CAMBRIDGE, Mass.–(BUSINESS WIRE)–Anumana announced US FDA clearance of its ECG-AI™ algorithm for cardiac amyloidosis.
Other News
Adagio Medical Receives IDE Approval from the FDA to Investigate Next-Generation vCLAS® Ventricular Ablation System
LAGUNA HILLS, Calif.–(BUSINESS WIRE)–Adagio Medical Holdings, Inc. (Nasdaq: ADGM) (“Adagio” or “the Company”), a leading innovator in catheter ablation technologies for the treatment of cardiac arrhythmias, today announced that the U.S. Food and Drug Administration (FDA) has granted Investigational Device Exemption (IDE) approval to expand the Company’s FULCRUM-VT trial to evaluate the safety and effectiveness of the Company’s next-generation vCLAS Ultra-Low Temperature Ablation (ULTA) Ventri
AliveCor Expands AI-Powered 12-lead Cardiac Diagnostics to Vietnam, Advancing Access to Early Detection and Modernized Cardiac Care
Kardia 12L ECG System
Kardia 12L ECG System Enables Faster, Easier Detection of Life-Threatening Cardiac Conditions Using Reduced Leadset
MOUNTAIN VIEW, Calif., April 07, 2026 (GLOBE NEWSWIRE) — AliveCor, the global leader in AI-powered cardiology, today announced the regulatory registration of its Kardia™ 12L electrocardiogram (ECG) System powered by KAI™ 12L AI technology in Vietnam, marking an important step toward expanding AI-enabled cardiac diagnostics across Southeast Asia. The clearance supports Vietnam’s growing commitment to healthcare modernization and strengthens clinicians’ ability to detect and manage cardiovascular disease earlier and more efficiently. Cardiovascular disease remains a leading cause of death globally and across Asia, accounting for 33% of all deaths in Vietnam. Rising rates of hypertension, diabetes, and urbanization-related lifestyle changes are contributing to increasing cardiac risk. At the same time, the Ministry of Health has prioritized digital transformation, hospital capacity optimization, and expanded access to quality care, particularly beyond major urban centers. AI-enabled ECG systems like Kardia 12L align with broader efforts to advance care delivery, support data-driven decision-making, and extend specialist-level insight into frontline settings. Expanding Access to Hospital-Grade Cardiac Insight Kardia 12L is the portable, AI-powered, 12-lead ECG solution whose measurements and ECG interpretation are highly similar to standard 12-lead ECG solutions, right at the point of care, enabling rapid acquisition of complete ECG information and offering a better patient experience. By enabling fast, high-quality diagnostics in clinics, community health centers, and outpatient settings, the platform helps clinicians detect arrhythmias and morphologies including serious conditions like acute myocardial infarction and the most common types of cardiac ischemia earlier, without reliance on traditional, fixed hospital infrastructure. This capability is particularly relevant in Vietnam’s diverse healthcare landscape, where care is delivered across densely populated cities and remote and rural provinces. Kardia 12L supports a more distributed model of cardiac diagnostics, empowering healthcare professionals with accurate, actionable insights wherever patients are seen. Strengthening Healthcare Efficiency and Clinical Confidence Vietnam’s healthcare system continues to manage high patient volumes in major hospitals. Streamlined diagnostic workflows can play a meaningful role in reducing bottlenecks and improving throughput. Kardia 12L’s rapid acquisition and reduced electrode setup simplify the ECG process, shortening preparation time while maintaining diagnostic rigor. The simplified five-electrode setup makes it less invasive for patients, who do not need to fully disrobe during a reading, and typically allows for faster acquisition. Research has shown nearly a 30% reduction in ECG acquisition time compared to standard 12-lead setups. Faster acquisition enables healthcare professionals to move from suspicion to confirmation more quickly, thereby improving triage decisions and accelerating treatment pathways when needed. By combining portability with advanced AI interpretations, the system supports efficient clinician decision making while optimizing patient flow, contributing to both quality and efficiency. Enabling the Next Phase of Digital Cardiac Care As Vietnam advances national digital health initiatives, connected diagnostic tools are becoming increasingly important. Kardia 12L now strengthens AliveCor’s cardiovascular portfolio by standing alongside KardiaMobile and KardiaMobile 6L, creating a unified ecosystem for holistic management of the entire cardiovascular pathway. With its offering expansion into Vietnam, AliveCor reinforces its commitment to supporting healthcare professionals across Southeast Asia with scalable, technology-enabled cardiac solutions designed to improve early detection and expand access to quality cardiovascular