Worldwide revenue of $123.6 million – an increase of 13.6% year over year (14.1% constant currency) Net loss of $6.7 million – an improvement of $6.5 million year over year Adjusted EBITDA of $8.8 million – an increase of $6.0 million year over year MASON, Ohio–(BUSINESS WIRE)–AtriCure, Inc. (Nasdaq: ATRC), a […]
Other News
United Therapeutics Corporation Reports First Quarter 2025 Financial Results
SILVER SPRING, Md. & RESEARCH TRIANGLE PARK, N.C.–(BUSINESS WIRE)–United Therapeutics Corporation (Nasdaq: UTHR), a public benefit corporation, today announced its financial results for the quarter ended March 31, 2025. Total revenues in the first quarter of 2025 grew 17 percent year-over-year to $794.4 million, compared to $677.7 million in the first […]
Viz.ai Launches Viz 3D CTA™ to Deliver Enhanced Imaging and Accelerate Clinical Decision-Making
ew feature provides AI-enhanced 3D imaging to improve neurovascular visualization and streamline workflows SAN FRANCISCO–(BUSINESS WIRE)–Viz.ai, the leader in AI-powered disease detection and intelligent care coordination, today announced the launch of Viz 3D CTA, a new solution that automatically converts computed tomography angiography (CTA) scans into high-resolution, AI-enhanced 3D images […]
TriSalus Life Sciences Announces $22.0 Million Private Placement Preliminary Q1 2025 Unaudited Financial Results
Funding Anticipated to Accelerate Applications Already in Use and New Additional Potential Applications Anticipated Preferred Stock Exchange to Simplify Capital Structure Private Placement Expected to Fund Business to Profitability Healthcare Focused Long-term Investors Support Strategic Vision DENVER–(BUSINESS WIRE)–TriSalus Life Sciences, Inc. (Nasdaq: TLSI) (the “Company”), an oncology focused medical technology […]
Surmodics Reports Second Quarter of Fiscal Year 2025 Financial Results; Introduces Fiscal Year 2025 Financial Guidance
EDEN PRAIRIE, Minn.–(BUSINESS WIRE)–Surmodics, Inc. (Nasdaq: SRDX), a leading provider of medical device and in vitro diagnostic technologies to the healthcare industry, today reported financial results for its second quarter ended March 31, 2025, and introduced its financial guidance for the fiscal year ending September 30, 2025. Second Quarter Fiscal […]
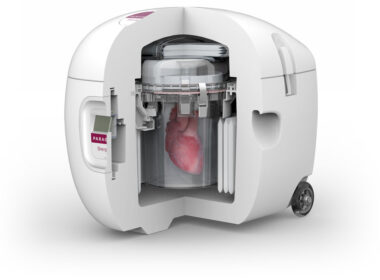
New Study Reports Increased 4-Year Survival in Heart Transplant Patients when Utilizing the Paragonix SherpaPak System
Data from the GUARDIAN Registry, presented at the 45th ISHLT Annual Meeting, shows the Paragonix SherpaPak ® Cardiac Transport System led to statistically significant improvements in short and long-term post-transplant survival and cardiac complications WALTHAM, MA. April 29, 2025 – Paragonix Technologies , a leader in organ transplant technologies and […]
Mach7 Technologies showcases advanced informatics innovations at SiiM25
Mach7 to showcase its Enterprise Imaging Platform with new applications designed to revolutionize the radiology workspace and deliver diagnostic viewing to all clinicians. SOUTH BURLINGTON, Vt., April 30, 2025 /PRNewswire/ — Mach7 Technologies, a company specializing in innovative…
Idorsia announces financial results for the first quarter 2025 – QUVIVIQ taking off in Europe and TRYVIO REMS removal increases the value of this outstanding asset
Ad hoc announcement pursuant to Art. 53 LR Allschwil, Switzerland – April 30, 2025Idorsia Ltd (SIX: IDIA) today announced its financial results for the first quarter of 2025. Business highlights Q1 2025 QUVIVIQ™ (daridorexant): Strong performance and accelerating sales in Q1 2025 with total Idorsia-led net sales of CHF 25 m.QUVIVIQ EUCAN: Demand grew by 50% from Q4 2024 to Q1 2025, strongly driven by reimbursed markets. Overall, more than 10 million nights of sleep prescribed in Q1 2025.Daridorexant: Positive data with daridorexant in patients with chronic insomnia and nocturia published in the Journal of Sleep Research and assessing the transition from night to day published in Sleep Medicine.TRYVIO™ (aprocitentan): REMS requirement removed by US FDA.Aprocitentan: Effect of reducing blood pressure and proteinuria in Black patients with resistant hypertension published in HypertensionRestructured convertible bond debt: Tailored approach to remove large debt overhang.New funding: Bondholders to provide CHF 150 m new money facility.Viatris collaboration: Updated agreement removed significant cash requirement for 2025. Financial highlights Net revenue Q1 2025 of CHF 59 m.US GAAP