TEL AVIV, lSRAEL, Dec. 04, 2017 (GLOBE NEWSWIRE) — InspireMD, Inc. (NYSE AMER:NSPR), a leader in embolic prevention systems (EPS) / thrombus management technologies and neurovascular devices, today announced it has signed Diverse Devices Pty Ltd as its exclusive distributor for Australia and New Zealand, and has signed Do Gia Production […]
Other News
CorInnova Awarded “2017 InnoSTARS” Prize for Its Non-Blood-Contacting, Soft Robotic Heart Assist Device as Alternative to LVADs for Treatment of Heart Failure
By Ronald Trahan, APR, Ronald Trahan Associates, 1-508-816-6730, rtrahan@ronaldtrahan.com HOUSTON, Dec. 4, 2017—CorInnova Inc., an emerging medical device company developing novel technology for the treatment of Heart Failure, announced today that it was awarded second prize in the “2017 InnoSTARS” life science competition for its EpicHeart™ soft-robotic heart assist device. […]
HeartFlow in rare Unicorn territory with $150 Million raise
HeartFlow is the newest unicorn after a new $150 million financing round The medical company is now valued at $1.4 billion. BY THEODORE SCHLEIFER@TEDDYSCHLEIFER The medical technology company HeartFlow is now valued at over $1 billion, the latest company to hit the category threshold for the most highly valued startups. HeartFlow […]
FDA Approves Amgen’s Repatha® (evolocumab) To Prevent Heart Attack And Stroke
THOUSAND OAKS, Calif., Dec. 1, 2017 /PRNewswire/ — Amgen (NASDAQ: AMGN) today announced that following priority review of its supplemental Biologics License Application, the U.S. Food and Drug Administration (FDA) approved Repatha® (evolocumab) as the first PCSK9 inhibitor to prevent heart attacks, strokes and coronary revascularizations in adults with established cardiovascular disease.1 “We are pleased […]
Vascular Dynamics Appoints Ed Roschak CEO
MOUNTAIN VIEW, Calif., Dec. 04, 2017 (GLOBE NEWSWIRE) — Vascular Dynamics, Inc. (VDI), a privately held medical device company which has developed an innovative treatment system for the treatment of hypertension, today announced the appointment of Mr. Ed Roschak as its president and chief executive officer. “We are fortunate that […]
Corindus Partners With Houston Methodist DeBakey Heart & Vascular Center to Launch Premier Robotic Training Site for Physicians
WALTHAM, Mass.–(BUSINESS WIRE)– Corindus Vascular Robotics (NYSE American: CVRS), a leading developer of precision vascular robotics, announced today that Houston Methodist DeBakey Heart & Vascular Center, a leading cardiovascular center located in the Texas Medical Center, has launched the first vascular robotic program in the Houston area with the installation of two CorPath® GRX […]
CryoLife Completes Acquisition of JOTEC
ATLANTA, Dec. 1, 2017 /PRNewswire/ — CryoLife, Inc. (NYSE: CRY), a leading medical device and tissue processing company focused on cardiac and vascular surgery, announced today that it has completed its previously announced acquisition of JOTEC AG, a German-based, privately-held developer of technologically differentiated endovascular stent grafts, and cardiac and vascular surgical […]
BioTelemetry Enters Agreement to Provide Cardiac Monitoring for Apple’s Heart Study
MALVERN, Pa., Nov. 30, 2017 (GLOBE NEWSWIRE) — BioTelemetry(NASDAQ:BEAT), the leading mobile and wireless medical technology company focused on the delivery of health information to improve quality of life and reduce cost of care, announced today a partnership with Apple(NASDAQ:AAPL) to provide cardiac monitoring services in conjunction with the Apple Heart Study. […]
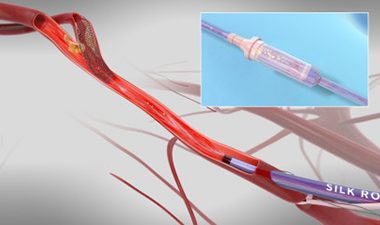
Updated TCAR Data From Silk Road Medical’s ROADSTER 1 and 2 Studies Presented at VEITHsymposium
SUNNYVALE, Calif., Nov. 29, 2017 /PRNewswire/ — Silk Road Medical, a company dedicated to preventing the devastating burden of stroke through surgical innovation, announced multiple presentations at the 45th annual VEITHsymposium highlighting the safety and efficacy of the company’s ENROUTE® Transcarotid Neuroprotection and Stent System, the first and only products specifically designed and […]
Cordis and Medinol Announce FDA Approval of the Innovative EluNIR Drug-Eluting Stent System
DUBLIN, Ohio, Nov. 30, 2017 /PRNewswire/ — Cordis, a Cardinal Health company, and Medinol today announced United States Food and Drug Administration (FDA) approval of the EluNIR™ drug-eluting stent (DES) for the treatment of patients with narrowing or blockages to their coronary arteries. The EluNIR™ stent system is designed with a novel metallic […]