care. For more information, sign up for exclusive updates on the Kardia 12L expansion outside of the U.S. here: https://alivecor.com/uk/forms/kardia-12l-interest-registration. About AliveCorAliveCor, Inc., the leading provider of FDA-cleared personal electrocardiogram (ECG) devices, is transforming cardiology with its medical-grade AI solutions. The company was named to the inaugural TIME World’s Top Health Tech Companies 2025 list – recognition of its commitment to delivering innovative devices and services that empower patients and physicians with personalized, actionable heart data. With over 350 million ECGs recorded, the company’s Kardia devices are the most clinically validated personal ECGs in the world and can remotely detect six of the most common heart arrhythmias in just 30 seconds. The company’s latest offering, Kardia 12L ECG System, powered by KAI 12L cleared to detect 39 cardiac conditions (with determination availability varying by geography), was designed exclusively for use by healthcare providers. AliveCor’s enterprise platform allows third-party providers to manage their patients’ and customers’ heart conditions simply using state-of-the-art tools that provide easy front-end and back-end integration to AliveCor technologies, addressing gaps in care and improving the treatment experience for patients across a range of disease areas. AliveCor is a privately held company headquartered in Mountain View, Calif. For more information, visit alivecor.com and follow us on LinkedIn, X, Instagram and Facebook. Media Contactpress@alivecor.com Sales ContactOUSenquiries@aliveor.com A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/7436ab48-0e5f-44b7-9289-6bc166f49740
Okami Medical Announces First Patient Enrolled in ALPHA Registry
ALISO VIEJO, Calif., April 7, 2026 /PRNewswire/ — Okami Medical, a medical technology company pioneering novel vascular embolization solutions, today announced the enrollment of the first patient in the ALPHA Registry, a prospective, multicenter registry evaluating the safety and…
InterVene Completes First 30 Cases Using the Recana® Thrombectomy Catheter System, Plans for Broader Commercial Launch
REDWOOD CITY, Calif.–(BUSINESS WIRE)–InterVene, Inc., a privately held medical device company advancing interventional care for venous disease, today announced the successful completion of 30 cases using the Recana® Thrombectomy Catheter System. The cases were performed across the company’s limited US market release and its European first-in-human study. “We’ve had the privilege of partnering with some of the most experienced and knowledgeable physicians to reach this important milestone,” sa
Johnson & Johnson Advances Pulsed Field Ablation Portfolio with the Launch of VARIPULSE Pro in Europe
IRVINE, Calif.–(BUSINESS WIRE)–Johnson & Johnson (NYSE: JNJ) today announced the launch of VARIPULSE Pro in Europe following CE Mark approval, further advancing its pulsed field ablation (PFA) portfolio. This new pulse sequence marks the next evolution of the VARIPULSE Platform and is designed to improve procedural efficiencyi,ii while reinforcing its established […]
HeartFocus and Inteleos Launch First AI Cardiac POCUS Certification
Rigorous training and certification gives clinicians a validated pathway to AI-enabled cardiac POCUS proficiencyROCKVILLE, Md. and BORDEAUX, France, April 07, 2026 (GLOBE NEWSWIRE) — HeartFocus, the AI-enabled cardiac imaging software developed by data-driven medtech company DESKi, today announced a partnership with Inteleos® through its Point-of-Care Ultrasound (POCUS) Certification Academy™. Together, they are launching the HeartFocus POCUS Certification, a fast, accessible, and comprehensive training pathway designed to provide healthcare professionals with a structured pathway to AI proficiency in cardiac ultrasound and to help them confidently perform high-quality heart scans at the point of care with HeartFocus. As cardiovascular disease continues to rise, AI-powered cardiac POCUS is emerging as a cost-effective way to expand access to earlier detection. While AI adoption in healthcare has accelerated, training and trust have lagged — this collaboration closes the gap by operationalizing competency and validation for frontline users. A recent HeartFocus study revealed that 69% of Americans are comfortable with AI-enabled cardiac care, signaling that clinicians and patients are ready for AI; what has been missing is a validated, competency-based route to use it responsibly. The HeartFocus POCUS Certification was developed with this in mind, combining DESKi’s HeartFocus AI-enabled cardiac imaging solution with the POCUS Certification Academy’s rigorous assessment standards to provide a structured course with guided hands-on scanning and expert image review to validate cardiac POCUS proficiency across diverse care settings. Key elements include: Structured training and