DurAVR ViV restores similar aortic valve gradients to initial post-surgical results June 13, 2024 06:00 AM Eastern Daylight Time BRISBANE, Australia & EAGAN, Minn.–(BUSINESS WIRE)–Anteris Technologies Ltd (ASX: AVR), a structural heart company developing DurAVR™ THV, a new class of TAVR and the world’s only balloon-expandable, single-piece biomimetic aortic replacement […]
Other News
FDA clears Clario’s SpiroSphere® with wireless ECG, streamlining data collection in clinical trials
With the FDA 510(k) clearance of the COR-12 wireless ECG device now integrated with the SpiroSphere®, sponsors can conduct comprehensive respiratory trials with cardiac safety ECG collection on one device.
Clario’s SpiroSphere® integration with the wireless COR-12 is now FDA 510(k) cleared, allowing for consecutive collection of spirometry and ECG data during a single site visit.
The flexibility of the wireless COR-12 ECG enables integrated respiratory and cardiac safety trials for on-site, hybrid or remote trial design considerations.
The wireless feature of the SpiroSphere® ECG improves patient experience by removing the need for lead wires while maintaining high-quality data collection and accelerating trial timelines.
PHILADELPHIA, June 13, 2024 /PRNewswire/ — Clario, a leading provider of technologies and endpoint data solutions for clinical trials, today announced the U.S. Food and Drug Administration (FDA) 510(k) clearance for its SpiroSphere® with the wireless COR-12 Electrocardiogram (ECG) device. This technological advancement allows for the consecutive collection of spirometry and ECG data during a single site visit through Clario’s SpiroSphere® platform, consolidating all data into a single, unified database. This streamlines the clinical trial process and eliminates the need for separate ECG devices or multiple databases for a study.
Originally launched in 2018, the SpiroSphere®, a smart and lightweight pulmonary function testing (PFT) device, initially did not include ECG functionality. Now, the introduction of the SpiroSphere® ECG features the wireless COR-12 ECG device. This advancement enables customers to run integrated respiratory and cardiac safety trials on a single device and single database, simplifying the clinical trial process.
“The launch of our FDA-cleared SpiroSphere® wireless ECG device represents a significant step in improving clinical trial technology,” said Tom Stuckey, Senior Vice President, Respiratory & Precision Motion at Clario. “This clearance and product launch emphasizes our commitment to optimizing data collection, efficiency, and consistency, further enhancing how we deliver respiratory trials with cardiac safety.”
Ellen Street, Executive Vice President, Cardiac, Respiratory & Precision Motion at Clario added, “Our SpiroSphere® wireless ECG focuses on the site and patient experience while maintaining the high-quality data collection standards as our wired ECG model. With this wireless technology, site clinicians can now comfortably record both ECG and spirometry data in a single session on a single device, which can reduce the necessity for multiple devices, reduce burden, and accelerate trial timelines.”
This new product not only represents a technological advancement but also offers a cost-effective solution for enhanced efficiency and patient experience. For more information about the SpiroSphere® ECG and other Clario endpoint solutions, please visit Clario.com.
About ClarioClario is a leading healthcare research and technology company that generates high clinical evidence for our pharmaceutical, biotech, and medical device partners. We offer comprehensive evidence-generation solutions that combine eCOA, cardiac solutions, medical imaging, precision motion, and respiratory endpoints.
Since our founding more than 50 years ago, Clario has delivered deep scientific expertise and broad endpoint technologies to help transform lives around the world. Our endpoint data solutions have supported clinical trials over 26,000 times in more than 100 countries. Our global team of science, technology, and operational experts have supported over 60% of all FDA drug approvals since 2019.
For more information, go to Clario.com or follow us on LinkedIn.