operating expenses Q1 2025 of CHF 5 m (income) were positively impacted by a one-off gain from the amendment of the Viatris deal with non-GAAP operating expenses Q1 2025 of CHF 78 m.US GAAP operating income Q1 2025 of CHF 67 m and non-GAAP operating loss of CHF 17 m. Guidance for 2025 – unforeseen events excluded QUVIVIQ net sales of around CHF 110 m.SG&A expenses of around CHF 210 m, R&D expenses of around CHF 100 m, leading to non-GAAP operating expenses of around CHF 325 m.US GAAP loss for global business of around CHF 125 m. André C. Muller, Chief Executive Officer of Idorsia, commented:“Beyond the transformation of Idorsia’s financial situation, we made significant progress on multiple fronts in the first quarter. QUVIVIQ is taking off in Europe with a particularly impressive performance in France following the commercial partnership to call on GPs initiated in October 2024; we hope Germany will follow suit, as a similar partnership will kick-in from April 2025. In the US, we have implemented a focused commercialization approach for QUVIVIQ to maintain sales until the potential descheduling of the dual orexin receptor antagonist (DORA) class can be achieved. We received great news from the FDA, with the removal of the REMS requirement for TRYVIO allowing a shift toward broad product availability in retail pharmacies. This, together with the early positive prescribing experience in leading US hypertension centers of excellence gives us confidence of the potential of our antihypertensive drug. Lastly, we streamlined the R&D organization to invest in our promising refocused pipeline. All these achievements put us on a solid path to reach our 2025 goals.” Financial results US GAAP results First Quarterin CHF millions, except EPS (CHF) and number of shares (millions) 20252024Net revenue 5910Operating expenses 520Operating income 6731Net income 6330Basic EPS 0.330.17Basic weighted average number of shares 188.9179.1Diluted EPS 0.230.13Diluted weighted average number of shares 270.8233.3 Net revenue of CHF 59 m in Q1 2025 resulted from QUVIVIQ product sales (CHF 25 m), product sales to partners (CHF 1 m), and contract revenues (CHF 32 m), comprising a one-off exclusivity fee of CHF 32 m paid by an undisclosed party in relation to a potential aprocitentan deal, and non-cash revenue related to the R-Bridge royalty monetization agreement of CHF 1 m. This compares to net revenue of CHF 10 m in Q1 2024 from QUVIVIQ product sales. US GAAP operating expenses of CHF 5 m (income) in Q1 2025 and CHF 20 m (income) in Q1 2024 were impacted by a one-off gain of CHF 90 m (Viatris deal amendment) in 2025 and CHF 125 m (Viatris deal) in 2024, respectively. Excluding these one-off gains, US GAAP operating expenses at Q1 2025 decreased by CHF 20 m, mainly driven by R&D expenses of CHF 27 m decreasing by CHF 6 m compared to Q1 2024 (CHF 33 m), and SG&A expenses of CHF 54 m decreasing by CHF 14 m compared to Q1 2024 (CHF 68 m). US GAAP net income in Q1 2025 of CHF 63 m (CHF 27 m net loss excluding Viatris deal amendment) and CHF 30 m in Q1 2024 (CHF 95 m net loss excluding Viatris deal). Excluding these one-offs, the reduced net loss in Q1 2025 was primarily due to lower operating expenses from cost savings through the effective restructuring efforts announced in November 2024 and higher revenue. The US GAAP net income resulted in a basic net income per share of CHF 0.33 (diluted net income per share of CHF 0.23) in Q1 2025, compared to a basic net income per share of CHF 0.17 (diluted net income per share of CHF 0.13) in Q1 2024. Non-GAAP* measures First Quarterin CHF millions, except EPS (CHF) and number of shares (millions) 20252024Net revenue 5810Operating expenses (78)(96)Operating loss (17)(85)Net loss (25)(86)Basic and diluted EPS (0.13)(0.48)Basic and diluted weighted average number of shares 188.9179.1 * Idorsia measures, reports, and issues guidance on non-GAAP operating performance. Idorsia believes that these non-GAAP financial measurements more accurately reflect the underlying business performance and therefore provide useful supplementary information to investors. These non-GAAP measures are reported in addition to, not as a substitute for, US GAAP financial performance. Non-GAAP net loss in Q1 2025 amounted to CHF 25 m; the difference versus US GAAP net income was mainly driven by the one-off gain from the amendment of the Viatris Deal (CHF 90 m). The non-GAAP net loss resulted in a net loss per share of CHF 0.13 (basic and diluted) in