MADRID, Jan. 28, 2026 /PRNewswire/ — The ReDS-SAFE HF II Trial, a large multicenter investigator-initiated randomized controlled study, funded by both the Instituto de Salud Carlos III and the Spanish Society of Cardiology, has officially commenced. The trial is designed to further…
Other News
Corify Care Introduces First Whole-Heart Mapping Technology, Delivering a Global View of Cardiac Arrhythmias in a Single Beat
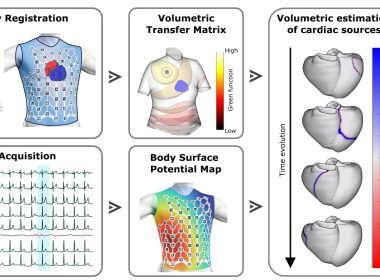
MADRID, Jan. 28, 2026 /PRNewswire/ — Corify Care today announced a major breakthrough in cardiac electrophysiology with the publication of its proprietary Global Volumetric Mapping technology in Nature Communications Medicine. The publication marks the first system capable of mapping all…
Pharyngeal Electrical Stimulation Recognized as Important Tool for Post-Stroke Recovery in New American Heart Association and American Stroke Association Guideline
Inclusion in new Acute Ischemic Stroke guidelines underscores clinical utility of Phagenyx for accelerating recovery from swallowing difficulties (dysphagia) in stroke patients NASHVILLE, Tenn., Jan. 27, 2026 /PRNewswire/ — Phagenesis, a commercial-stage company with a first-of-its-kind…
New Scientific Statement Guides Clinicians on How to Integrate Digital Health Tools into Heart Failure Care
WASHINGTON, Jan. 27, 2026 /PRNewswire/ — A new scientific statement provides clinicians with practical guidance on how to integrate digital health tools into everyday heart failure care – moving beyond isolated devices toward coordinated, team-based, and actionable systems of care….
Da Vinci 5 Cleared for Cardiac Procedures
FDA 510(k) clearance expands da Vinci 5 indications to include certain cardiac procedures FDA 510(k) clearance expands da Vinci 5 indications to include certain cardiac procedures
Milestone Pharmaceuticals Announces U.S. Availability of CARDAMYST™ (etripamil) Nasal Spray, the First and Only FDA-Approved Self-Administered Treatment for Adults with Paroxysmal Supraventricular Tachycardia (PSVT)
CARDAMYST is now conveniently available to patients through retail pharmaciesNational sales force hired with promotional launch in mid-FebruaryExpected $25 copay cap for eligible commercially insured patientsLaunch supported by comprehensive patient assistance program offering benefits verification and reimbursement support MONTREAL and CHARLOTTE, N.C., Jan. 26, 2026 (GLOBE NEWSWIRE) — Milestone Pharmaceuticals Inc. (Nasdaq: MIST) today announced that CARDAMYST™ (etripamil) nasal spray, its first commercial product, is now available through U.S. retail pharmacies. CARDAMYST is a prescription medication indicated for the conversion of acute symptomatic episodes of paroxysmal supraventricular tachycardia (PSVT) to sinus rhythm in adults. Implementation of a rapid commercialization plan is underway with a national sales force to be deployed in mid-February 2026. CARDAMYST will launch with the support of a patient assistance platform as Milestone continues to negotiate with insurers for formulary placement and coverage. The program provides benefits verification, reimbursement support, and copay assistance for most patients with commercial insurance that covers CARDAMYST, helping to reduce financial and administrative barriers to treatment. For eligible commercially insured patients, copays are expected to be capped at $25, reinforcing Milestone’s commitment to making CARDAMYST affordable for patients who need it. “Many patients have been waiting for help to take greater control of their PSVT condition. Now that CARDAMYST is available, patients have access to a convenient, self-administered option designed to treat episodes of PSVT wherever and whenever they occur,” said Joseph Oliveto, President and Chief Executive Officer of Milestone Pharmaceuticals. “Beyond availability, our team continues to engage insurers with the goal of having CARDAMYST widely covered and affordable.” The U.S. Food and Drug Administration (FDA) approved CARDAMYST on December 12, 2025, marking the first FDA approval of a treatment for PSVT in more than 30 years. About Paroxysmal Supraventricular Tachycardia (PSVT)An estimated two million people in the United States are currently diagnosed with PSVT, which is a type of arrhythmia or abnormal heart rhythm. PSVT is characterized by episodes of sudden onset rapid heartbeats often exceeding 150 to 200 beats per minute. The heart rate spike is unpredictable and may last several hours. The rapid heart rate often causes disabling severe palpitations, shortness