— Completed business combination with Alpha Healthcare Acquisition Corp. and raised $242M in proceeds — — Progressed advanced-stage clinical- and early-stage programs of the human acellular vessel (HAV™) and other engineered tissue platform applications — DURHAM, N.C., Nov. 12, 2021 (GLOBE NEWSWIRE) — Humacyte, Inc. (Nasdaq: HUMA), a clinical-stage biotechnology […]
Other News
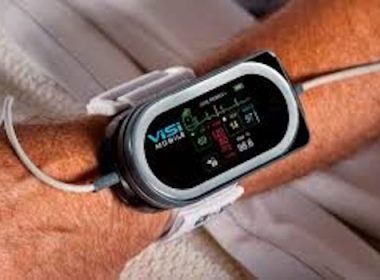
ViSi Mobile Chosen As Remote Monitoring Solution For Busy Emergency Department
Sotera Wireless, Inc., a company specializing in healthcare technology, has announced that their remote patient monitoring system was successfully implemented in an emergency department. San Diego, United States, Nov. 11, 2021 (GLOBE NEWSWIRE) — The ViSi Mobile System from San Diego-based Sotera Wireless was recently implemented to help negate overcrowding […]
Acutus Medical Reports Third Quarter 2021 Financial Results
CARLSBAD, Calif., Nov. 11, 2021 (GLOBE NEWSWIRE) — Acutus Medical, Inc. (“Acutus” or the “Company”) (Nasdaq: AFIB), an arrhythmia management company focused on improving the way cardiac arrhythmias are diagnosed and treated, today reported results for the third quarter of 2021. Recent Highlights: Significant progress was achieved in our three […]
Investor Insights: Tom Hertzberg, Managing Director, Lifesciences and Healthcare, Silicon Valley Bank
Investor Insights highlights some of the top investors in Life Science and MedTech. This week host Ken Dropiewski interviews Tom Hertzberg, Managing Director, Lifesciences and Healthcare, Silicon Valley BanK https://vimeo.com/626976813
NEW RESEARCH FINDS CHALLENGES IN SYMPTOM RECOGNITION AND DIAGNOSTIC TESTING CAN IMPACT PATIENT SATISFACTION FOR PEOPLE WITH VASCULAR DISEASE
– One in four physicians surveyed feel the “lack of technology or equipment to make an accurate diagnosis of coronary artery disease and peripheral artery disease” is a key obstacle to an accurate diagnosis – Over a third of surveyed patients with Coronary Artery Disease (CAD) or Peripheral Artery Disease […]
AngelMed Announces CMS Approval of Transitional Pass-Through (TPT) Payment Category For The Guardian™ Effective January 1, 2022
EATONTOWN, N.J., Nov. 11, 2021 /PRNewswire/ — Angel Medical Systems, Inc., (dba AngelMed) a company focused on improving long-term management of high-risk coronary disease in patients who have survived one or more heart attacks, announced today the Center for Medicare & Medicaid Services (CMS) has granted The Guardian™ implantable acute coronary syndrome […]
US FDA accepts supplemental New Drug Application and grants Priority Review for Jardiance® for adults with heart failure independent of left ventricular ejection fraction
If approved, Jardiance would be the first clinically proven treatment for adults across the full spectrum of heart failure regardless of ejection fraction RIDGEFIELD, Conn. and INDIANAPOLIS, Nov. 11, 2021 /PRNewswire/ — The U.S. Food and Drug Administration (FDA) has accepted a supplemental New Drug Application (sNDA) and granted Priority Review for Jardiance® (empagliflozin) 10 […]
BioCardia Reports Third Quarter 2021 Financial Results and Business Highlights
SAN CARLOS, Calif., Nov. 10, 2021 (GLOBE NEWSWIRE) — BioCardia, Inc. [Nasdaq: BCDA], a developer of cellular and cell-derived therapeutics for the treatment of cardiovascular and pulmonary diseases, today reported financial results for the third quarter of 2021 and filed its quarterly report on Form 10-Q for the three and nine months […]
Windtree Therapeutics Reports Third Quarter 2021 Financial Results and Provides Key Business Updates
WARRINGTON, Pa., Nov. 10, 2021 (GLOBE NEWSWIRE) — Windtree Therapeutics, Inc. (NasdaqCM: WINT), a biotechnology company focused on advancing multiple late-stage interventions for acute cardiovascular and pulmonary disorders, today reported financial results for the third quarter ended September 30, 2021 and provided key business updates. “I am very pleased to […]
Stereotaxis Reports 2021 Third Quarter Financial Results
ST. LOUIS, Nov. 11, 2021 (GLOBE NEWSWIRE) — Stereotaxis (NYSE: STXS), the global leader in innovative robotic technologies for the treatment of cardiac arrhythmias, today reported financial results for the third quarter ended September 30, 2021. “We are proud of the progress made on multiple fronts in the third quarter,” said David […]