Q1 2025, compared to a net loss per share of CHF 0.48 (basic and diluted) in Q1 2024. Viatris collaborationIn March 2024, Idorsia entered into a global research and development collaboration with Viatris, for the global development and commercialization rights to selatogrel and cenerimod. Idorsia received an upfront payment of USD 350 million (CHF 308 million) with Idorsia obligated to contribute USD 200 million for the development of selatogrel and cenerimod. Idorsia is entitled to potential development and regulatory milestone payments, and certain contingent payments of additional sales milestone payments and tiered royalties in the mid-single to low-double digit percentages on annual net sales. In February 2025, Idorsia reached an agreement with Viatris to update the terms of the collaboration. In exchange for a USD 100 million reduction to Idorsia’s contribution to the development costs due in 2025, Idorsia has agreed to a USD 250 million reduction in future potential regulatory and sales milestone payments, and an expansion of territorial rights to Viatris for cenerimod. The agreed royalties on future sales remain unchanged. Under the updated terms, Idorsia’s contribution for the development of selatogrel and cenerimod is reduced to USD 100 million with no commitment in 2025. Idorsia has contributed USD 73 million in 2024 for the performance of development services, and the remaining USD 27 million will be paid in 2026. Restructured convertible bond debt and new funding securedOn February 26, 2025, Idorsia announced that it has reached an agreement with more than two-thirds of the holders of its outstanding convertible bond debt on the main terms of a holistic restructuring of the bonds and a CHF 150 million new money facility, to alleviate the short- to mid-term debt overhang of CHF 800 million while retaining upside potential of key assets beyond the value of the debt. As part of the holistic restructuring Idorsia will issue up to 27.5 million shares and up to 25.5 million warrants. When complete, the tailored solution secures future operations of Idorsia into 2026. More information can be found in the dedicated press release. Capital increaseIn connection with the holistic restructuring of the convertible bond debt and raising of additional funds, 35 million registered shares with a nominal value of CHF 0.05 each were created out of capital band and were listed on March 4, 2025. Financial guidance for 2025As previously announced, for the Idorsia-led portfolio in 2025, the company expects a continued acceleration of QUVIVIQ with net sales of around CHF 110 million, COGS of around CHF 15 million, SG&A expenses of around CHF 210 million, and R&D expense of around CHF 100 million, leading to non-GAAP operating expenses of around CHF 325 million. This performance would result in an Idorsia-led business non-GAAP operating loss of around CHF 215 million and US-GAAP operating loss of around CHF 260 million. The company expects US-GAAP EBIT for the partnered business of around CHF 135 million – updated to reflect the positive impact of the one-off exclusivity fee paid by an undisclosed party in Q4 2024 but recognized in Q1 2025 – and mainly driven by the amended deal with Viatris. This would result in a US-GAAP loss for the global business of around CHF 125 million. All amounts exclude unforeseen events and potential revenue related to additional business development activities. Arno Groenewoud, Chief Financial Officer, commented:“The updated agreement with Viatris, and the convertible debt restructuring, together with the new money facility agreed with our bondholders, has significantly changed the financial situation of Idorsia. That said, there are still several steps to implement in order to realize what was agreed. The restructuring of the bonds is moving forward with the first step approved by the court, allowing us to proceed to the next bondholder meetings. We are also making progress with putting the new money facility in place. The excellent uplift with QUVIVIQ in Europe and the tight cost-control means we are well on track with our financial performance targets.” Liquidity and indebtednessAt the end of the first quarter of 2025, Idorsia’s liquidity amounted to CHF 51 million. (in CHF millions) Mar 31, 2025Dec 31, 2024Liquidity Cash and cash equivalents 51106Total liquidity* 51106 Indebtedness Convertible loan 335335Convertible bond 797797Other financial debt 190189Total indebtedness 1,3221,321 *rounding