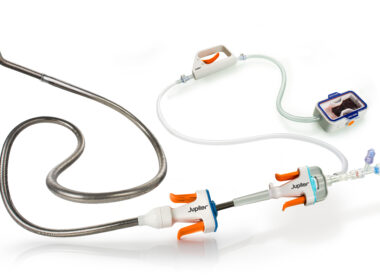
MENLO PARK, Calif.–(BUSINESS WIRE)–Jupiter Endovascular, Inc., a medical technology startup developing a new class of endovascular procedures using Endoportal Control™, announced today that the first two patients have been treated in SPIRARE I (NCT06571760), a multicenter study of the Vertex Pulmonary Embolectomy System using the company’s Endoportal Control™ platform technology. […]
Other News
NewVue Announces Strategic Partner Agreement with Konica Minolta Healthcare to Enhance Radiology Workflow
TAMPA, Fla. and WAYNE, N.J., Sept. 19, 2024 (GLOBE NEWSWIRE) — NewVue, a leader in cloud-native radiology workflow solutions, and Konica Minolta Healthcare Americas, Inc., a leading provider of medical diagnostic imaging and healthcare information technology, announce a strategic partner agreement. This partnership allows Konica Minolta to offer NewVue’s innovative EmpowerSuite Radiology Workflow Orchestrator and Command Center to its extensive network of healthcare customers across the US, integrating seamlessly with Konica Minolta’s Exa® Platform to enhance radiology workflows. EmpowerSuite is a key component of Konica Minolta’s enterprise imaging portfolio.
CorMedix Inc. Announces New Commercial Agreement
BERKELEY HEIGHTS, N.J., Sept. 19, 2024 (GLOBE NEWSWIRE) — CorMedix Inc. (Nasdaq: CRMD), a biopharmaceutical company focused on developing therapeutic products for life-threatening diseases and conditions, today announced that it has entered into a new commercial supply contract with a top-tier mid-sized dialysis operator for the supply of DefenCath® (taurolidine and heparin) to dialysis clinics in the US. This new agreement will provide access to DefenCath for adult patients with kidney failure receiving hemodialysis through a central venous catheter at more than 250 outpatient dialysis clinics located across the United States.
Astellas Announces FDA Listing of DIGITIVA™ for the Management of Heart Failure
– New digital health solution for heart failure management puts patients at the center of their care by providing ability for at-home disease monitoring – TOKYO, Sept. 18, 2024 /PRNewswire/ — Astellas Pharma Inc. (TSE: 4503, President and CEO: Naoki Okamura, “Astellas”) today announced…
CARMAT Launches a Capital Increase for an Initial Amount of €10.3 Million to Finance Its Activities Until at Least the End of 2024
This capital increase will be carried out through a public offering without preferential subscription rights and with a 7 trading days priority period, on an irreducible and reducible basis, for the benefit of CARMAT’s shareholders The initial amount of the capital increase may be increased up to €11.8 million in […]
Edgewise Therapeutics to Host Webcast Event to Discuss Top-Line Data from Phase 1 trial in Healthy Subjects and Phase 2 CIRRUS-HCM Trial in Patients with Obstructive Hypertrophic Cardiomyopathy (HCM) on Thursday, September 19 at 8:30 am Eastern Time
BOULDER, Colo.–(BUSINESS WIRE)–Edgewise Therapeutics, Inc., (Nasdaq: EWTX), a leading muscle disease biopharmaceutical company, today announced that members of the management team will hold a live webcast to discuss top-line data of EDG-7500 from the Phase 1 trial in healthy subjects and the single-dose arm of the Phase 2 CIRRUS-HCM trial in […]
BioCardia Regains Full Compliance with Nasdaq Listing Requirements
SUNNYVALE, Calif., Sept. 18, 2024 (GLOBE NEWSWIRE) — BioCardia, Inc. [Nasdaq: BCDA], a global leader in cellular and cell-derived therapeutics for the treatment of cardiovascular and pulmonary diseases, announced today that the Company has regained full compliance with the Nasdaq Capital Market’s Listing Requirements as required by the Hearing Panel’s (the “Panel”) decision on May 13, 2024.
Elutia Announces New Peer Reviewed Publication Highlighting the Robustness of EluPro™, Company’s Antibiotic-Eluting BioEnvelope for Implantable Devices
EluPro eradicated bacteria commonly associated with cardiac implant-related infections in an established preclinical infection model
Edwards Launches SAPIEN 3 With Alterra Prestent in Europe for Transcatheter Pulmonic Valve Implantation
September 16, 2024 07:01 PM Eastern Daylight Time NYON, Switzerland–(BUSINESS WIRE)–Edwards Lifesciences today announced the launch in Europe of the SAPIEN 3 transcatheter pulmonary valve implantation (TPVI) system with Alterra adaptive prestent, expanding minimally invasive treatment options to a broader range of patients with congenital heart conditions. “I am proud […]
Novostia Announces New CEO and Secures Additional CHF 5.6 million in Funding
September 17, 2024 07:47 AM Eastern Daylight Time LAUSANNE, Switzerland–(BUSINESS WIRE)–Novostia, a pioneering medical technology company, specializing in the development of a disruptive heart valve prosthesis, at clinical stage, today announces a leadership transition alongside the successful completion of a CHF 5.6 million fund raise to further accelerate the company’s […]