MOUNTAIN VIEW, Calif., April 22, 2025 (GLOBE NEWSWIRE) — R3 Vascular Inc., a medical device company dedicated to developing and providing novel, best-in-class bioresorbable scaffolds for treating peripheral arterial disease (PAD), is pleased to announce that the first patient in its ELITE-BTK pivotal trial was treated by Dr. Brian DeRubertis, FACS, Chief of the Division of Vascular & Endovascular Surgery at New York-Presbyterian and Weill Cornell Medicine in New York City. The trial evaluates R3 Vascular’s next generation drug eluting bioresorbable scaffold, MAGNITUDE®, for below-the-knee (BTK) PAD, which according to the American Heart Association affects more than 200 million people globally, and 10 to 12 million in the U.S. older than the age of 40. The most common type of PAD affects the lower-extremity where blood flow is reduced to the legs and feet. Each year, approximately 150,000 leg amputations are performed in the U.S. alone.
Other News
FIELD MEDICAL CLOSES $40 MILLION SERIES A FINANCING TO REDEFINE PULSED FIELD ABLATION FOR VENTRICULAR TACHYCARDIA
CARDIFF-BY-THE-SEA, Calif., April 22, 2025 /PRNewswire/ — Field Medical Inc., a pioneer in cardiac pulsed field ablation (PFA) technology, today announced the successful closing of $40 million in Series A financing. The round includes $20 million in new capital and the conversion of $20…
Transverse Medical Inc. Announces Completion of First Series of Patients in Feasibility Clinical Study at Victorian Heart Hospital, Melbourne, Australia, Using Point-Guard Cerebral Embolic Protection.
DENVER–(BUSINESS WIRE)–Transverse Medical Inc. is excited to announce the completion of the first series of patients under its Feasibility Clinical Study at the prestigious Victorian Heart Hospital in Melbourne, Australia. The patients undergoing TAVR procedures (Transcatheter Aortic Valve Replacement) were treated using the company’s next generation POINT-GUARD™ Cerebral Embolic Protection […]
The Future of AI & Electrophysiology Takes Center Stage at Heart Rhythm 2025 with Vektor Medical
SAN DIEGO–(BUSINESS WIRE)–Vektor Medical, a leader in non-invasive, AI-powered arrhythmia analysis technology, will showcase its transformative vMap technology at Heart Rhythm 2025, taking place April 25–27 in San Diego. Through live demonstrations at adjacent booths (#1523 and #1622), scientific abstract presentations, and a highly anticipated Rhythm Theater session featuring leading […]
CathVision to Exhibit the Latest Advancements in Electrophysiology at Heart Rhythm Society 2025 in San Diego
COPENHAGEN, Denmark, April 21, 2025 /PRNewswire/ — CathVision, a leader in advanced cardiac electrophysiology (EP) technology, is pleased to announce its participation and exhibition at the Heart Rhythm Society (HRS) 2025 Annual Scientific Sessions at the San Diego Convention Center in…
Elutia Initiates EluPro™ Registry Study Designed to Generate Evidence Supporting the Use of EluPro in Real-World Clinical Practice
— Integration of clinical and patient-reported outcomes expected to further differentiate EluPro’s utility in cardiac implantable electronic device (CIED) procedures — SILVER SPRING, Md., April 21, 2025 (GLOBE NEWSWIRE) — Elutia Inc. (Nasdaq: ELUT) (“Elutia” or the “Company”), a pioneer in drug-eluting biomatrix technologies, today announced the initiation of an EluPro™ clinical study designed to collect patient outcome data in real-world clinical practice. EluPro, the first and only FDA-cleared antibiotic-eluting bioenvelope designed for use with cardiac implantable electronic devices (CIEDs) and neurostimulators, was commercially launched earlier this year. The first patient was enrolled at UC San Diego Health. “Every innovation we pursue is driven by a commitment to improving patient care,” said Kimberly Mulligan, PhD, Vice President and General Manager of Cardiovascular at Elutia. “With EluPro, we combined trusted antibiotics with a soft, regenerative biomatrix to protect the implant, facilitate implantation, and support healing. This study will allow us to collect data on these differentiating characteristics in real-world practice.” The multi-center clinical study is a prospective, post-market study designed to evaluate the use of EluPro in standard clinical practice and its performance across a diverse population of patients undergoing CIED implantation. Data on clinical and patient-reported outcomes will be collected, which will include assessments of key complications of interest following CIED implantation – such as infection, hematoma, lead dislodgement, device migration or erosion, and implant site complications. The study plans to enroll 100 patients, who will be followed for 12 months after device implantation. Each year, more than 600,000 CIEDs are implanted in the U.S., with overall complication rates up to 5-7%, including infections linked to higher morbidity and mortality. EluPro is cleared for use across all major CIED brands including pacemakers and implantable defibrillators, as well as for a wide range of neurostimulation devices. Unlike synthetic alternatives, EluPro addresses this critical need by combining the antibiotics rifampin and minocycline with a soft, regenerative biomatrix that promotes healing and helps reduce other complications, such as migration and erosion. The CIED protection market is valued at $600 million in the U.S. To learn more, visit www.elutia.com/products/elupro/. About Elutia Elutia develops and commercializes drug-eluting biomatrix products to improve compatibility between medical devices and the patients who need them. With a growing population in need of implantable technologies, Elutia’s mission is humanizing medicine so patients can thrive without compromise. For more information, visit www.Elutia.com. Forward Looking Statements This press release contains “forward-looking statements” within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. Forward-looking statements can be identified by words such as “projects,” “may,” “will,” “could,” “would,” “should,” “believes,” “expects,” “anticipates,” “estimates,” “intends,” “plans,” “potential,” “promise” or similar references to future periods. All statements contained in this press release that do not relate to matters of historical fact should be considered forward-looking statements, including any statements and information concerning the EluPro Registry Study, including the timing and anticipated data, and the value of the CIED protection market. These forward-looking statements are based on our management’s beliefs and assumptions and on information currently available to us. Such beliefs and assumptions may or may not prove to be correct. Additionally, such forward-looking statements are subject to a number of known and unknown risks, uncertainties and other important factors that may cause our actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied in the forward-looking statements, including, but not limited to the following: our ability to successfully commercialize, market and sell our newly approved EluPro product; our ability to continue as a going concern; our ability to achieve or sustain profitability; the risk of product liability claims and our ability to obtain or maintain adequate product liability insurance; our ability to defend against the various lawsuits and claims related to our recalled FiberCel and other viable bone matrix products and avoid a material adverse financial consequence from those lawsuits and claims; the continued and future acceptance of our products by the medical community; our ability to enhance our products, expand our product indications and develop, acquire and commercialize additional product offerings; our dependence on our commercial partners and independent sales agents to generate a substantial portion of our net sales; our dependence on a limited number of third-party suppliers and manufacturers, which, in certain cases are exclusive suppliers for products essential to our business; our ability to successfully realize the anticipated benefits of the November 2023 sale of our Orthobiologics business; physician awareness of the distinctive characteristics, benefits, safety, clinical efficacy and cost-effectiveness of our products; our ability to compete against other companies, most of which have longer operating histories, more established products and/or greater resources than we do; pricing pressure as a result of cost-containment efforts of our customers, purchasing groups, third-party payors and governmental organizations could adversely affect our sales and profitability; our ability to obtain regulatory approval or other marketing authorizations by the FDA and comparable foreign authorities for our products and product candidates; and our ability to obtain, maintain and adequately protect our intellectual property rights; and other important factors which can be found in the “Risk Factors” section of Elutia’s public filings with the Securities and Exchange Commission (“SEC”), including Elutia’s Annual Report on Form 10-K for the year ended December 31, 2024, as such factors may be updated from time to time in Elutia’s other filings with the SEC, accessible on the SEC’s website at www.sec.gov and the Investor Relations page of Elutia’s website at https://investors.elutia.com. Because forward-looking statements are inherently subject to risks and uncertainties, you should not rely on these forward-looking statements as predictions of future events. Any forward-looking statement made by Elutia in this press release is based only on information currently available and speaks only as of the date on which it is made. Except as required by applicable law, Elutia expressly disclaims any obligations to publicly update any forward-looking statements, whether written or oral, that may be made from time to time, whether as a result of new information, future developments or otherwise. Investors:Matt SteinbergFINN Partnersmatt.steinberg@finnpartners.com This press release was published by a CLEAR® Verified individual.
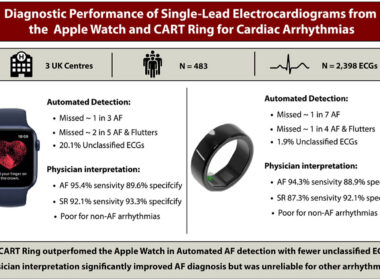
UK Clinical Study Finds Sky Labs’ Smart Ring ‘CART-I’ More Sensitive Than Apple Watch for AFib Detection
Published in the international journal Heart Rhythm O2, the clinical study demonstrates that CART-I achieved 84.6% sensitivity – higher than Apple Watch’s 69.1% Sky Labs also recognized with Korea’s top tech honor: IR52 Jang Young-sil Award – Prime Minister’s Award SEOUL, South Korea,…
Venova Medical Announces Completion of Enrollment for VENOS-2 IDE Study of the VelocityTM Percutaneous AVF System
Los Gatos, California – Venova Medical, a privately held company developing a next generation technology for the creation of percutaneous arteriovenous fistulas (pAVF) for hemodialysis access, today announced the successful completion of patient enrollment for the company’s VENOS-2 early feasibility study (NCT06712251). This multi-center study is being performed under an Investigational […]
Eight-Year Data Confirm Long-Term Durability of Edwards’ RESILIA Tissue
CAIRO–(BUSINESS WIRE)–Edwards Lifesciences (NYSE: EW) today announced new eight-year data showing that patients receiving aortic surgical valves treated with its proprietary RESILIA tissue technology have significantly improved long-term outcomes compared to those receiving non-RESILIA tissue bioprosthetic valves. For more than 65 years, Edwards has been the leader in heart valve […]
Adagio Medical Holdings, Inc. Receives FDA Breakthrough Device Designation for the vCLASTM Cryoablation System
Agency’s Breakthrough Device Designation Program Allows for Priority Review of Ablation Technology for Ventricular Tachycardia LAGUNA HILLS, Calif.–(BUSINESS WIRE)–Adagio Medical Holdings, Inc. (Nasdaq: ADGM) (“Adagio” or “the Company”), a leading innovator in catheter ablation technologies for the treatment of cardiac arrhythmias, today announced that it has received Breakthrough Device designation […]