differences may occur Commercial operationsIn the first quarter of 2025, QUVIVIQ™ (daridorexant) in the US, Germany, Italy, Switzerland, Spain, UK, Canada, Austria, France, and Sweden generated total product sales of CHF 25 million. Europe and Canada ProductMechanism of actionIndicationCommercially availableDual orexin receptor antagonistTreatment of adult patients with insomnia characterised by symptoms present for at least three months and considerable impact on daytime functioningSweden: Sept. 2024France: Mar. 2024Austria: Feb. 2024UK: Oct. 2023Spain: Sept. 2023Switzerland: Jun. 2023Germany: Nov. 2022Italy: Nov. 2022 Management of adult patients with insomnia, characterized by difficulties with sleep onset and/or sleep maintenanceCanada: Nov. 2023 QUVIVIQ (daridorexant) net sales in the first quarter of 2025 reached CHF 19.4 million in the Europe and Canada (EUCAN) region, a significant increase from CHF 3.5 million in the first quarter of 2024. In France, QUVIVIQ is reimbursed for moderate and severe chronic insomnia patients after, or as an alternative to, cognitive behavioral therapy for insomnia. The outstanding launch in France is driven by a combination of co-promotion with Menarini reflected by a new-to-brand share growing from 1.1% in September 2024 to 9.3% in January 2025 in the general practitioner (GP) segment, and a strong positioning to specialists in retail and hospital settings reflected by a solid new-to-brand share evolution from 9.7% in September 2024 to 14.4% in January 2025 in the psychiatrist segment. In Germany, QUVIVIQ was launched in November 2022 and is the only sleep medication in Germany that can be prescribed for long-term treatment of chronic insomnia. The progress made in Germany is reflected by the performance of QUVIVIQ on the market, with demand increasing by 20% quarter on quarter. In February 2025, Idorsia successfully concluded negotiations for the reimbursement price in Germany. Idorsia is expanding its commercial reach from specialist prescribers to GPs through a commercial partnership with Berlin-Chemie (a wholly owned subsidiary of the Menarini Group), which started in April 2025. In the UK, QUVIVIQ is recommended as first-line pharmaceutical treatment for patients with chronic insomnia, after, or as an alternative to, cognitive behavioral therapy for insomnia (CBT-I). QUVIVIQ was launched in October 2023 at NICE approval. The priority in the UK in 2024 was to secure regional access, and the team has achieved reimbursement throughout 85% of the UK, as well as raising awareness of QUVIVIQ among general practitioners. Increased access and awareness have started to translate into strong demand in the UK which grew by 48% from Q4 2024 to Q1 2025. In Canada, after being approved in April 2023, QUVIVIQ was launched in November 2023 to the private market, representing 55% of the Canadian insomnia market. To date, 85% of private Canadian lives are covered. The focus is now on public payers; the company submitted public reimbursement dossiers and expects decisions by the end of 2025. QUVIVIQ demand in Canada grew by 25% from Q4 2024 to Q1 2025. Austria will soon become the fourth EUCAN country to grant public reimbursement to QUVIVIQ, starting from June 1, 2025. This is a significant achievement in a country that has strict reimbursement rules and underpins the value QUVIVIQ brings to patients, physicians and the healthcare system. In Italy, QUVIVIQ is now officially available for all prescribers, following its publication in the Official Gazette in mid-March 2025, this includes GPs who represent nearly 80% of the total insomnia market. In Switzerland, Spain, and Sweden, where we are still negotiating for reimbursement, launches have been very successful despite the out-of-pocket costs for patients, particularly in Switzerland where we see a strong demand. Benjamin Limal, President of Europe and Canada region, commented:“Demand has grown by an impressive 50% quarter on quarter, mainly driven by reimbursed markets. Across Europe and Canada, more than 10 million nights of sleep have been prescribed in the first quarter of 2025. Recent successes in access and pricing, notably in Austria and Germany, reinforce our confidence for continued growth moving forward. QUVIVIQ has been strongly adopted by specialists and more and more general practitioners start prescribing QUVIVIQ due to our increased efforts with recent partnerships for the GP market.” For more information about QUVIVIQ in the EU, see