Media Contact:Alexis NavratilCommunications Manager[email protected]
SOURCE Clario
Xeltis receives IDE approval from FDA for initiation of US pivotal trial for aXess™, its restorative vascular access conduit
aXess is a restorative arteriovenous dialysis conduit which allows vascular access for hemodialysis through the creation of a new, permanent, living vessel
EINDHOVEN, The Netherlands, June 13, 2024 /PRNewswire/ — Xeltis, a leading developer of transformative implants that enable the natural creation of living and long-lasting vessels, announces that it has gained approval from the US Food and Drug Administration (FDA) for an Investigational Device Exemption (IDE) submission to begin enrolling patients into a pivotal study for aXess.
aXess is a restorative conduit that enables the creation of a new, permanent, living vessel for hemodialysis vascular access, combining the safety and patency of an arteriovenous fistula (AVF), with the speed to treatment of an arteriovenous graft (AVG). The aXess vascular access conduit offers an improved dialysis patient experience and avoids the frequent reinterventions and complications, such as infections, faced by many renal disease patients.
Xeltis’ groundbreaking implants platform is an important development in vascular replacement technology as, over time, its implants are gradually replaced by the patients’ own living healthy tissue. Xeltis’ novel technology has already treated over 100 patients across different clinical trials.
Eliane Schutte, Chief Executive Officer, Xeltis commented: “We have already shown outstanding 12-month data from our first-in-human study in Europe and are looking forward to starting this pivotal trial in the US. We are very proud of the potential for aXess to transform the field of vascular access by stopping the cycle of interventions and infections and bringing our unique restorative solution to hemodialysis patients worldwide.”
Paulo Neves, Chief Medical Officer, Xeltis said: “Our focus is on improving the outcomes for patients on dialysis. aXess offers this potential through its avoidance of the reinterventions and complications associated with other vascular access solutions. This pivotal study is important in assessing and demonstrating this and marks a significant milestone in our clinical strategy in the US.”
The US-based pivotal trial follows strong 12-month data from the first-in-human trial in Europe in comparison to hemodialysis vascular access solutions. aXess is also undergoing an EU pivotal trial, recruiting up to 110 patients in nine EU countries.
About XeltisXeltis is a medtech company developing transformative implants that enable the natural creation of living and long-lasting vessels. Xeltis seeks to address the limitations of currently available options for the millions of people requiring hemodialysis access grafts or cardiovascular replacements every year. The Company’s proprietary endogenous tissue restoration (ETR) platform utilizes an advanced polymer implant which regenerates the patient’s own tissue before gradually being absorbed and leaving new, living, and long-lasting vessels in place. Xeltis’ most advanced product currently under clinical development is aXess™, an implantable blood vessel for hemodialysis vascular access. Xeltis’ groundbreaking technology has high potential to be applied to other major vascular and cardiovascular diseases.
Xeltis is based in The Netherlands and the USA. Its investors include DaVita Venture Group, EQT Life Sciences, Kurma Partners, VI Partners, Ysios Capital, Grand Pharma Group, the European Innovation Council and Invest-NL, in addition to other public and private investors.
About aXessaXess is a restorative conduit which enables the creation of a new, permanent, living vessel for hemodialysis vascular access. It combines the safety and patency of a fistula with the speed to treatment of an AV graft. The aXess vascular access conduit offers an improved dialysis patient experience and avoids the frequent reinterventions and complications, such as infections, faced by renal disease patients.
A first-in-human trial of aXess demonstrated a significant improvement in performance compared to hemodialysis vascular access solutions. A pivotal trial of aXess is currently enrolling up to 110 patients in nine EU countries.
Go to axesspivotal.com for more details.
