ST. PAUL, Minn.–(BUSINESS WIRE)– Cardiovascular Systems, Inc. (CSI) (NASDAQ: CSII), a medical device company developing and commercializing innovative interventional treatment systems for patients with peripheral and coronary artery disease, today announced two new partnerships broadening the company’s product portfolio. CSI is now the exclusive U.S. distributor of OrbusNeich balloon products. Additionally, the […]
Other News
BD Announces Completion of Enrollment in the Lutonix 014 Drug Coated Balloon Below-the-Knee Trial
FRANKLIN LAKES, N.J., Jan. 18, 2018 /PRNewswire/ — Becton, Dickinson and Company (NYSE: BDX), a leading global medical technology company, today announced the completion of enrollment in the Lutonix below-the-knee (BTK) trial and plans to submit a pre-market approval application in late 2018. The Lutonix BTK trial is a prospective, multicenter, randomized, […]
Bluegrass Vascular Announces Enrollment of First Patients in U.S. Pivotal Trial
SAN ANTONIO, Jan. 18, 2018 /PRNewswire/ — Bluegrass Vascular Technologies, a private medical technology company focused on innovating lifesaving devices and methods for vascular access, today announced the enrollment of its first patients in the SAVE-US (Surfacer® System to Facilitate Access in VEnous Occlusions – United States) pivotal trial. The SAVE-US clinical trial is a pre-market […]
Neovasc Reducer Featured in Live Case at Berlin Symposium
VANCOUVER, Jan. 18, 2018 /PRNewswire/ – Neovasc Inc. today reported its Neovasc Reducer™ (“Reducer”) was featured in a “live case” broadcast to more than 800 participants at the Kardiologie Symposium 2018 held in Berlin, Germany. The successful live case was performed by Dr. Spyrantis and Professor Banai in the Sana-Klinikum Lichtenberg. “The […]
FDA adds safety alert for Zoll LifeVest
Patient death prompts safety warning by FDA. Zoll LifeVest 4000 Wearable Cardioverter Defibrillator: FDA Safety Communication – Potential Lack of Treatment (Shock) Delivery Due to Device Failure by FDA.gov ISSUE: FDA is providing information and recommendations regarding the Zoll LifeVest 4000 due to concerns that the device may fail to […]
PENUMBRA ANNOUNCES KEY EVENTS AT INTERNATIONAL STROKE CONFERENCE 2018 MEETING
ALAMEDA, Calif. – January 17, 2018 – Penumbra, Inc. (NYSE: PEN), a global healthcare company focused on innovative therapies, today announced key events at the upcoming International Stroke Conference 2018 meeting (ISC 2018) to be held January 24-26 at the Los Angeles Convention Center. These events include results from two […]
BIOLIFE4D Targets High Growth Healthcare 3D Printing Market as it Readies for Regulation A+ Offering
CHICAGO, Jan. 17, 2018 (GLOBE NEWSWIRE) — BIOLIFE4D, a biotech pioneer leveraging advances in tissue engineering to 3D print human organs viable for transplant, today commented on the high growth healthcare 3D printing market as the Company readies for its Regulation A+ equity crowdfunding offering. A recent report projects the global market to […]
International Biophysics Wins FDA Nod for the FloPump 32 Centrifugal Heart Pump
AUSTIN, Texas, Jan. 17, 2018 /PRNewswire/ — International Biophysics Corporation, a global medical device manufacturer based in Austin, Texas, announced they have received FDA A 510k clearance for their FloPump 32 centrifugal disposable heart pump. The FloPump 32 is one of the smallest centrifugal pumps in the world at 32ml prime. The FloPump […]
Secant Group Develops the First Synthetic Regenerative Cardiovascular Graft
TELFORD, Pa.–(BUSINESS WIRE)–Secant Group, in partnership with its sister company SanaVita Medical, announce game changing technology to advance cardiovascular regenerative medicine with the development of a synthetic, small bore, vessel that encourages endogenous regeneration and new vessel formation. The technology is based on the company’s sophisticated textile forming capabilities that […]
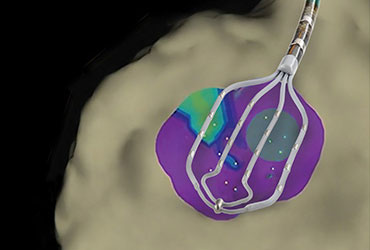
Abbott announces European launch of Advisor HD Grid Mapping Catheter, Sensor Enabled
ABBOTT PARK, Ill., Jan. 11, 2018 – Abbott today announced CE Mark approval for the company’s new Advisor™ HD Grid Mapping Catheter, Sensor Enabled™, a product designed to advance cardiac mapping during cardiac ablation to treat patients with complex cardiac arrhythmias. With the European launch of this latest addition to […]