the Summary of Product Characteristics. For more information about QUVIVIQ in Switzerland, see the Patient Information and Information for Healthcare Professionals. For more information on the marketing authorization of QUVIVIQ in Canada, see the Product Monograph. United States ProductMechanism of actionIndicationCommercially available sinceDual orexin receptor antagonistTreatment of adult patients with insomnia, characterized by difficulties with sleep onset and/or sleep maintenanceMay 2022 QUVIVIQ® (daridorexant) net sales in the first quarter of 2025 amounted to CHF 5.9 million in the US, compared to CHF 6.5 million in the first quarter of 2024. As of the end of the first quarter of 2025, more than 180,000 patients have been treated with QUVIVIQ since launch in the US, over 600,000 prescriptions have been dispensed, and the product has been prescribed by more than 50,000 healthcare professionals. Michael Moye, President and General Manager of Idorsia US, commented:“We have implemented a streamlined, focused, and more cost-efficient commercialization approach for QUVIVIQ to maintain sales until the potential descheduling of the dual orexin receptor antagonist (DORA) class can be achieved. Our commercialization partner, Syneos Health, is fully operational and executing a highly targeted digital marketing plan supporting 20 virtual sales reps. Syneos is now also executing educational programming and market access activities in support of the virtual representatives. We are seeing early, positive prescribing results in key customer areas.” For more information about QUVIVIQ in the US, see the Full Prescribing Information (PI and Medication Guide). ProductMechanism of actionIndicationCommercially available sinceDual endothelin receptor antagonistTreatment of hypertension in combination with other antihypertensive drugs, to lower blood pressure in adult patients who are not adequately controlled on other drugsOctober 2024 On March 19, 2024, the US Food and Drug Administration (FDA) approved TRYVIO™ (aprocitentan) for the treatment of hypertension in combination with other antihypertensive drugs, to lower blood pressure in adult patients who are not adequately controlled on other drugs. Lowering blood pressure reduces the risk of fatal and non-fatal cardiovascular events, primarily strokes and myocardial infarctions. The recommended dosage of TRYVIO is 12.5 mg orally once daily, with or without food. Following the approval, the US team rapidly established the positioning, branding, websites, materials, training and educational platforms, and field sales force and MSL coverage plans. TRYVIO was made available for prescription in October 2024 via Walgreens Specialty Pharmacy. There is ongoing engagement with hypertension experts at major cardiovascular and nephrology congresses and encouraging discussions with payors. In March 2025, the US FDA fully released TRYVIO from its REMS (Risk Evaluation and Mitigation Strategy) requirement to minimize the burden on the healthcare delivery system of complying with the REMS. The US FDA has determined that a REMS is no longer necessary to ensure the benefits of TRYVIO outweigh the risk of embryo-fetal toxicity and that labeling is sufficient for conveying the safety information. As a result, a rapid transition from specialty pharmacy to a wide retail pharmacy distribution model is underway. Funding for a field sales force and promotional activities continues to be dependent on a partnership deal. Michael concluded:“Early prescribing experience in leading US hypertension centers of excellence has been very positive, with prescribers confirming that they are seeing blood pressure reductions, safety and tolerability consistent with the Phase 3 study. The REMS removal and shift toward broad product availability in retail pharmacies has profoundly improved the potential of TRYVIO to reach millions of patients struggling with their hypertension on existing medication regimens.” For more information see the Full Prescribing Information including BOXED Warning (PI and Medication Guide). Research & DevelopmentOur drug discovery engine has produced innovative drugs with the potential to transform the treatment paradigm in multiple therapeutic areas, including CNS, cardiovascular, and immunological disorders, as well as orphan diseases. The company also has a vaccine platform for the discovery and development of glycoconjugate vaccines to prevent infection. The company has focused its drug discovery