of breath, chest discomfort, dizziness or lightheadedness, and distress, forcing patients to limit their daily activities. The uncertainty of when an episode of PSVT will strike or how long it will persist can provoke anxiety in patients and negatively impact their day-to-day life between episodes. The impact and morbidity from an attack can be especially detrimental in patients with underlying cardiovascular or medical conditions, such as heart failure, obstructive coronary disease, or dehydration. Many health care providers are dissatisfied with the lack of effective treatment options, with patients often electing to pursue prolonged, burdensome, and costly trips to the emergency department or even undergo invasive cardiac ablation procedures. About CARDAMYSTCARDAMYST™ (etripamil) nasal spray is approved by the U.S. Food and Drug Administration (FDA) for the conversion of acute symptomatic episodes of paroxysmal supraventricular tachycardia (PSVT) to sinus rhythm in adults. It is a novel calcium channel blocker nasal spray designed as a self-administered rapid response therapy for patients, thereby bypassing the need for immediate medical oversight. The product is intended to provide health care providers with a new treatment option to enable on-demand care and patient self-management. This portable treatment may provide patients with active management and a greater sense of control over their condition. CARDAMYST is well studied with a robust clinical trial program that includes a completed Phase 3 clinical-stage program for the treatment of PSVT. Currently, etripamil is in Phase 2 development for treatment of PSVT in pediatric patients and Phase 3 development for acute treatment of atrial fibrillation with rapid ventricular rate (AFib-RVR) in adults. For more information, please visit CARDAMYST.com. IndicationCARDAMYST is indicated for the conversion of acute symptomatic episodes of paroxysmal supraventricular tachycardia (PSVT) to sinus rhythm in adults. IMPORTANT SAFETY INFORMATION FOR CARDAMYST (etripamil) What is CARDAMYST?CARDAMYST is a prescription medicine used to help restore normal sinus heart rhythm in adults who have symptoms of sudden episodes of fast heartbeat called paroxysmal supraventricular tachycardia (PSVT). It is not known if CARDAMYST is safe and effective in children. Do not use CARDAMYST if you: are allergic to CARDAMYST or any of its ingredients. See the Patient Information for a complete list of ingredients in CARDAMYST.have limitations in activities due to heart failure (moderate to severe heart failure).have Wolff-Parkinson-White (WPW) syndrome, Lown-Ganong-Levine syndrome, or an abnormal heart rhythm pattern called pre-excitation (delta wave) on an electrocardiogram (ECG).have sick sinus syndrome without a permanent pacemaker.have second degree or higher atrioventricular (AV) block. Before using CARDAMYST, tell your healthcare provider about all of your medical conditions, including if you: have a history of fainting.have low blood pressure.are pregnant or plan to become pregnant. It is not known if CARDAMYST will harm your unborn baby.are breastfeeding or plan to breastfeed. It is not known if CARDAMYST passes into your breast milk. You should stop breastfeeding for 12 hours after treatment with CARDAMYST. During this time, pump and throw away your breast milk. Talk to your healthcare provider about the best way to feed your baby after using CARDAMYST. Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. What are the possible side effects of CARDAMYST? CARDAMYST may cause serious side effects, including: Fainting due to CARDAMYST effects on blood pressure, heart rate, and electrical activity of the heart. CARDAMYST may cause dizziness and fainting, especially in people with a history of fainting and certain heart problems, or people with a history of fainting during an episode of PSVT. Use CARDAMYST while sitting in a safe area where you will not fall if you become dizzy or lightheaded. Lie down if you feel dizzy or lightheaded after using CARDAMYST. If fainting occurs after using CARDAMYST, caregivers should place you on your back and seek medical help. The most common side effects of CARDAMYST include: nasal discomfortnasal congestionrunny nose throat irritationnosebleed These are not all of the possible side effects for CARDAMYST. Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.Please see the full Prescribing Information https://milestonepharma.com/etripamilprescribinginformation.pdf for CARDAMYST. About Milestone PharmaceuticalsMilestone Pharmaceuticals Inc. (Nasdaq: MIST) is a biopharmaceutical company developing and commercializing innovative cardiovascular medicines to benefit people living with certain heart conditions. Milestone’s lead product is CARDAMYST™ (etripamil) nasal spray, a novel calcium channel blocker, which is FDA approved for the conversion of acute symptomatic episodes of paroxysmal supraventricular tachycardia (PSVT) to sinus rhythm in adults. Etripamil is also in development for the treatment of symptomatic episodic attacks associated with AFib-RVR. Cautionary Note on Forward-Looking StatementsThis press release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. Words such as “believe,” “continue,” “could,” “demonstrate,” “designed,” “develop,” “estimate,” “expect,” “may,” “pending,” “plan,” “potential,” “progress,” “will”, “intend” and similar expressions (as well as other words or expressions referencing future events, conditions, or circumstances) are intended to identify forward-looking statements. These forward-looking statements are based on Milestone’s expectations and assumptions as of the date of this press release. Each of these forward-looking statements involves risks and uncertainties. Actual results may differ materially from these forward-looking statements. Forward-looking statements contained in this press release include statements regarding: the retail availability of CARDAMYST; Milestone’s expectations regarding the promotional launch of CARDAMYST; expectations in regards to etripamil’s efficacy; Milestone’s ability to make CARDAMYST widely covered and affordable, including any applicable copay caps; and other statements not related to historical facts. Important factors that could cause actual results to differ materially from those in the forward-looking statements include, but are not limited to, whether Milestone’s future interactions with the EMA will have satisfactory outcomes; whether and when, if at all, Milestone’s MMA for etripamil will be approved by the EMA; uncertainties related to the timing of initiation, enrollment, completion, evaluation and results of Milestone’s clinical trials; risks and uncertainty related to the complexity inherent in cleaning, verifying and analyzing trial data; and whether the clinical trials will validate the safety and efficacy of etripamil for PSVT or other indications, among others, general economic, political, and market conditions, including deteriorating market conditions due to investor concerns regarding inflation, international tariffs, Russian hostilities in Ukraine and ongoing disputes in the Middle East and overall fluctuations in the financial markets in the United States and abroad, risks related to pandemics and public health emergencies, and risks related the sufficiency of Milestone’s capital resources and its ability to raise additional capital in the current economic climate. These and other risks are set forth in Milestone’s filings with the U.S. Securities and Exchange Commission (SEC), including in its annual report on Form 10-K for the year ended December 31, 2025 and its quarterly report on Form 10-Q for the quarter ended September 30, 2025, in each case under the caption “Risk Factors,” as such discussions may be updated from time to time by subsequent filings Milestone may make with the SEC. Except as required by law, Milestone assumes no obligation to update any forward-looking statements contained herein to reflect any change in expectations, even as new information becomes available. Contact: Investor Relations Kevin Gardner, kgardner@lifesciadvisors.com Media Relations Rebecca Novak, rnovak@milestonepharma.com A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/55ffc562-1e02-4b4f-8ace-822b39d104f4
Concept Medical Announces First Patient Enrollment in STARS DAPT Randomized Clinical Trial Evaluating Abluminus NP Polymer-Free DES
TAMPA, Fla.–(BUSINESS WIRE)–Concept Medical Inc., a global pioneer in innovative drug delivery technology, is proud to announce the successful enrollment of the first patients in the STARS DAPT (ST-Segment Elevation Myocardial InfArction treated with a Polymer-Free Sirolimus-based NanocarrieR Eluting Stent and a P2Y12 inhibitor-based Single Antiplatelet Strategy after a Short Dual Antiplatelet Therapy […]
Affera™ momentum continues as Medtronic announces CE Mark in Europe and U.S. IDE first cases for Sphere-360™ PFA catheter to treat paroxysmal atrial fibrillation