SOURCE Xeltis
Simpson Interventions Announces Appointment of Dr. Joseph Knight as CEO
June 12, 2024 10:00 AM Eastern Daylight Time CAMPBELL, Calif.–(BUSINESS WIRE)–Simpson Interventions, Inc., a pioneering medical technology company specializing in cardiovascular interventional devices, today announced the appointment of Dr. Joseph Knight to the role of Chief Executive Officer, effective May 1st, 2024. He replaces Simpson Interventions’ Founder and previous CEO, […]
ABE Technology Introduces AI-Enhanced Cardiac Imaging System ‘CardioVision’
HONG KONG–(BUSINESS WIRE)–ABE Technology has announced the launch of its latest innovation, CardioVision, an AI-powered cardiac imaging system designed to improve the efficiency and accuracy of cardiac diagnostics. This new system integrates advanced artificial intelligence to streamline imaging processes and enhance patient care. CardioVision: Enhancing Cardiac Diagnostics CardioVision utilizes advanced […]
Positron Corporation Enters Agreement to Acquire FDA 510(k) for NeuSight PET-CT
Niagara Falls, NY, June 12, 2024 (GLOBE NEWSWIRE) — Positron Corporation (“Positron” or the “Company”) (OTC: POSC), a leading molecular imaging medical device company offering PET and PET-CT (Positron Emission Tomography/Computed Tomography) imaging systems and clinical support services, is pleased to announce it has entered an agreement to acquire the FDA 510(k) for the NeuSight PET-CT from its cooperative partner Neusoft Medical Systems’ subsidiary, Shenyang Intelligent Neuclear Medical Technology Co. A 510(k) is a premarket submission made to the FDA (U.S. Food and Drug Administration) to demonstrate that the device to be marketed is as safe and effective, that is, substantially equivalent, to a legally marketed device. Positron’s ownership of the existing 510(k) and FDA clearance will enable the marketing and sale of the NeuSight PET-CT 64 slice imaging system. This 510(k) also will facilitate future FDA clearances, including for the Company’s Affinity PET-CT 4D 64 slice system. This agreement allows Positron to immediately market and sell the NeuSight PET-CT, with plans to introduce Positron’s Affinity PET-CT 4D pending acceptance of an amended 510(k) submission. Adel Abdullah, President of Positron stated, “Gaining the 510(k) is a significant milestone in Positron’s mission to offer outstanding PET-CT technology and solutions to healthcare practices and hospitals serving both the cardiac and oncology segments of molecular imaging. Our PET imaging product line will now feature PET-CT which will expand access for physicians and practitioners seeking to utilize the full capabilities of the PET modality in nuclear cardiology and oncology. PET-CT imaging remains at the forefront of oncology studies and has become vital to the future of nuclear cardiology. With the addition of PET-CT, Positron has a range of core products that blend cutting edge technology with strong economics, appealing to most nuclear imaging specialist and practices.” Mr. Abdullah further added, “We appreciate the full support of Neusoft Medical Systems whose unwavering support and expertise in imaging technologies and extensive R&D continues to enhance our current and future capabilities. I am confident in Positron’s important role in advancing the PET modality and our commitment to delivering the best value in the industry.” About Positron Corporation Positron Corporation is a medical technology company that co-develops, manufactures, and sells state-of-the-art PET and PET-CT imaging systems and clinical services to nuclear medicine healthcare providers throughout North America. Positron specializes in the field of cardiac Positron Emission Tomography (PET) imaging, the gold standard in cardiac diagnostics. Positron’s innovative PET/PET-CT technologies, clinical services and practice solutions enables healthcare providers to accurately diagnose coronary artery disease and improve patient outcomes while practicing cost effective medicine. Positron’s Attrius® PET and NeuSight PET-CT imaging systems and distinct market position are substantial advantages unique to Positron that will facilitate the adoption of cardiac PET and the growth of the nuclear imaging market. Positron will soon offer a state-of-the-art PET-CT 4D molecular imaging device in the Affinity PET-CT 4D 64-Slice. Positron’s PET-CT(s) will enable nuclear cardiologists to utilize the full capabilities of