efforts, reducing the number of active projects in research and development and preparing some for out-licensing. The prioritization has resulted in a portfolio of assets where Idorsia intends to develop to the next inflection point before partnering, or when feasible and appropriate, developing further ourselves. The company expects new lucerastat data from a kidney biopsy sub-study (to the ongoing Phase 3 open-label extension study) in the second quarter of 2025, with further discussions on the regulatory pathway to follow. The results from a Phase 1 study of our Clostridium difficile infection vaccine are also expected in the coming months. The company will need to further prioritize activities in order to reduce costs and the decisions on which assets to advance will be taken based on the data when available and the results of ongoing out-licensing discussions for early-stage assets. In March 2025, “A randomized cross-over trial of daridorexant for the treatment of chronic insomnia and nocturia” was published in the Journal of Sleep Research. The new data provides evidence of the benefit of daridorexant, at a daily dose of 50 mg, in patients aged >=55 years with chronic insomnia and comorbid nocturia, with efficacy data on symptoms of both conditions, improvement in daytime functioning, and a good safety and tolerability profile. In April 2025, the “Effect of daridorexant on nighttime wakefulness and next-morning sleepiness: assessing the transition from night to day in insomnia disorder” was published in Sleep Medicine. The analysis of the Phase 3 data provides evidence that daridorexant reduces wakefulness throughout the entire night, while decreasing morning sleepiness and improving daytime functioning and alertness in patients with chronic insomnia disorder. Idorsia-led portfolioThe company will develop each asset to the next inflection point or seek a partner. CompoundMechanism of actionTarget indicationStatusQUVIVIQ™ (daridorexant)Dual orexin receptor antagonistInsomniaCommercialized by Idorsia in the US, Germany, Italy, Switzerland, Spain, the UK, Canada, Austria, France, and Sweden; approved throughout the EU.LucerastatGlucosylceramide synthase inhibitorFabry diseasePhase 3 open-label extension study ongoing – kidney biopsy sub-study results expected in Q2 2025 – regulatory pathway to be further discussed with FDA.DaridorexantDual orexin receptor antagonistPediatric insomniaPhase 2 in pediatric insomnia is ongoing.ACT-777991CXCR3 receptor antagonistVitiligoProof-of-concept study in preparation for patients with vitiligo. Unique precision medicine with a dual targeting of CD8+ CXCR3+ T cells offers potential for a first-in-class targeted systemic therapy for effective and safer treatment of immuno-dermatology and autoimmune disorders.ACT-1004-1239ACKR3 (CXCR7) receptor antagonistProgressive multiple sclerosis Proof-of-concept study in preparation for patients with progressive MS. Unique combination of re-myelination and anti-inflammatory effect with decreased inflammatory cell infiltration.IDOR-1117-2520CCR6 receptor antagonistPsoriasisProof-of-concept study in preparation for patients with psoriasis. Unique potential as a first-in-class, oral, targeted systemic therapy for effective treatment of Th17-driven immuno-dermatology and autoimmune disorders.ACT-1016-0707LPA 1 receptor antagonistImmune-mediated and fibrosis related disordersEntry-into-human package complete. Potential best-in-class due to insurmountable binding mode – proven inhibitory activity in preclinical models of inflammation and fibrosis.IDOR-1134-9712CFTR Type-IV correctorCystic FibrosisEntry-into-human package in progress. A unique corrector targeting an Idorsia-identified binding site on the Cystic Fibrosis Transmembrane regulator (CFTR) protein. Potential synergy with other molecules.IDOR-1141-8472Orexin 2 receptor agonistOrexin-related CNS disordersEntry-into-human package ready to begin. Potential best-in-class – sustained chronic efficacy in a preclinical model of narcolepsy.IDOR-1126-6421Undisclosed mechanismOrgan injury / fibrosisEntry-into-human package in progress. Broad potential of undisclosed mechanism for inhibiting organ injury and fibrosis – proven effectiveness in several preclinical models of organ injury. Synthetic Glycan Vaccine PlatformIdorsia will seek a partner for the platform or individual vaccines.IDOR-1134-2831Synthetic glycan vaccineClostridium difficile infectionIdorsia is conducting a Phase 1 clinical pharmacology