 – EMBOTRAP® Device, a device designed to remove clots from the brain following an ischemic stroke, demonstrated high success rates in restoring blood flow and patients achieved high rates of functional independence, according to […]
Endologix, Inc. to Announce Fourth Quarter and Fiscal Year 2017 Financial Results on February 21, 2018
IRVINE, Calif.–(BUSINESS WIRE)– Endologix Inc. (Nasdaq: ELGX), a developer and marketer of innovative treatments for aortic disorders, today announced that it will report financial results for the fourth quarter and fiscal year 2017 after the market close on Wednesday, February 21, 2018. The Company’s management will host a conference call at 4:30 […]
Stryker’s Wingspan Stent Impresses in Post-Market Study
KALAMAZOO, Michigan, USA, Jan. 25, 2018 /PRNewswire/ — Yesterday the American Heart Association and American Stroke Association updated the ischemic stroke guidelines. The new recommendation expanded the treatment window from six to 16 hours based on overwhelming clinical evidence from the Stryker-sponsored DAWN Trial and confirmed by DEFUSE 3. The guidelines state that […]
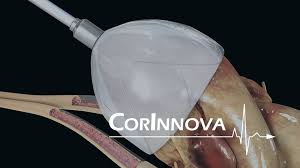
CorInnova Selected to Present at “STS/AATS Tech-Con 2018” – Sixth Honor in Five Months
HOUSTON–(BUSINESS WIRE)– CorInnova, an emerging medical device company developing breakthrough technology for the treatment of Heart Failure, announced today that it has been selected to present at the Society for Thoracic Surgeons (STS) / American Association for Thoracic Surgery (AATS) Tech-Con 2018 event to be held this coming Saturday, Jan. 27, […]
Advanced Bifurcation Systems Appoints Industry Veteran Thomas O’Neill to Board of Directors
LOS ANGELES, Jan. 25, 2018 (GLOBE NEWSWIRE) — Advanced Bifurcation Systems (“ABS” or the “Company”), a clinical stage medical device company developing an innovative stenting platform which overcomes the limitations of current approaches for the treatment of bifurcation lesions in coronary angioplasties, today announced the addition of Thomas O’Neill to the Board […]