molecular imaging and nuclear medicine. Positron’s PET-CT systems will also enable the Company to fully service and meet the demands of the vast oncology imaging segment of nuclear medicine. Positron is committed to expanding the cardiac and oncology PET modality by delivering the best technology and value to imaging specialists and will continue to advance its technology through its co-developer, supplier, and R&D venture with Shenyang Intelligent Neuclear Technology Co. a subsidiary of Neusoft Medical Systems. About Neusoft Medical Systems Co., Ltd. Neusoft Medical Systems is a leading global clinical diagnosis and treatment solution provider, headquartered in China. Neusoft Medical Systems is constantly innovating its portfolio of medical imaging diagnosis and clinical solutions in CT, MRI, DSA, XR, PET/CT, RT, US and IVD. Neusoft Medical Systems also is developing MDaaS (Medical Devices & Data as a Service), a strategic product line built using the Internet, big data, artificial intelligence, combined with other technologies improving medical institutions’ ability to diagnose and treat patients, achieving operational excellence. Innovation is always the driving force of Neusoft Medical Systems. Neusoft Medical Systems is collaborating with global scientists and medical institutions dedicated to advancing the technology of medical imaging solutions. Together with 45,000 installations in more than 110 countries, Neusoft Medical Systems provides advanced, high-quality medical imaging solutions to patients around the world. Neusoft Medical Systems is dedicated to being an excellent value innovator of global healthcare services. Through innovation and excellent operations, Neusoft Medical Systems is advancing healthcare products across a wide range of medical solutions and services to enhance global healthcare for all. Forward-Looking Statements This press release contains statements which may constitute “forward-looking statements” within the meaning of the Securities Act of 1933 and the Securities Exchange Act of 1934, as amended by the Private Securities Litigation Reform Act of 1995. Those statements include statements regarding the intent, belief or current expectations of Positron Corporation, and members of its management as well as the assumptions on which such statements are based. Words such as “anticipate,” “if,” “believe,” “plan,” “estimate,” “expect,” “intend,” “may,” “could,” “should,” “will,” and similar expressions are forward-looking statements and involve risks, uncertainties and contingencies, many of which are beyond our control, which may cause actual results, performance, or achievements to differ materially from anticipated results, performance, or achievements. Prospective investors are cautioned that any such forward-looking statements are not guarantees of future performance and involve risks and uncertainties, and that actual results may differ materially from those contemplated by such forward-looking statements. The Company undertakes no obligation to update or revise forward-looking statements to reflect changed assumptions, the occurrence of unanticipated events or changes to future operating results even if new information becomes available in the future. FOR FURTHER INFORMATION, please visit the company’s website at www.positron.com, or contact: investor@positron.com
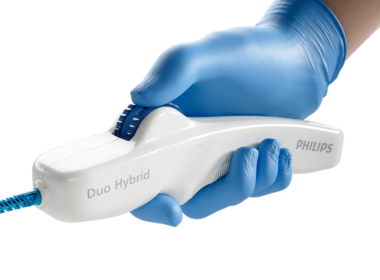
Philips launches Duo Venous Stent System for treatment of symptomatic venous outflow obstruction
June 12, 2024 Sanger Heart & Vascular Institute, Atrium Health, treated the first patient with the new implantable medical device following FDA premarket approval Amsterdam, the Netherlands – Royal Philips (NYSE: PHG, AEX: PHIA), a global leader in health technology, today announced the first implant of the Duo Venous Stent System, an implantable medical device indicated to treat symptomatic venous outflow obstruction in patients with chronic venous insufficiency (CVI), following premarket approval (PMA) from the U.S. Food and Drug Association (FDA). On June 11, Dr Erin Murphy – vascular surgeon and director of the Venous and Lymphatic Program at the renowned Sanger Heart & Vascular Institute, Atrium Health, in Charlotte, N.C., and an investigator in the VIVID study, which contributed to the device’s FDA approval – successfully used the Duo Venous Stent System for the first time outside of a clinical trial. Impacting 25 million