assessment: covering core cardiac ultrasound fundamentals and the use of HeartFocus, followed by a final knowledge assessment within a competency-based framework.Practical skills validation: remote evaluation of images created using HeartFocus, by experts to assess diagnostic quality and confirm consistent performance.Recognized certification: a personal credential demonstrating verified competency in cardiac POCUS using HeartFocus, enabling clinicians and institutions to demonstrate responsible AI use. “POCUS is rapidly becoming a core competency for clinicians everywhere, but education and objective skills validation must keep pace with technology,” said Jasmine Rockett, Inteleos® POCUS Certification Academy Director. “While AI adoption in healthcare has accelerated, training and trust have lagged — this collaboration closes that gap by providing a validated, competency-based route to use AI responsibly. Through this partnership with DESKi and the launch of the HeartFocus POCUS Certification, we are offering a scalable, competency-based pathway that helps clinicians gain confidence in cardiac ultrasound assisted by AI while helping health systems standardize quality and reduce variability in care.” Bertrand Moal, PhD, MD, CEO of DESKi said, “HeartFocus was created to make world-class echocardiography accessible to any trained healthcare professional at the bedside. The response since our launch has been overwhelmingly positive, but clinicians have communicated that confidence is a barrier. This certification closes that gap, giving healthcare professionals a practical way to build skills and bring high-quality cardiac imaging into everyday care.” The certification is available for healthcare institutions or medical professionals globally, and helping organizations operationalize trusted, competency-based AI use in cardiac POCUS. To learn more about The HeartFocus POCUS Certification, visit www.pocus.org/heartfocus About Inteleos Inteleos™ is a non-profit organization dedicated to ensuring equitable access to quality healthcare globally. It oversees the American Registry for Diagnostic Medical Sonography® (ARDMS®), the Alliance for Physician Certification & Advancement™ (APCA™), and the Point-of-Care Ultrasound Certification Academy™ (PCA), collectively representing 150,000+ certified medical professionals worldwide. About HeartFocusHeartFocus is an FDA-cleared, AI-powered cardiac ultrasound application designed to expand access to diagnostic-quality heart imaging at the point of care. Using real-time AI guidance and automatic image quality validation, it enables healthcare professionals – including physicians, nurses, PAs, and NPs – to capture clinical-grade cardiac ultrasound scans after just hours of training. Built on proprietary algorithms trained on millions of data points and validated through clinical studies, HeartFocus supports standard transthoracic views and integrates with existing ultrasound and DICOM/PACS workflows. Developed by DESKi, HeartFocus is driven by the belief No heart can wait™, helping clinicians identify cardiac conditions earlier across primary care, emergency, and frontline settings. Learn more at www.heartfocus.ai HeartFocus by DESKi Inquiries: Pernille Bruun-Jensen, Chief Growth Officermediarelations@deski.ai Inteleos Inquiries: Lisa Jordan, Director of Brand Strategy and Communications Lisa.jordan@inteleos.org
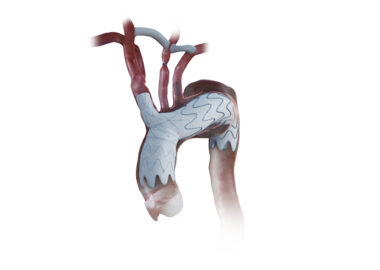
Endospan Receives FDA Approval for the NEXUS® Aortic Arch Stent Graft System
TEL AVIV, Israel–(BUSINESS WIRE)– #aortaEd–Endospan, a privately-held company pioneering solutions for the endovascular repair of aortic arch disease, today announced that the U.S. Food and Drug Administration (FDA) has approved the NEXUS® Aortic Arch Stent Graft. FDA approval was supported by the one-year results of the TRIOMPHE Investigational Device Exemption (IDE) Study evaluating the NEXUS® System shown to effectively treat aortic arch disease, including chronic aortic dissections, in patients at
Route 92 Medical Announces $50 Million Growth Financing
New investor Sectoral Asset Management joins growth financing round to support acceleration of commercial growth April 7, 2026 WEST JORDAN, Utah— Route 92 Medical, Inc., a privately held medical technology company dedicated to improving outcomes for patients undergoing neurovascular intervention, today announced a $50 million growth financing. New investor Sectoral Asset Management joined […]
Merakris Therapeutics Provides FDA Update and Reports Interim Findings for MTX-001 in Venous Leg Ulcers
RESEARCH TRIANGLE PARK, N.C., April 07, 2026 (GLOBE NEWSWIRE) — Merakris Therapeutics today provided an update on recent communications with the U.S. Food and Drug Administration (FDA) related to its Investigational New Drug (IND) application for MTX-001, an investigational biologic being developed for venous leg ulcers (VLUs).