Company advanced its pipeline, launched new products, won share, and delivered earnings growth despite COVID-19 impact on healthcare procedure volumes DUBLIN, Feb. 22, 2022 /PRNewswire/ — Medtronic plc (NYSE: MDT) today announced financial results for its third quarter of fiscal year 2022, which ended January 28, 2022. Key Highlights Revenue of $7.8 billion was flat year-over-year […]
Other News
MicroVention Announces First U.S. Patient Treated with FRED™ X™ Flow Diverter Featuring X Technology
ALISO VIEJO, Calif., Feb. 22, 2022 /PRNewswire/ — MicroVention, Inc., a subsidiary of Terumo and a global neurovascular company, announced the first U.S. clinical case of its next generation Flow Diverter, the FRED X device, at Thomas Jefferson University Hospital located in Philadelphia. MicroVention received FDA PMA approval for the FRED X device in September […]
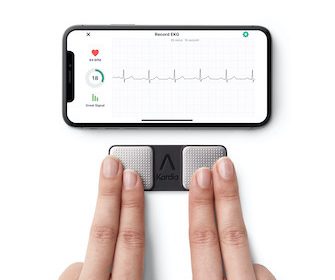
TLV recommends AliveCor’s smartphone-based ECG as a more cost-effective option for the detection of atrial fibrillation compared to Holter ECG
Sweden’s Dental and Pharmaceutical Benefits Agency, TLV evaluated the combination of the KardiaMobile® and KardiaPro personal electrocardiogram (ECG) and software compared to the current standard of care, Holter ECG The health economic assessment found a higher degree of atrial fibrillation (AF) detection and resource-saving with KardiaMobile compared to Holter ECG 24 hours1 KardiaMobile […]
Esperion Reports Fourth Quarter and Full Year 2021 Financial Results and Provides Company Update
– Unprecedented CLEAR Outcomes Study Achieved 90% MACE Accumulation in February 2022 – – U.S. Net Product Revenue of NEXLETOL® (bempedoic acid) Tablets and NEXLIZET® (bempedoic acid and ezetimibe) Tablets Grew 12% Sequentially to $12.2 Million in the Fourth Quarter 2021 and Over 200% for the Full Year 2021 – – Prescriptions Grew 9% […]
Claritas Announces Receipt of Comments from Australian Ethics Committee Regarding the Company’s Phase 1 Clinical Study of R-107 and Closing of Financing
SAN FRANCISCO, CA and TORONTO, ON , Feb. 22, 2022 (GLOBE NEWSWIRE) — Claritas Pharmaceuticals, Inc. (TSX VENTURE: CLAS and OTC: KALTF) (the “Company” or “Claritas“) today announced that it has received comments from the Australian Human Research Ethics Committee (the “HREC”) regarding the Company’s Phase 1 clinical study of R-107, and […]
Silence Therapeutics Appoints Craig Tooman President and Chief Executive Officer
Mark Rothera steps down as President and CEO LONDON–(BUSINESS WIRE)–Silence Therapeutics plc, Nasdaq: SLN (“Silence” or “the Company”), a leader in the discovery, development and delivery of novel short interfering ribonucleic acid (“siRNA”) therapeutics for the treatment of diseases with significant unmet medical need, today announced that Craig Tooman, previously […]
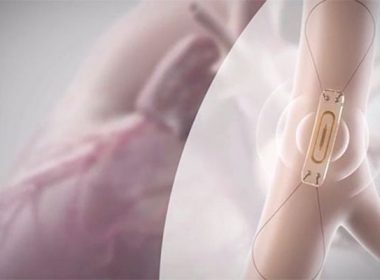
Abbott’s CardioMEMS™ HF System Receives FDA Approval to Support Patients Battling Earlier-Stage Heart Failure
– New expanded indication provides patients suffering from earlier stages of heart failure access to the CardioMEMS™ HF System, a small implantable sensor that can flag early warning signs of worsening heart failure – FDA approval was supported by data from the GUIDE-HF trial, which suggested that the CardioMEMS sensor […]
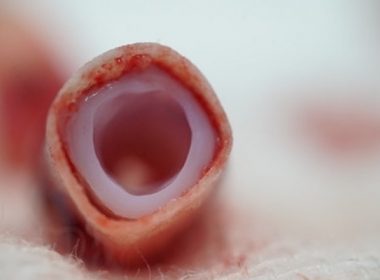
Revotek Receives Clearance from the National Health Commission of China for the World First Clinical Trial for 3D Bioprinted Stem Cell Vascular Grafts
CHENGDU, China, Feb. 21, 2022 /PRNewswire/ — Revotek announced that it has received clearance from the National Health Commission of China to begin a clinical study in the West China Hospital, Chengdu, with its first stem cell 3D bioprinting product, REVOVAS. “The clearance marks an important milestone for Revotek. We are excited to advance REVOVAS, our […]
Medtronic receives FDA expanded approval for cardiac cryoablation catheters for pediatric treatment of a common heart rhythm condition
Family of focal ablation catheters are the only approved option to treat specific type of supraventricular tachycardia (SVT) DUBLIN, Feb. 18, 2022 /PRNewswire/ — Medtronic plc (NYSE:MDT), a global leader in healthcare technology, today announced that the Freezor™ and Freezor™ Xtra Cardiac Cryoablation Catheters are approved by the U.S. Food and Drug Administration […]
Breakthrough Study Uses Genetic Testing to Evaluate Risk of Heart Disease
PHOENIX, Feb. 17, 2022 /PRNewswire/ — Dignity Health in Arizona is launching the first research study in North America in which genetic testing is used to identify men and women at risk of developing heart disease based on the makeup of their DNA. If proven effective in clinical trials, this form of genetic testing may be adopted globally to prevent […]