people globally [1], deep venous disease results from venous thromboembolism, a condition that occurs when a blood clot forms in the vein [2]. It is the third most common cardiovascular disease [2]. Deep venous anatomy and obstructions can present a multitude of complexities and mechanical challenges. Engineered for the unique demands of venous anatomy and obstructions, the Duo Venous Stent System is comprised of two stents – Duo Hybrid and Duo Extend – of various sizes. Duo Hybrid has a distinct integrated design that combines multiple zones of differing mechanical properties into a single stent [3]. For long lesions, Duo Extend smoothly overlaps with the Duo Hybrid to extend therapy. These two stents are designed to work together and minimize the risk of stent fracture and corrosion, while providing an option to stent within caudal veins with smaller diameters [3]. “Duo is the first stent that offers a differential design for the challenges of venous anatomy – a focal area that withstands the forces of compression as well as the flexibility to accommodate curvature of the vessel,” said Dr Kush Desai, a highly regarded Interventional radiologist and associate professor of Radiology, Surgery and Medicine at Northwestern University in Chicago, as well as a leading enroller and investigator for the VIVID study. “Consequently, Philips is well positioned to support CVI treatment by offering a robust portfolio of medical technology that includes both intravascular ultrasound and a differentiated venous stenting system.” VIVID studyThe VIVID study is a global, prospective, multi-center, single-arm, non-blinded clinical trial conducted in the U.S. and Poland, evaluating the safety and efficacy of the Philips Duo Venous Stent System in the treatment of nonmalignant iliofemoral occlusive disease. It enrolled 162 subjects at 30 centers with three patient populations – non-thrombotic iliac vein lesion (NIVL), post-thrombotic syndrome (PTS) and acute deep vein thrombosis (aDVT). The VIVID study is now in 36-month follow-up and upon FDA PMA approval transitioned from an investigational device exemption (IDE) study to a post-approval study (PAS): NCT04580160. The VIVID study met all of its primary safety and efficacy performance goals. The 12-month effectiveness endpoint for primary patency reached 90.2%, which exceeded the performance target goal of 77.3%. The 12-month primary safety endpoint of 98.7% also exceeded the corresponding performance goal of 89%. In addition, quality of life and venous functional assessments that were performed in the VIVID study – including Clinical-Etiology-Anatomy-Pathophysiology (CEAP), Venous Clinical Severity Score (VCSS), Villalta, EQ-5D-3L and VEINES scores – showed sustained improvements compared to baseline at 12 months. “The VIVID study’s 12-month results demonstrate the safety and efficacy of the Duo Venous Stent System in the treatment of chronic venous insufficiency, a vascular condition affecting millions of people worldwide,” said principal investigator Dr Mahmood Razavi, M.D., an interventional radiologist with St. Joseph Vascular in Orange County, Calif. “Duo represents a meaningful addition to the tools that clinicians can use to treat CVI patients,” Dr Razavi added, “especially when used in conjunction with intravascular ultrasound, or IVUS. Ultimately, the new device promises to enable excellent clinical outcomes and drive significant quality of life improvements.” The VIVID study was the first clinical trial to mandate IVUS use to aid in lesion assessment and stent sizing prior to device implantation. According to prior published research, IVUS supports accurate diagnosis of venous disease and has been shown to change 57% of treatment plans compared to venography alone [4]. Led by Philips, intravascular imaging is used in more than 70% of venous stent procedures [5]. “The launch of the Duo Venous Stent System represents another step forward in achieving our aspiration to innovate interventional procedures with advanced medical technology,” said Heather Hudnut Page, Vice President and Business Leader of Peripheral Vascular at Philips. “In this context, we look forward to bringing the combined offering of intravascular ultrasound and Duo to the interdisciplinary teams – from vascular surgeons to interventional radiologists and interventional cardiologists – who share our overarching goal of enhancing patient care.” [1] Market Model Sources- DVD: 1 US Physician Quant Survey- Leveraged for NIVL prevalence assumption as ~25% of Symptomatic DVD 2 Thrombosis: a major contributor to the global disease burden. J Thromb Haemost 2014; 12: 1580–90. – Leveraged for DVT incidence in some countries (ex. EU5) 3 DRG VTE Epidemiology Reports- Leveraged for DVT incidence in some countries 4 Inari Medical presentation and Khan, SR, Arch Intern Med 2004- Leveraged for assumption of PTS.