study which will test the immune response of the vaccine and evaluate its safety and tolerability. Results expected in Q2 2025.IDOR-1142-0810Synthetic glycan vaccineKlebsiella pneumonia infection Entry-into-human package in progress. Further details including the current status of each project in our portfolio can be found in our innovation fact sheet.Idorsia partner-led portfolioFor Idorsia, partnerships are a way of gaining strategic access to technologies or products and fully exploiting our discovery engine and clinical pipeline. We seek suitable external project partners to maximize the value of internal innovation. On March 19, 2024, the US Food and Drug Administration (FDA) approved TRYVIO™ (aprocitentan) for the treatment of hypertension in combination with other antihypertensive drugs, to lower blood pressure in adult patients who are not adequately controlled on other drugs. See the commercial operations section above. On June 27, 2024, the European Commission (EC) approved JERAYGO™ (aprocitentan) for the treatment of resistant hypertension in adult patients in combination with at least three antihypertensive medicinal products. The recommended dose is 12.5 mg orally once daily. The dose can be increased to 25 mg once daily for patients tolerating the 12.5 mg dose and in need of tighter blood pressure (BP) control. For more information about JERAYGO in the EU, see the Summary of Product Characteristics. Aprocitentan is an innovative and highly differentiated drug, commercially available in the US and approved in Europe and UK for the millions of patients who are unable to bring their hypertension under control with existing medications. As the first drug to target the endothelin pathway in systemic hypertension, aprocitentan has blockbuster potential in uncontrolled hypertension, particularly for difficult to treat patients with chronic kidney disease and hypertension, and further potential beyond hypertension. The priority remains to partner aprocitentan, having been released from the exclusivity constraint with the undisclosed party, the company will resume discussions with alternative potential partners that recognize the value of aprocitentan. In April 2025, “Aprocitentan for Blood Pressure Reduction in Black Patients” was published in Hypertension. The publication reports preplanned analyses of the efficacy, tolerability and safety of aprocitentan in the subgroup of African American patients enrolled in the Phase 3 PRECISION study in patients with confirmed resistant hypertension. Aprocitentan, when added to a combination of at least three antihypertensive drugs (four in more than 50% of patients), produced clinically meaningful and sustained blood pressure reductions. Aprocitentan also markedly decreased proteinuria in the patients with proteinuria at baseline. As reported by the authors, aprocitentan was safe and well tolerated, even in those Black patients with chronic kidney disease. CompoundMechanism of actionTarget indicationPartner/statusTRYVIO™ (aprocitentan) Dual endothelin receptor antagonistSystemic hypertension in combination with other antihypertensivesTo be defined: worldwide development and commercialization rightsCommercially available in the USJERAYGO™ (aprocitentan)Dual endothelin receptor antagonistResistant hypertension in combination with other antihypertensivesTo be defined: worldwide development and commercialization rights Approved in the EU and UK; Marketing authorization applications under review in Canada, and SwitzerlandQUVIVIQ™ (daridorexant)Dual orexin receptor antagonistInsomniaNxera Pharma: license to develop and commercialize for Asia-Pacific region (excluding China)Launched for the treatment of insomnia in Japan; Phase 3 ongoing in South KoreaDaridorexantDual orexin receptor antagonistInsomniaSimcere: license to develop and commercialize for Greater China regionNDA submitted in Greater China; approved for the treatment of insomnia in Hong-KongSelatogrelP2Y12 inhibitorAcute myocardial infarctionViatris: worldwide development and commercialization rightsPhase 3 “SOS-AMI” program ongoingCenerimodS1P1 receptor modulatorSystemic lupus erythematosusViatris: worldwide development and commercialization rightsPhase 3 “OPUS” program ongoingDaridorexantDual orexin receptor antagonistPosttraumatic stress disorder (PTSD)US Department of Defense (DOD): Idorsia is supporting a clinical study sponsored by the US DOD to develop new therapies to treat PTSDACT-1002-4391EP2/EP4 receptor