ALISO VIEJO, Calif., Sept. 26, 2017 /PRNewswire/ — MicroVention, a wholly owned subsidiary of Terumo Corporation and a global neurovascular company, today announced that the Grand Opening of its New Innovation Center was held on Monday, September 25. The opening of the new facility, located in the heart of the Summit Office Campus, also […]
Other News
W. L. Gore & Associates Completes Patient Enrollment In U.S. Pivotal Clinical Study Of GORE CARDIOFORM ASD Occluder
FLAGSTAFF, Ariz.–(BUSINESS WIRE)–W. L. Gore & Associates, Inc. (Gore) has completed enrollment for the pivotal phase of the Gore ASSURED Clinical Study. This investigational device exemption (IDE) trial is researching the new GORE®CARDIOFORM ASD Occluder for the interventional closure of Atrial Septal Defects (ASDs), sized 8 to 35 mm. The […]
FDA Classifies Spectranetics’ Bridge Occlusion Balloon as Class I Recall
Bridge Occlusion Balloon Catheter Model 590-001 by Spectranetics: Class I Recall – Risk of Blocked Guidewire Lumen Preventing Balloon Utilization ISSUE: Spectranetics is recalling its Bridge Occlusion Balloon Catheter due to the possibility of a blocked guidewire lumen in some device units. If a device with a blocked guidewire lumen […]
FEops closes EUR 6 million Series B financing
GENT, Belgium, Sept. 22, 2017 — FEops, a leader in personalized computer modeling for structural heart interventions, announced it has secured a 6 million euros investment, led by Valiance, and joined by existing investors Capricorn and PMV. The funding will be used to aggressively implement the company’s product roadmap, centered […]
Aortica Reports Successful Results Of First 30 Fenestrated EVAR Patients Using Automated Case Planning Software
9/25/2017 8:56:46 AM BELLEVUE, Wash.–(BUSINESS WIRE)–Aortica Corp. today announced that Dr. Benjamin Starnes, Chief of Vascular Surgery at the University of Washington (UW), has reported successful results for the first 30 patients undergoing Fenestrated Endovascular Aneurysm Repair (FEVAR) planned and performed using Aortica’s AORTAFIT™ automated case planning software. The cases […]
Boston Scientific (BSX) Nabs FDA OK for MRI Labeling on High-Voltage Devices, U.S. Launch of Resonate Devices
MARLBOROUGH, Mass., Sept. 25, 2017 /PRNewswire/ — Boston Scientific (NYSE: BSX) has launched the Resonate family of implantable cardioverter defibrillator (ICD) and cardiac resynchronization therapy defibrillator (CRT-D) systems featuring the HeartLogic Heart Failure Diagnostic to help physicians improve heart failure (HF) management. The new devices, which are approved by the U.S. Food […]
FDA OKs Abbott (ABT)’ MRI-Compatibility for the Company’s Ellipse ICD
ABBOTT PARK, Ill., Sept. 22, 2017 /PRNewswire/ — Abbott (NYSE: ABT) today announced U.S. Food and Drug Administration (FDA) approval for magnetic resonance (MR)-conditional labeling for one of Abbott’s most widely-used implantable cardioverter defibrillators (ICD) and associated high voltage leads. The approval of MR-conditional labeling for the Ellipse ICD with the Tendril MRI […]
InspireMD Announces Distribution Agreement For CGuard EPS In Portugal
TEL AVIV, ISRAEL–(Marketwired – September 21, 2017) – InspireMD, Inc. (NYSE MKT: NSPR) (NYSE American: NSPR) (NYSE MKT: NSPR.WS) (NYSE American: NSPR.WS) (“InspireMD” or the “Company”), a leader in embolic prevention systems (EPS) / thrombus management technologies and neurovascular devices, today announced it has signed an agreement with Medicinália Cormédica MC Medical Lda, a […]
Medtronic (MDT) Announces Clinical Study To Evaluate The Corevalve Evolut PRO System In ‘Everyday’ Clinical Practice
DUBLIN – September 21, 2017 – Medtronic plc (NYSE: MDT) today announced a new post-market clinical study to evaluate its CoreValve(TM) Evolut(TM) PRO valve in everyday clinical practice. Studying patients with severe symptomatic aortic stenosis at an intermediate, high or extreme risk for open heart surgery, the FORWARD PRO Clinical Study […]
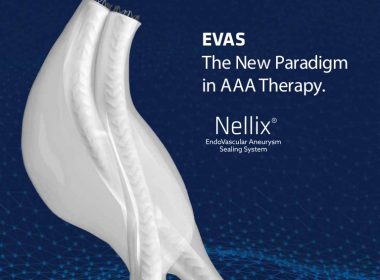
Endologix Announces CE Mark Approval For The Nellix Endovascular Aneurysm Sealing System With The Refined Indications For Use
9/21/2017 7:24:55 AM IRVINE, Calif.–(BUSINESS WIRE)–Endologix, Inc. (NASDAQ: ELGX), a developer and marketer of innovative treatments for aortic disorders, today announced the CE Mark approval for its Nellix® EndoVascular Aneurysm Sealing System (“Nellix”) with the refined Indications for Use (IFU). Nellix is being studied in the U.S. under an Investigational Device […]