[2] Scheres LJJ, Lijfering WM, Cannegieter SC. Current and future burden of venous thrombosis: Not simply predictable. Res Pract Thromb Haemost. 2018 Apr 17;2(2):199-208. doi: 10.1002/rth2.12101. PMID: 30046722; PMCID: PMC6055567.[3] Data on file: D062749[4] Gagne PJ, Tahara RW, Fastabend CP, et al. Venography versus intravascular ultrasound for diagnosing and treating iliofemoral vein obstruction. J Vasc Surg Venous Lymphat Disord. 2017;5:678-687.[5] Divakaran S, Meissner MH, Kohi MP, et al. Utilization of and Outcomes Associated with Intravascular Ultrasound during Deep Venous Stent Placement among Medicare Beneficiaries. J Vasc Interv Radiol. 2022;33(12):1476-1484.e2. doi:10.1016/j.jvir.2022.08.018 Regulatory disclosures Developed by Vesper Medical, Inc., a wholly owned subsidiary of Philips, the Duo Venous Stent System is being marketed under the Philips brand. Its FDA approval order, along with related regulatory information, can be found here: PMA approval for Duo Venous Stent System. Caution: Federal law restricts this device to sale by or on the order of a licensed healthcare practitioner. For further information, please contact:Joost MalthaPhilips Global Press OfficeTel. : +31 (6) 1055 8116Email : joost.maltha@philips.com About Royal PhilipsRoyal Philips (NYSE: PHG, AEX: PHIA) is a leading health technology company focused on improving people’s health and well-being through meaningful innovation. Philips’ patient- and people-centric innovation leverages advanced technology and deep clinical and consumer insights to deliver personal health solutions for consumers and professional health solutions for healthcare providers and their patients in the hospital and the home. Headquartered in the Netherlands, the company is a leader in diagnostic imaging, ultrasound, image-guided therapy, monitoring and enterprise informatics, as well as in personal health. Philips generated 2023 sales of EUR 18.2 billion and employs approximately 69,100 employees with sales and services in more than 100 countries. News about Philips can be found at www.philips.com/newscenter.
Attachments
Duo Venous Stent System
Duo Venous Stent System 2
University Hospitals Transforms Patient Care with Aidoc’s AI-Enabled Solutions Across Its Entire Health System
Aidoc aiOS™ integration across 13 hospitals focuses on enhancing clinical outcomes and streamlining workflow with access to 17 FDA-cleared AI algorithmsCLEVELAND, June 12, 2024 /PRNewswire/ — University Hospitals (UH) has announced a major advancement in its AI strategy with the deployment of Aidoc’s comprehensive AI platform (aiOS™) across 13 of its hospitals and dozens of its outpatient locations. This initiative marks a significant leap forward in leveraging cutting-edge technology for superior patient outcomes.
Continue Reading
Image of University Hospitals Medical Center Campus in Cleveland, Oh. (PRNewsfoto/Aidoc)
University Hospitals Logo (PRNewsfoto/Aidoc)
Driven by a commitment to provide the highest standard of care, UH sought a standardized, hyper-accurate AI platform that could be seamlessly integrated across its network of academic hospitals and outpatient locations. Having utilized AI in various small-scale applications, UH chose to expand its capabilities with Aidoc to empower care teams with the information they need to address some of today’s most pressing medical issues.
Patients at UH will benefit from faster diagnosis and treatment of acute conditions such as pulmonary embolism, aortic dissection, vertebral compression fracture and pneumothorax. When a patient arrives at a UH facility and undergoes a CT scan for an injury or pain, Aidoc’s AI analyzes the scan using its comprehensive algorithms. The technology identifies both expected and unexpected findings, helps physicians prioritize urgent cases and ensures all flagged conditions are reviewed by the care team. Aidoc’s AI facilitates communication between the care team to enhance collaboration and speed up treatment.
