BILLERICA, Mass.–(BUSINESS WIRE)–Quanterix Corporation (NASDAQ: QTRX), a company digitizing biomarker analysis with the goal of advancing the science of precision health, today announced that it has appointed Shawn Stetson as the Company’s interim chief financial officer, effective May 11, 2021. Mr. Stetson will assume his new responsibilities upon the departure of […]
Other News
BioStable Science & Engineering Announces CE Mark Approval for the HAART 200 Aortic Annuloplasty Device & Surpasses 1,000 Patients Treated Worldwide with the HAART Devices
AUSTIN, Texas–(BUSINESS WIRE)–BioStable Science & Engineering, Inc. (“BioStable”) announced today the company has received CE Mark approval for the HAART 200 Aortic Annuloplasty Device for use during bicuspid aortic valve repair. With CE Mark approval of both the HAART 300 and HAART 200 Aortic Annuloplasty Devices, BioStable will be able to […]
Bluegrass Vascular Announces Publication Reporting Experience with Cardiac Resynchronization Therapy Device Lead Placement following Use of Surfacer System
San Antonio, TX – April 21, 2021 – Bluegrass Vascular Technologies, a private medical technology company focused on innovating lifesaving devices and methods for vascular access procedures, announced today the publication of a report documenting the clinical utility of the Surfacer System to gain venous access prior to the placement […]
Vivasure Medical Begins Clinical Evaluation of Next-Generation PerQseal+ Device
First patient enrolled in Frontier V study evaluating enhanced fully absorbable, patch-based large-bore percutaneous closure device GALWAY, Ireland–(BUSINESS WIRE)–Vivasure Medical®, a company pioneering novel fully absorbable technology for percutaneous vessel closure, today announced the first patient was enrolled in the Frontier V study, a European multicenter study evaluating PerQseal®+, the […]
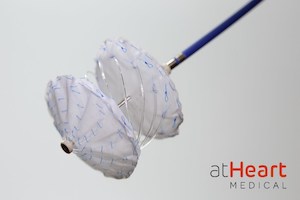
Newly Formed Company, atHeart Medical, Aims to Evolve Septal Closure with the reSept™ ASD Occluder
Company prepares for U.S. IDE clinical trial of its novel metal-free frame designed to reduce complications and preserve long-term treatment options BAAR, Switzerland and MOUNTAIN VIEW, Calif., April 20, 2021 /PRNewswire/ — atHeart Medical, a medical device company dedicated to establishing the new standard of care for closure of atrial septal defects (ASD), announced today that […]
Ra Medical Systems Provides Enrollment Update for its Pivotal Atherectomy Clinical Trial
42 total subjects enrolled as of April 19, 2021 12 subjects enrolled since mid-March CARLSBAD, Calif.–(BUSINESS WIRE)–Ra Medical Systems, Inc. (NYSE American: RMED), a medical device company focused on commercializing excimer laser systems to treat vascular and dermatological diseases, announces an increase in enrollment in its pivotal clinical trial to […]
ABBOTT REPORTS FIRST-QUARTER 2021 RESULTS
– Sales growth of 35.3 percent; organic sales growth of 32.9 percent – GAAP diluted EPS growth from continuing operations of 233.3 percent; adjusted diluted EPS growth of 103.1 percent – All four major businesses achieved strong sales growth in the quarter – Projected full-year EPS remains unchanged; reflects growth […]
MyoStrain® Provides Comprehensive Assessment of Myocardial Deformation to Help Physicians Risk-Stratify Heart Failure Patients for Individualized Cardiac Treatment
MyoStrain provided a uniquely sensitive and reproducible measure of segmental heart function to help physicians quantify differences in heart failure patients and risk-stratify patient populations based on the individual extent and severity of dysfunction MORRISVILLE, N.C., April 19, 2021 /PRNewswire/ — A new article published in the European Society of Cardiology (ESC) Heart […]
Zynex Appoints Donald Gregg as Vice President of Sales and Operations of Monitoring Solutions Division
ENGLEWOOD, Colo., April 20, 2021 /PRNewswire/ — Zynex, Inc. (Nasdaq: ZYXI), an innovative medical technology company specializing in the manufacture and sale of non-invasive medical devices for pain management, stroke rehabilitation, cardiac monitoring and neurological diagnostics today announced that it has hired Donald Gregg as the Vice President of Sales and Operations of its Monitoring Solutions Division. In […]
IN.PACT™ AV Drug-Coated Balloon Is First and Only to Show Superior and Sustained Results Through Two Years Compared to PTA in Treating Arteriovenous Fistulae Lesions
IN.PACT AV Access Trial 24 Month Results Presented as a Podium First at Charing Cross Symposium DUBLIN and LONDON, April 20, 2021 /PRNewswire/ — Medtronic plc (NYSE:MDT), the global leader in medical technology, today announced the safety and effectiveness results through 24 months for the IN.PACT AV Access clinical study. The data, which were […]