First-of-its-kind, rotation-free single-shot PFA catheter supported by strong safety and efficacy data adds to the groundbreaking Affera family of technologies in Europe Successful completion of first cases at multiple sites kicks off Horizon 360 IDE pivotal trial in the United States…
CVRx Announces Initiation of BENEFIT-HF, a Landmark Heart Failure Trial Evaluating Barostim in Significantly Expanded Population
Study is Expected to Be One of the Largest Therapeutic Cardiac Device Trials in Heart Failure Ever Performed, and is Supported by CMS Category B IDE CoverageMINNEAPOLIS, Jan. 22, 2026 (GLOBE NEWSWIRE) — CVRx, Inc. (NASDAQ: CVRX), a medical device company focused on developing, manufacturing and commercializing innovative neuromodulation solutions for patients with cardiovascular diseases, today announced initiation of the BENEFIT-HF clinical trial, a landmark randomized study supported by CMS Category B IDE coverage and designed to evaluate the impact of Barostim™ in a significantly expanded heart failure population. Barostim™ is an implantable device that delivers electrical signals to baroreceptors located on the carotid artery, increasing baroreflex signaling, rebalancing the autonomic nervous system, and improving heart failure symptoms. Barostim™ received FDA approval in 2019 following its designation under the FDA’s Breakthrough Devices Program and is now commercially available in both the U.S. and Europe. The BENEFIT-HF trial is expected to be one of the largest therapeutic cardiac device trials ever performed in heart failure, randomizing 2,500 patients in approximately 150 centers in the United States and Germany. The primary endpoint will be a composite of all-cause mortality and heart failure decompensation events. Enrollment is expected to begin in the first half of 2026. The BENEFIT-HF trial is designed to evaluate Barostim in patients with heart failure who: Remain symptomatic after receiving optimized guideline-directed medical and device therapies (GDMT)Have a left ventricular ejection fraction (LVEF)
Anteris Announces Strategic Investment from Medtronic to Continue Advancing TAVR in $320 Million Aggregate Capital Raises
MINNEAPOLIS and BRISBANE, Australia, Jan. 22, 2026 (GLOBE NEWSWIRE) — Anteris Technologies Global Corp. (Anteris or the Company) (NASDAQ: AVR, ASX: AVR) a global structural heart company committed to designing, developing, and commercializing cutting-edge medical devices to restore healthy heart function, announced today it has completed a $90 million strategic investment from Medtronic, plc (Galway, Ireland) (NASDAQ: MDT) (Medtronic), the world’s largest medical technology company. The investment closed immediately following Anteris’ underwritten offering of common stock, for a combined $320 million in gross proceeds, which supports execution of the global pivotal PARADIGM trial and advances the Company toward global commercialization of the DurAVR® Transcatheter Heart Valve (THV). Anteris and Medtronic are aligned around the belief that reshaping the Transcatheter Aortic Valve Replacement (TAVR) market requires advancing clinical science and valve design while maintaining rigorous standards for durability, hemodynamics, and long‑term patient outcomes. “This strategic investment, along with our underwritten offering of common stock, represent an important milestone for our company. It also provides strong validation of our program from the capital markets and a major strategic innovator,” said Wayne Paterson, Anteris Vice Chairman and CEO. “The investment is one aspect of a collaboration that may expand into other strategic areas in the future. Anteris has developed a clinically important, evidence-supported product designed to improve the lives of patients with aortic stenosis as we advance toward regulatory approval.” “The Board is excited to welcome Medtronic to its stockholder community,” said John Seaberg, Chairman of the Anteris Board. “This investment aligns two organizations in their commitment to advancing valve science in a way that is disciplined and focused on long‑term patient benefit.” “Medtronic is a pioneer in TAVR innovation and evidence,” said Jorie Soskin, Vice President and General Manager of the Structural Heart business in Medtronic’s Cardiovascular portfolio. “Our investment in differentiated innovation like the DurAVR® THV technology — which has the potential to offer improved valve performance in a balloon-expandable platform — is core to our commitment to define and drive the future of TAVR, meeting the needs of more aortic stenosis patients and heart teams with a comprehensive