Key Benefits of Aidoc for University Hospitals
Seamless integration across the entire health network
Immediate access to critical patient information
Expedited care and improved patient outcomes
Access to all 17 FDA-cleared AI algorithms for triage, quantification and care coordination
“Today’s care delivery is incredibly complex with numerous moving parts,” shared Dr. Donna Plecha, Chair of Radiology at University Hospitals.” Aidoc’s AI technology assists our radiologists in evaluating various patient images, allowing our clinicians to access precise, actionable data quickly. The AI technology enables our care teams to be more accurate and efficient leading to even more exceptional care for our patients.”The agreement leverages Aidoc’s proprietary aiOS™, a groundbreaking platform that addresses common AI implementation challenges such as integrating with existing IT systems, ensuring EHR compatibility, and managing data and reporting. This comprehensive approach ensures that UH can provide leading-edge care while maintaining operational efficiency and clinical excellence.”We are thrilled to collaborate with University Hospitals to drive healthcare innovation through our AI solutions,” said Elad Walach, CEO, Aidoc. “University Hospitals’ visionary approach to healthcare and dedication to integrating technology sets a new standard for patient care. UH is not only enhancing care coordination and streamlining workflows but also ensuring every patient receives the precise, timely and exceptional care they deserve. UH’s commitment to medical ingenuity and excellence is truly transformative.”By integrating Aidoc’s AI platform, University Hospitals ensures seamless deployment and minimal technical challenges, allowing for a collaborative approach to continuous monitoring and governance. This partnership not only enhances patient safety and care but also enables UH to maintain high standards in AI performance and reliability. Through this collaborative effort, UH can focus more on patient care, ensuring every patient receives the best possible treatment with the aid of AI technology.About University Hospitals Founded in 1866, University Hospitals serves the needs of patients through an integrated network of 21 hospitals (including five joint ventures), more than 50 health centers and outpatient facilities, and over 200 physician offices in 16 counties throughout northern Ohio. The system’s flagship quaternary care, academic medical center, University Hospitals Cleveland Medical Center, is affiliated with Case Western Reserve University School of Medicine, Northeast Ohio Medical University, Oxford University, the Technion Israel Institute of Technology and National Taiwan University College of Medicine. The main campus also includes the UH Rainbow Babies & Children’s Hospital, ranked among the top children’s hospitals in the nation; UH MacDonald Women’s Hospital, Ohio’s only hospital for women; and UH Seidman Cancer Center, part of the NCI-designated Case Comprehensive Cancer Center. UH is home to some of the most prestigious clinical and research programs in the nation, with more than 3,000 active clinical trials and research studies underway. UH Cleveland Medical Center is perennially among the highest performers in national ranking surveys, including “America’s Best Hospitals” from U.S. News & World Report. UH is also home to 19 Clinical Care Delivery and Research Institutes. UH is one of the largest employers in Northeast Ohio with more than 30,000 employees. Follow UH on LinkedIn, Facebook and Twitter. For more information, visit UHhospitals.org.About AidocAidoc is a pioneering force in clinical AI. We focus on aiding and empowering healthcare teams to optimize patient treatment, which results in improved economic value and clinical outcomes. Our clinically proven AI solutions eliminate silos, increase efficiencies, and improve outcomes by delivering critical information when and where care teams need it leading to immediate collective action. Built on Aidoc’s proprietary aiOS™, we analyze and aggregate medical data to enable care teams to operationalize the unexpected and work seamlessly with a continued focus on the patient. Used in more than 1,000 medical centers worldwide, Aidoc has the most FDA clearances (17) in clinical AI and its AI-based solutions cover 75 percent of patient populations, enabling physicians to make informed decisions based on real-time data. Aidoc AI is always on, running in the background to change the foreground. Visit Aidoc.com to see how we are connecting all points of care with always on AI.Photo – https://mma.prnewswire.com/media/2436134/Aidoc_UH.jpgLogo – https://mma.prnewswire.com/media/2015772/4756850/Aidoc_Always_On_AI_Logo.jpgLogo – https://mma.prnewswire.com/media/2436135/University_Hospitals_Logo.jpgSOURCE Aidoc
Milestone Pharmaceuticals to Host KOL Event for Investors “Learnings from the Field: Expert Perspectives on PSVT in the Community Setting” on June 20, 2024
