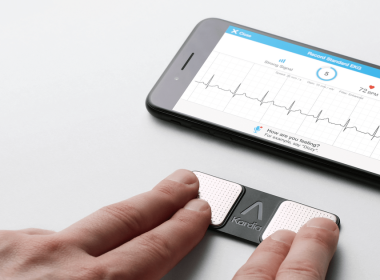
MOUNTAIN VIEW, Calif., Sept. 6, 2017 /PRNewswire/ — AliveCor, the leader in FDA-cleared personal electrocardiogram (EKG) technology, today announced the results of four clinical research presentations that demonstrate that AliveCor’s hyper-fast, 30 second, digital EKG can positively impact and potentially even save the lives of millions of people around the world who suffer […]
Other News
Cerebrotech Appoints Neurovascular Veteran Martin Dieck to Executive Chairman
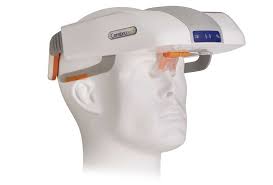
PLEASANTON, Calif.–(BUSINESS WIRE)–Cerebrotech Medical Systems, an innovative medical device company focused on the development of a portable brain bioimpedance monitor, today announced that board member Martin Dieck, an experienced entrepreneur with expertise in developing novel neurovascular solutions, will step up to executive chairman, effectively immediately. “I am thrilled to have […]
Cardiac Insight, Inc. Continues Steady Beat of Progress with New $4.5 Million Investment and Attracts Prestigious Healthcare M&A Executive, Clifford J. Stocks, to Its Board of Directors
KIRKLAND, Wash.–(BUSINESS WIRE)–Cardiac Insight, Inc., a U.S. developer of wearable medical devices and diagnostic software, announced today that the company continues to experience a steady beat of market traction and industry endorsement with a recent infusion of $4.5 million into their new C-1 round of funding, currently capped at $10 […]
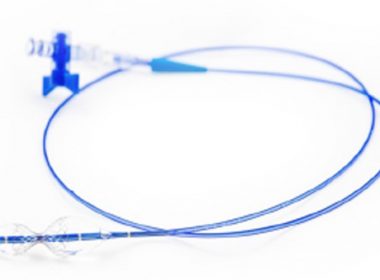
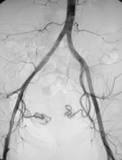
NaviGate Cardiac Structures, Inc. (“NCSI”) reports world’s first successful transcatheter implantation of 52-mm tricuspid valve into a transplanted-heart patient
LAKE FOREST, Calif., Aug. 30, 2017 – NaviGate Cardiac Structures Inc. (“NCSI”) announced today that its GATE™ catheter-guided tricuspid atrioventricular valved stent (A VS) was implanted through the jugular vein six weeks ago into a patient’s transplanted heart that was failing due to severe tricuspid valve insufficiency. The successful implantation […]
Data Published in The Lancet Shows Positive Results at Primary Endpoint in the Treatment of Resistant Hypertension with MobiusHD Device
MOUNTAIN VIEW, Calif., Sept. 01, 2017 (GLOBE NEWSWIRE) — Vascular Dynamics, Inc. (VDI), a privately held medical device company developing novel solutions for the treatment of hypertension, today announced European results of the company’s first-in-human trial of its MobiusHD®implant published in The Lancet. The publication reported an average reduction of 24-hour […]
Neovasc’s Reducer System Reviewed in European Heart Journal
VANCOUVER, Sept. 5, 2017 /PRNewswire/ – Neovasc Inc. (“Neovasc” or the “Company“) (NASDAQ, TSX: NVCN) today announced the European Heart Journal published a clinical review of its Neovasc Reducer™ (“Reducer”) device for the treatment of refractory/persistent angina pectoris, a common and disabling medical condition, and a major public health problem that affects […]
Women largely unaware of minimally invasive treatment for uterine fibroids
FAIRFAX, Va., Aug. 31, 2017 /PRNewswire-USNewswire/ — U.S. women are largely unaware of uterine fibroid embolization (UFE), a minimally invasive treatment for uterine fibroids that is less painful, preserves the uterus and allows women to get back to their lives sooner than surgical options, according to results from a new nationwide poll […]
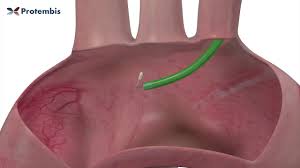
Protembis Announces Successful First-in-Human Use of Its ProtEmbo® Cerebral Protection System in European Trial
AACHEN, Germany–(BUSINESS WIRE)–Protembis GmbH, a privately held medical device company, announced today the first clinical applications of its ProtEmbo® Cerebral Protection System to complement a transcatheter aortic valve replacement (TAVR) procedure. The ProtEmbo® System is an intra-aortic filter device that deflects embolic material arising during TAVR away from the brain. Darren Mylotte, […]
Spectranetics Recalls Bridge Occlusion Balloons
Philips subsidiary Spectranetics recalls Bridge occlusion balloon AUGUST 31, 2017 BY BRAD PERRIELLO , MassDevice Royal Philips (NYSE:PHG) subsidiary Spectranetics recalled its Bridge occlusion balloon after receiving reports of blocked guidewire lumens. The Bridge device is used to temporarily block the superior vena cava for emergency control of hemorrhage. Spectranetics said the August 25 recall was […]
HeartFlow Announces Positive Medical Coverage Decisions on Non-Invasive HeartFlow® FFRct Analysis from Anthem Blue Cross Blue Shield, Blue Shield of California and Blue Cross and Blue Shield of Alabama
REDWOOD CITY, Calif.–(BUSINESS WIRE)–HeartFlow, Inc. today announced that Anthem Blue Cross Blue Shield, Blue Shield of California, and Blue Cross and Blue Shield of Alabama have each issued a positive medical policy for the HeartFlow® FFRct Analysis, a first-of-its-kind noninvasive technology that helps clinicians diagnose and treat patients with suspected coronary artery […]