portfolio.” The Medtronic investment and the underwritten public offering of common stock position Anteris to continue to execute the global PARADIGM Trial and to continue investing in research that can transform the treatment of aortic stenosis. Wells Fargo Securities acted as sole placement agent in connection with the Medtronic investment. About the PARADIGM Trial The PARADIGM Trial is a prospective randomized controlled trial (RCT) which will evaluate the safety and effectiveness of the DurAVR® THV compared to commercially available transcatheter aortic valve replacements (TAVRs). This head-to-head study will enroll approximately 1000 patients in the ‘All Comers Randomized Cohort’ with 1:1 randomization of patients who will receive either the DurAVR® THV or TAVR using commercially available and approved THVs. The PARADIGM Trial will assess non-inferiority on a primary composite endpoint of all-cause mortality, all stroke and cardiovascular hospitalization at one year post procedure. The PARADIGM Trial has commenced recruitment with the first patients enrolled and implanted during the fourth quarter of 2025. For further information, please refer to ClinicalTrials.gov (ClinicaTrials.gov ID NCT07194265). The planned expansion across other geographies includes additional cohorts. About Anteris Anteris Technologies Global Corp. (NASDAQ: AVR, ASX: AVR) is a global structural heart company committed to designing, developing, and commercializing cutting-edge medical devices to restore healthy heart function. Founded in Australia, with a significant presence in Minneapolis, USA, Anteris is a science-driven company with an experienced team of multidisciplinary professionals delivering restorative solutions to structural heart disease patients. Anteris’ lead product, the DurAVR® Transcatheter Heart Valve (THV), was designed in partnership with the world’s leading interventional cardiologists and cardiac surgeons to treat aortic stenosis – a potentially life-threatening condition resulting from the narrowing of the aortic valve. The balloon-expandable DurAVR® THV is the first biomimetic valve, which is shaped to mimic the performance of a healthy human aortic valve and aims to replicate normal aortic blood flow. DurAVR® THV is made using a single piece of molded ADAPT® tissue, Anteris’ patented anti-calcification tissue technology. ADAPT® tissue, which is FDA-cleared, has been used clinically for over 10 years and distributed for use in over 55,000 patients worldwide. The DurAVR® THV System is comprised of the DurAVR® valve, the ADAPT® tissue, and the balloon-expandable ComASUR® Delivery System. Forward-Looking Statements This announcement contains forward-looking statements, including statements in the quotations contained herein and regarding the use of proceeds from the Private Placement and the underwritten public offering, future collaboration between the company, the PARADIGM Trial, the potential for approval of DurAVR® THV and its ability to improve the lives of patients with aortic stenosis, and the testing of the DurAVR® THV technology. Forward-looking statements include all statements that are not historical facts. Forward-looking statements generally are identified by the words “believe,” “project,” “expect,” “anticipate,” “estimate,” “intend,” “budget,” “target,” “aim,” “strategy,” “plan,” “guidance,” “outlook,” “may,” “should,” “could,” “will,” “would,” “will be,” “will continue,” “will likely result” and similar expressions, although not all forward-looking statements contain these identifying words. These forward-looking statements are subject to a number of risks, uncertainties, and assumptions, including those described under “Risk Factors” in Anteris’ Annual Report on Form 10-K for the fiscal period ended December 31, 2024 and Quarterly Report on Form 10-Q for the period ended September 30, 2025, each of which was filed with the SEC and ASX. Readers are cautioned not to put undue reliance on forward-looking statements, and except as required by law, Anteris does not assume any obligation to update any of these forward-looking statements to conform these statements to actual results or revised expectations. For more information: Investor Relations Investor Relations (US)investor@anteristech.com mchatterjee@bplifescience.comDebbie Ormsby Malini Chatterjee, Ph.D.Anteris Technologies Global Corp. Blueprint Life Science Group+61 1300 550 310 | +61 7 3152 3200 +1 917 330 4269 Websitewww.anteristech.com X@AnterisTech LinkedInhttps://www.linkedin.com/company/anteristech