MONTREAL and CHARLOTTE, N.C., June 12, 2024 (GLOBE NEWSWIRE) — Milestone Pharmaceuticals Inc. (Nasdaq: MIST), a biopharmaceutical company focused on the development and commercialization of innovative cardiovascular medicines, today announced that it will host Part I of its virtual investor educational series on Thursday, June 20, 2024, at 8:00 AM ET. To register for “Learnings from the Field: Expert Perspectives on Managing PSVT in the Community Setting,” click here. The event will feature George Mark, MD, FACC and Vivek Sailam, MD from The Heart House and Cooper University Health Care, who will discuss the burden of paroxysmal supraventricular tachycardia (PSVT) on patients and their practice, current treatments and unmet needs, and expectations for how the treatment landscape will evolve in coming years. A live question and answer session will follow the formal presentation. About George Mark, MD, FACC George Mark, MD, FACC is a cardiac electrophysiologist at The Heart House and Cooper University Health Care who specializes in treating rhythm disorders of the heart. His practice includes medical device implantation for the prevention of sudden cardiac death and cardiac ablation to treat abnormal heart rhythms. Dr. Mark graduated from Wesleyan University with a degree in Molecular Biology and Biochemistry and received his medical degree from Temple University. Dr. Mark remained at Temple for his residency in Internal Medicine, where he served for a year as Chief Medical Resident. Dr. Mark completed his fellowship training in Cardiovascular Diseases and Clinical Cardiac Electrophysiology at Thomas Jefferson University Hospital. About Vivek Sailam, MDVivek Sailam, MD is a clinical cardiologist at The Heart House and Cooper University Health Care with more than 16 years of experience. He completed his internal Medicine Residency at Hahnemann University and pursued additional training in cardiovascular disease. His dedication and expertise have been acknowledged with awards such as South Jersey Top Docs, Guardian Angel Award, and NJ Top Docs. Dr. Sailam has contributed to the medical field through publications in the NY Times 2021. Beyond medicine, Dr. Sailam is passionate about Community Health programs: “Walk with a Doc, Workout with a Doc, Eat with a Doc, Shop with a Doc,” nutrition, fitness, and racing automobiles, finding his balance and inspiration outside the clinic or hospital. About Paroxysmal Supraventricular Tachycardia An estimated two million people in the United States are currently diagnosed with PSVT which is a type of arrhythmia or abnormal heart rhythm. PSVT is characterized by episodes of sudden onset rapid heartbeats often exceeding 150 to 200 beats per minute. The heart rate spike is unpredictable and may last several hours. The rapid heart rate often causes disabling severe palpitations, shortness of breath, chest discomfort, dizziness or lightheadedness, and distress, forcing patients to limit their daily activities. The uncertainty of when an episode of PSVT will strike or how long it will persist can provoke anxiety in patients and negatively impact their day-to-day life between episodes. The impact and morbidity from an attack can be especially detrimental in patients with underlying cardiovascular or medical conditions, such as heart failure, obstructive coronary disease, or dehydration. Many health care providers are dissatisfied with the lack of effective treatment options with patients often requiring prolonged, burdensome, and costly trips to the emergency department or even invasive cardiac ablation procedures. About Milestone Pharmaceuticals Milestone Pharmaceuticals Inc. (Nasdaq: MIST) is a biopharmaceutical company developing and commercializing innovative cardiovascular solutions to improve the lives of people living with complex and life-altering heart conditions. The Company’s focus on understanding unmet patient needs and improving the patient experience has led us to develop new treatment approaches that provide patients with an active role in self-managing their care. Milestone’s lead investigational product is CARDAMYST (etripamil) nasal spray, a novel calcium channel blocker that is being studied for patients to self-administer without medical supervision to treat symptomatic episodic attacks associated with PSVT and AFib-RVR. Contact: Kim Fox, Vice President, Communications, kfox@milestonepharma.com Investor Relations Chris Calabrese, ccalabrese@lifesciadvisors.com Kevin Gardner, kgardner@lifesciadvisors.com Source: Milestone Pharmaceuticals Inc.
Kardium Announces $104M in New Financing for Innovative Atrial Fibrillation Treatment
VANCOUVER, British Columbia–(BUSINESS WIRE)–Kardium Inc., developer of the Globe® Mapping and Ablation System for the treatment of atrial fibrillation, has raised US $104 million in a new financing round. The round is led by existing investor Fidelity Management & Research Company, together with follow-on participation by funds and accounts advised by T. Rowe […]