NEW YORK, Feb. 5, 2024 /PRNewswire/ — Acarix, a leader in advanced acoustic-based cardiac diagnostic devices today announces the CADScor® System has been added to the United States Federal Supply Schedule (FSS). The nationwide contract simplifies purchasing for the Veterans Health…
Other News
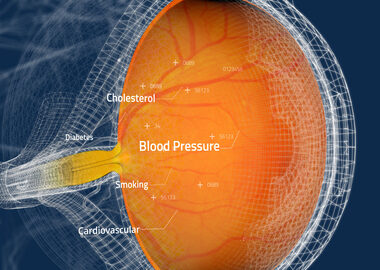
Toku Obtains CE and UKCA Marks for AI Cardiovascular Risk Assessments Through the Eye
SAN DIEGO–(BUSINESS WIRE)–Toku, Inc., a commercial medical device company specializing in AI assessment of retinal images, announced today that it has obtained CE and UKCA Marks for its patented CLAiR technology. This significant milestone underscores Toku’s commitment to maintaining the highest standards of quality and safety and allows access to […]
8 Best WordPress Training Courses for Beginners in 2024
WordPress is a powerful content management system (CMS) that pretty much anyone can use to make a stunning, custom website. You don’t even have to know how to code to use it. But, if you’ve never used the platform before, there’s a bit of a learning curve. If that’s exactly […]
8 Best WordPress Training Courses for Beginners in 2024
WordPress is a powerful content management system (CMS) that pretty much anyone can use to make a stunning, custom website. But, if you’ve never used the platform before, there’s a bit of a learning curve. In this post, we’re going to share eight of the best WordPress training courses for […]
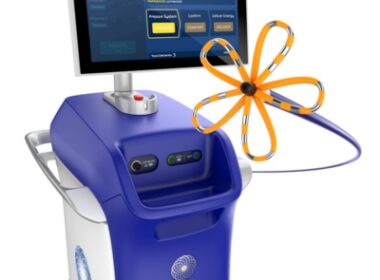
Boston Scientific Receives FDA Approval for FARAPULSE™ Pulsed Field Ablation System
MARLBOROUGH, Mass., Jan. 31, 2024 /PRNewswire/ — Boston Scientific Corporation (NYSE: BSX) announced it has received U.S. Food and Drug Administration (FDA) approval for the FARAPULSE™ Pulsed Field Ablation (PFA) System. The FARAPULSE PFA System is indicated for the isolation of pulmonary veins in the treatment of drug-refractory, recurrent, symptomatic, paroxysmal (i.e., […]
Edwards’ EVOQUE Valve Replacement System First Transcatheter Therapy to Earn FDA Approval for Tricuspid Valve
IRVINE, Calif.–(BUSINESS WIRE)– Edwards Lifesciences Corporation (NYSE: EW) today announced the company’s EVOQUE tricuspid valve replacement system is the first transcatheter therapy to receive U.S. Food and Drug Administration (FDA) approval for the treatment of tricuspid regurgitation (TR). The EVOQUE system is indicated for the improvement of health status in patients […]
Complete Tutorial: How to Build a Membership Site on WordPress
Creating a membership site on WordPress can be valuable to your new or existing business. The site may serve to offer online courses and exclusive content or simply to build an online community. Regardless of your budget, you can build a membership site without spending too much. All you need […]
Sirius Therapeutics Begins Phase 1 Clinical Trial of Next-Generation, Long-Acting Factor XI siRNA Anticoagulant for Treatment of Thromboembolic Disorders
SAN DIEGO & SHANGHAI–(BUSINESS WIRE)– Sirius Therapeutics, announced it has dosed the first subject in a Phase 1, first-in-human clinical trial in Australia of SRSD107 on January 30th 2024, its next generation siRNA therapeutic targeting coagulation Factor XI (FXI), for the prevention and treatment of thromboembolic disorders. SRSD107 is the second clinical […]
Artivion Announces Release Date and Teleconference Call Details for Fourth Quarter and Full Year 2023 Financial Results
ATLANTA, Feb. 1, 2024 /PRNewswire/ — Artivion, Inc. (NYSE: AORT), a leading cardiac and vascular surgery company focused on aortic disease, announced today that fourth quarter and full year 2023 financial results will be released on Thursday, February 15, 2024 after the market closes. On…
Surmodics Reports First Quarter of Fiscal Year 2024 Financial Results; Updates Fiscal Year 2024 Financial Guidance
EDEN PRAIRIE, Minn.–(BUSINESS WIRE)–Surmodics, Inc. (Nasdaq: SRDX), a leading provider of medical device and in vitro diagnostic technologies to the healthcare industry, today reported financial results for its first quarter ended December 31, 2023, and updated its financial guidance for fiscal year ending September 30, 2024. “We delivered total revenue […]