antagonistImmuno-oncologyOwkin: global license to develop and commercializePhase 1 ongoing Further details including the current status of each project in our partner-led portfolio can be found in our innovation fact sheet. Note to ShareholdersThe Annual General Meeting (AGM) of Shareholders to approve the Annual Report of the year ending December 31, 2024, will be held on Wednesday, May 28, 2025. In order to attend and vote at the AGM, shareholders must be registered in the company’s shareholder register by May 19, 2025, 17:00 CEST, at the latest. Results Day CenterInvestor community: To make your job easier, we provide all relevant documentation via the Results Day Center on our corporate website: www.idorsia.com/results-day-center. Events Annual General Meeting of Shareholders on May 28, 2025Half-Year 2025 Financial Results reporting on July 30, 20259-Month 2025 Financial Results reporting on October 30, 2025 Notes to the editor About IdorsiaIdorsia Ltd is reaching out for more – we have more passion for science, we see more opportunities, and we want to help more patients. The purpose of Idorsia is to challenge accepted medical paradigms, answering the questions that matter most. To achieve this, we will discover, develop, and commercialize transformative medicines – either with in-house capabilities or together with partners – and evolve Idorsia into a leading biopharmaceutical company, with a strong scientific core. Headquartered near Basel, Switzerland – a European biotech hub – Idorsia has a highly experienced team of dedicated professionals, covering all disciplines from bench to bedside; QUVIVIQ™ (daridorexant), a different kind of insomnia treatment with the potential to revolutionize this mounting public health concern; strong partners to maximize the value of our portfolio; a promising in-house development pipeline; and a specialized drug discovery engine focused on small-molecule drugs that can change the treatment paradigm for many patients. Idorsia is listed on the SIX Swiss Exchange (ticker symbol: IDIA). For further information, please contactInvestor & Media RelationsIdorsia Pharmaceuticals Ltd, Hegenheimermattweg 91, CH-4123 Allschwil+41 58 844 10 10 investor.relations@idorsia.com – media.relations@idorsia.com – www.idorsia.com The above information contains certain “forward-looking statements”, relating to the company’s business, which can be identified by the use of forward-looking terminology such as “estimates”, “believes”, “expects”, “may”, “are expected to”, “will”, “will continue”, “should”, “would be”, “seeks”, “pending” or “anticipates” or similar expressions, or by discussions of strategy, plans or intentions. Such statements include descriptions of the company’s investment and research and development programs and anticipated expenditures in connection therewith, descriptions of new products expected to be introduced by the company and anticipated customer demand for such products and products in the company’s existing portfolio. Such statements reflect the current views of the company with respect to future events and are subject to certain risks, uncertainties and assumptions. Many factors could cause the actual results, performance or achievements of the company to be materially different from any future results, performances or achievements that may be expressed or implied by such forward-looking statements. Should one or more of these risks or uncertainties materialize, or should underlying assumptions prove incorrect, actual results may vary materially from those described herein as anticipated, believed, estimated or expected.
Attachment
Press Release PDF
Impulse Dynamics®’ INTEGRA-D Trial Completes Efficacy Phase
First phase of enrollment of single device for CCM® and ICD therapy complete MARLTON, N.J., April 28, 2025 (GLOBE NEWSWIRE) — Impulse Dynamics® plc, a global medical device company dedicated to improving the lives of people with heart failure (HF), is proud to announce that it reached an important milestone in the […]
Transverse Medical Inc. Announces Completion of Series B2 Capital Raise
Point-Guard Protected™ Safeguard the Brain, Treat the Heart. DENVER–(BUSINESS WIRE)–Transverse Medical Inc., a leading innovator in Cerebral Embolic Protection for stroke prevention during TAVR (Transcatheter Aortic Valve Replacements) and early-stage medical device technology company, today announced the successful completion of its Series B2 financing round. The company secured over $10 million […]